Reinthaler M1,2,3,4,*, Braune S1,2,4, Curio J3, Fröhlich G3,4, Jung F1,2,4 and Landmesser U1,3,4
1Institute of Biomaterial Science, Berlin-Brandenburg Centre for Regenerative Therapies (BCRT), Germany
2Helmholtz Institution, Kantstrasse 55, Teltow, Germany
3Department for Cardiology, Charité Universitätsmedizin Berlin, Campus Benjamin Franklin; Berlin Institute of Health, Deutsches Zentrum für Herz-Kreislaufforschung, Berlin, Germany
4Joint Laboratory, Kantstrasse 55, Teltow, Germany
- Corresponding Author:
- Markus Reinthaler
Charité Universitätsmedizin Berlin
Campus Benjamin Franklin, Department for Cardiology,
Hindenburgdamm 30, 12200 Berlin
Tel: +49 30 450539418
E-mail: markus.reinthaler@gmx.at
Received date: Apr 21, 2015, Accepted date: May 24, 2016, Published date: May 26, 2016
Citation: Reinthaler M, Braune S, Curio J, Fröhlich G, Jung F, et al. Trans-Catheter Pulmonary Valve Implantation: Procedure, Limitations and Future Directions. Interv Cardiol J 2016, 2:1.
Keywords
Trans-catheter pulmonary valve; Patch
repaired RVOT; Joint laboratory
Introduction
The indications for treatment of pulmonary valve stenosis
and incompetence are based on published guidelines (Tables 1 and 2) and reflected in the treatment indications used by many authors [1,2]. In terms of feasibility for TPVI there has
been a change during the past years. Initially, TPVI was
advocated only for previously operated patients with surgical
conduits.
Treatment indication in asymptomatic patients with normal RV function when:
- Systolic gradient > 50 mmHg (infants < 5 years)
- Systolic gradient > 40 mmHg (infants > 6 years) |
| Special care needs to be taken to the pathology of the pulmonary valve as this may have implications on the type of procedure performed (commissurotomy, separation of fused valve leaflets, “shaving” of thickened leaflets, moderate annuloplasty, resection of infundibular/muscular/subvalvular parts of the stenosis) |
| Primary treatment is balloon valvuloplasty |
| Secondary treatment is surgical approach after failure of valvuloplasty or |
| Trans-catheter pulmonary valve implantation (TPVI) |
| Recommendations according to the German guidelines of pediatric cardiology [1] |
Table 1: Treatment indications for pulmonary valve stenosis
| Regurgitation fraction >30% (measured by MRI), in addition to: |
| - Clinical symptoms: apparent right heart failure, impaired exercise performance (VO2 max <70%) |
| - New onset/persistent and symptomatic atrial or ventricular arrhythmia |
| - Significant or progressive right ventricular dilatation (EDV160, ESV 80 ml/m2) |
| - Significant or progressive right ventricular dysfunction (EF <45%) |
| - New onset significant tricuspid regurgitation with RV dilatation |
| - QRS>180 ms or increase of QRS >3.5 msec/year |
| Pulmonary regurgitation is NOT a benign structural cardiac problem. It has major impact on survival and quality of life. Reconstruction or valve replacement is the treatment of choice. Recommendations according to the german guidelines of pediatric cardiology [1] |
Table 2: Treatment indications for pulmonary valve regurgitation
The larger diameter of the SAPIEN valves and the use of prestenting,
nowadays allow treating many more types of outflow
tracts, whether there has been a conduit, a trans-annular
patch, a surgical valve or a native pulmonary valve [3].
Medtronic Melody Valve
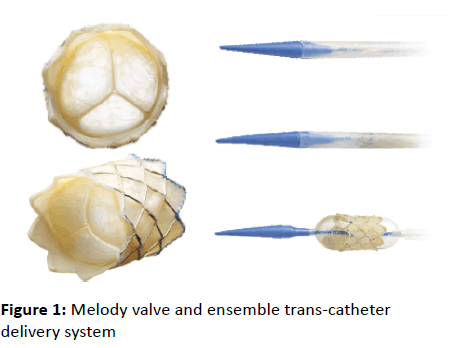
The Medtronic Melody valve is a balloon-expandable valve,
which is available in two sizes; 16 mm and 18 mm. The Device
is made of a bovine jugular vein valve (Contegra pulmonary
valved Conduit, Medtronic Inc., Minneapolis, MN, USA),
sutured within a Cheatham Platinum stent (CP stent, NuMED
Inc., Hopkinton, NY). The CP stent has a closed cell design.
Three different dedicated balloon in balloon delivery
catheters (Ensemble Trans-catheter Delivery System, 22F)-
available in 18, 20 and 22 mm outer balloon size-allow precise
deployment according to RVOT diameters (Figure 1).
Figure 1: Melody valve and ensemble trans-catheter
delivery system
Trans-catheter pulmonary valve replacement with the
Melody valve is a safe and effective treatment for patients
with post-operative RVOT conduit dysfunction [4]. It has been
proven to relief conduit obstruction, to restore valve function,
to delay the next surgical intervention and to significantly
improve functional status as well as quality of life [2,4-6]. Most
recently, data from the French registry demonstrated a
sustained improvement of right ventricular (RV) function [7,8].
Edwards Pulmonic Valve
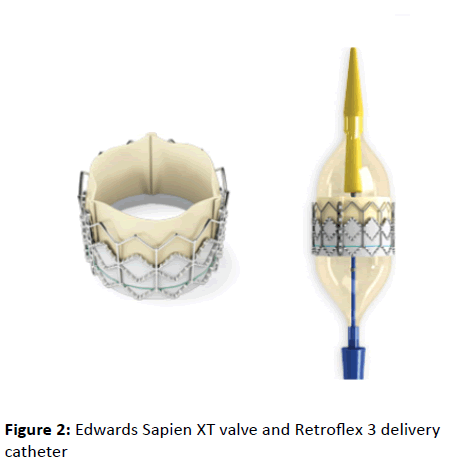
The Edwards Sapien valve is also a balloon-expandable
valve, which is available in a 23 mm and a 26 mm size
(covering RVOT dimensions from 21-23 and 23-26 mm). There
is also a 29 mm valve available, which is currently not cleared
for use in pulmonic position. However, some experience is
available with the larger valve, which demonstrated that it may
be feasible for RVOT diameters up to 29 mm [8]. The Edwards
valve is implanted through the Retroflex delivery system,
which consists of a balloon catheter and a deflectable guiding
catheter (Figure 2).
Figure 2: Edwards Sapien XT valve and Retroflex 3 delivery
catheter
It requires a 22 Fr introducer sheath for the 23 mm and a 24
F sheath for the 26 mm valve, presuming the valve is crimped
on the catheter and loaded on the balloon after passing the
introducer sheath. Access from the jugulary vein may even
require larger sheath sizes, as mounting the valve directly on
the balloon may be necessary. However, there is also some
experience with the latest Edwards trans-catheter aortic valve
implantation (TAVI) technology in pulmonary position, the
Sapien III valve, which is implantable through a 14 F e-sheath
[9].
Two recent large studies investigated the feasibility and
safety of the Sapien valves in pulmonic position. The
compassion trial, which is still ongoing, included data from 36
patients with follow up of up to 4 years, showed a procedural
success rate of 85% with no mortality after 6 months [10].
These results are confirmed by the most recent Data of the
PREMIER Registry, which included 131 patients in 16 centers and also showed no mortality and a 91% procedural success
rate after one year [11]. Both studies confirm good
hemodynamic and functional results throughout the follow-up
period.
Procedural Considerations
Congenital heart disease patients usually have a history of
complex surgical interventions. However, trans-catheter
technologies have expanded over the past few years, and
nowadays serve as important complementary treatment
modalities. Therefore, every type of surgery performed in
childhood, should take into consideration possible
percutaneous procedures a patient may need to undergo in
the future. For example, RVOT morphology is different in patch
and conduit repair and may therefore influence the choice of
equipment and technique in case of TPVI. Great care needs to
be paid to the coronary anatomy, due to the risk of external
compression after RVOT stenting. According to previous
published data, 5% of patients requiring a percutaneous
pulmonary valve were not suitable for this procedure due to
an increased risk of coronary artery compression [12].
In general, coronary vessels at risk are detected by
simultaneous coronary angiography and inflation of a large
non-compliant balloon in the RVOT. The balloon size should be
large enough to cause complete abolition of systemic pressure
after inflation. This will ensure adequate simulation of the
external RVOT and pulmonary artery dimensions after
percutaneous repair. Pre-stenting the RVOT with covered or
open cell stents is recommended in all patients to improve the
landing zone of the trans-catheter valve and to minimize
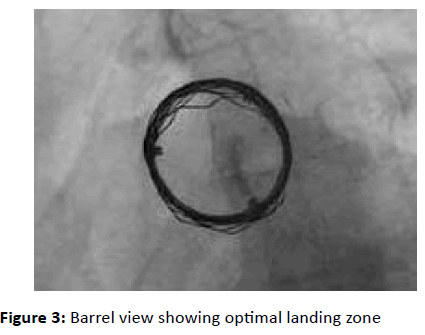
residual gradients [13]. The stent formed “conduit” needs to
be circular to enable optimal acute and longterm function of
the implanted valve as leaflet coaptation could be impaired by
non-circular deployment. A ‘down the barrel’ view is ideal to
visualize the anatomy (Figure 3).
Figure 3: Barrel view showing optimal landing zone
Balloon sizing with low pressure balloons may help to
determine the RVOT diameter and subsequent size of stents
selected for preparation of the landing zone of the valve. In
outflow tracts with severe calcification, high pressure and
shear resistant balloons (i.e. Atlas balloons) may be used.
Keeping in mind that a normal RVOT in adults usually
measures about 25 mm in diameter, the valve size should be
chosen accordingly for the first implant in a patient if possible
[14,15]. A sub-investigation of the Framingham heart study
revealed mean pulmonary artery diameters of 26 ± 2.7 mm in
men and 24 ± 2.7 mm in women [15]. Once these steps are
completed, the trans-catheter valve can be delivered and
deployed, preferably at the position of the native valve.
Important attention needs to be paid to the orientation of the
valve when mounted on the delivery catheter. An overview of
procedural steps, along with the generally used technical
equipment is provided in Table 3.
| Procedural steps |
Equipment |
| Accessing pulmonary artery |
Standard catheters |
| Acquiring hemodynamics and angiograms |
Standard pigtail or Multitrack catheter (NuMED, Inc.) for good visualization. |
| Balloon sizing, Pulmonary valve valvuloplasty, exclusion of coronary obstruction |
In the presence of pulmonary valve stenosis, stiffer wires (e.g. COOK ultrastiff wire) and medium pressure balloons (e.g. Mullins-X, Z-med, Zmed II - NuMED Inc., VACS III - Osypka GmbH) may be used for dilatation
If severe calcification is visible, a predilation with robust balloons, i.e. Atlas Balloons (BARD) is often necessary
In large outflow tracts, moderate pressure balloons are recommended (e.g. PTS, PTS-X sizing balloon - NuMED Inc., Cristal balloon – Abmedica). For balloon interrogation a VACS II (Osypka GmbH) can be used. |
| RVOT Stenting |
Usually straight 14-16 F Check-Flo Performer Introducers and medium pressure balloons for stent deployment are used (e.g. Mullins-X, Z-med, Z-med II-NuMED, Inc., VACS III-Osypka GmbH or BiB balloon)
Covered CP-stents (NuMED, Inc.) may reduce blood extravasation; non-covered stents (i.e. CP Stents, Andra stents) are suitable in selected cases.
In wide, non-calcified RVOTs, Andra stents (Andramed, Inc.) are preferred by many operators since they may improve the “grip” on the vessel wall.
Preparation of a round landing zone for the TPV is important and can be checked in a barrel view (Figure 3) |
| Valve implantation |
23, 26 mm SAPIEN valve (Edwards Lifesciences, LLC) RVOT dimension: 21-26 mm
Melody valve (Medtronic, Inc.) RVOT dimension: 16-22 mm. |
Table 3: Procedural steps and equipment.
Complex Anatomies
TPVI implanters often face technically challenging anatomies
and limited availability of dedicated technical equipment.
These tricky situations include dilated RVOTs and the presence
of surgical pulmonic valves.
Annulus diameters of more than 26 mm are most likely
discovered in patients with previous trans-annular patch (TAP)
repair or in dilated native RVOTs. Patch-repaired pulmonary
arteries may be less expandable (dependent on the implanted
patch size) and the stent apposition area may be narrow. This
is why anatomy after RVOT patch repair is often considered
unfavorable for TPVI and is therefore not recommended by
current guidelines. However, a recent study showed that this
procedure may be feasible with the SAPIEN valves [16].
Furthermore, earlier rather than later TPVI, to prevent severe
RVOT dilatation, may be considered in these patients. Dilated
RVOT anatomies may result in an increased risk of valve
migration in case of immediate valve implantation. Therefore,
a staged ‘two sessions’ procedure (meaning that the valve
delivery is postponed for one to two months, until fixation of
the implanted stents has taken place) may be an additional
option to achieve the best possible results. However, this
approach may be inadequate in patients with poor right
ventricular function, as free PR is likely to cause rapid
hemodynamic deterioration in this cohort. Having the valve
prepared for immediate implantation may be the most
effective rescue strategy in case of rapid hemodynamic
deterioration after RVOT-stenting in these patients.
Most importantly, stent/valve sizing plays a major role in
avoiding stent/valve migration. Some operators favor balloon
interrogation, using a low pressure compliant balloon.
Applying a gentle tug to the inflated balloon may also help to
predict stent malapposition. If the stenosis disappears after
inflation or in case of balloon dislodgement after the tug, the
procedure should be aborted. Procedural data from 10 TPVIs
in trans-annular patches showed that good results were
achieved by slight oversizing of the balloon used for stent
deployment (Outer BiB balloon diameter to RVOT ratio=1.1)
[17]. Similar recommendations can be given in terms of valve
sizing. The RVOT diameter just above or below the balloon
waist should be chosen for valve sizing. For large anatomies
(>26 mm RVOT diameter) there is currently only the 29 mm
SAPIEN XT (or Sapien III) valve available (not CE marked for this
indication). Self-expandable stents may be used for prestenting,
if the RVOT is larger than the available balloons for
stent deployment. As mentioned earlier, a circular RVOT
should be prepared to allow full expansion of the implanted
heart valve. Post-dilatation of the implanted stents with
noncompliant balloons or implantation of a second or even
several stents may help to improve the results. Multiple stent
implantations may also be considered to downsize the RVOT
diameter and allow implantation of the largest available valve.
In general, the longest available stents (crimped on a BiB
balloon) should be used and placed as proximally as possible
to cover the RVOT. However, the use of multiple stents also has
serious risks including distortion of the previous stent, damage
to the pulmonic artery wall, and compromise of the main pulmonary branches, which has to be excluded after stentimplantation.
A large low pressure balloon may help to define
the correct implantation site. Compared to conventional
pulmonary angiography, this approach may improve flare and
will reduce the amount of contrast used.
Patients with previous bio-prosthetic surgical pulmonary
valve replacements also require a different approach.
Although the risk of coronary obstruction and annular rupture
is lower in some valve types, correct positioning of the transcatheter
valve, trans-valvular gradients and pulmonary
regurgitation may be a problem in these procedures. Prestenting
despite the presence of a surgical bio-prosthesis may
be considered to improve the landing zone especially when
the Melody valve is used, due to its longer Stent frame
compared to the Edwards devices. However, this approach
may increase the trans-valvular gradient, especially in small
stenotic valves. On the other hand, direct valve implantation
without pre-stenting may result in significant regurgitation due
to lack of leaflet-coaptation, caused by extreme overexpansion
of the distal part of the trans-catheter valve, which is not
caged in the surgical bio-prosthesis. Extensive oversizing
should therefore be avoided, especially in valve-in-valve
procedures. In conclusion, the design, size, early postoperative
gradients and degree of calcification of the surgical
valve, as well as careful valve sizing and positioning are major
components to achieve the best possible results.
Future Directions
Requirements for valve substitutes include features such as
a lower introducer profile <20 F, low inflammatory response,
long durability, low opening resistance with maximal valve
area, as well as fast and reliable closure and as for all
cardiovascular implants non-thrombogenicity. In addition, the
trans-catheter valve should be able to compensate for
suboptimal geometrical expansion in potentially oval-shaped
landing zones. Extensive oversizing is associated with
pulmonary regurgitation due to lack of leaflet coaptation,
implying that the availability of several valve sizes or even
patient specific valves is key to achieve good results. The latest
Edwards Sapien technology provides a broader range of sizes
(23-26-29 mm), lower profile (16-20 F e-Sheath), a more
flexible system and semi-closed resting position leaflet
configuration. A disadvantage compared to the Medtronic
Melody device is that once the Sapien valve has been
introduced into the vessel, it cannot be retrieved, except
removing the introducer sheath, until the valve is deployed.
Importantly, Stent-fractures, which impacts the durability of
the valve, is more likely with the Melody valve. Stent design
and the used materials will therefore play a key role in future
technologies. Furthermore, structural valve failure is also
caused by calcification which is histologically evident within 3
years of valve implantation [18]. Mechanical stresses including
mounting the valve on various catheters and distortion of the
valve or incomplete valve expansion have been identified as
risk factors for early valve failure. The trend to reduce sheath
sizes and consequently the reduction of leaflet thickness is
also known to impair the durability of percutaneous valves [18]. Self-healing materials may offer a solution to overcome
these limitations in the future [19].
Taken together, current trans-catheter valve technology is
moving in the right direction to achieve the best possible
outcomes. However, it is only a transient solution so far.
Upcoming technologies will offer new features which may
further improve the outcome of congenital heart disease
patients.
References
- https://www.kinderkardiologie.org/leitlinien/
- Eicken A, Ewert P, Hager A, Peters B, Fratz S, et al. (2011) Percutaneous pulmonary valve implantation: two-centre experience with more than 100 patients. Eur Heart J 32: 1260-1265.
- Haas NA, Moysich A, Neudorf U, Mortezaeian H, Abdel-Wahab M, et al. (2013) Percutaneous implantation of the Edwards SAPIEN pulmonic valve: initial results in the first 22 patients. Clin Res Cardiol 102: 119-128.
- Zahn EM, Hellenbrand WE, Lock JE, McElhinney DB (2009) Implantation of the melody transcatheter pulmonary valve in patients with a dysfunctional right ventricular outflow tract conduit early results from the U.S. Clinical trial. J Am Coll Cardiol 54: 1722-1729.
- McElhinney DB, Hellenbrand WE, Zahn EM, Jones TK, Cheatham JP, et al. (2010) Short- and medium-term outcomes after transcatheter pulmonary valve placement in the expanded multicenter US melody valve trial. Circulation 122: 507-516.
- Vezmar M, Chaturvedi R, Lee KJ, Almeida C, Manlhiot C, et al. (2010) Percutaneous pulmonary valve implantation in the young 2-year follow-up. JACC Cardiovasc Interv 3: 439-448.
- Fraisse A, Aldebert P, Malekzadeh-Milani S, Thambo JB, Piéchaud JF, et al. (2014) Melody® transcatheter pulmonary valve implantation: results from a French registry. Arch Cardiovasc Dis 107: 607-614.
- Wilson WM, Benson LN, Osten MD, Shah A, Horlick EM (2015) Transcatheter Pulmonary Valve Replacement with the Edwards Sapien System: The Toronto Experience. JACC Cardiovasc Interv 8: 1819-1827.
- Rockefeller T, Shahanavaz S, Zajarias A, Balzer D (2016) Transcatheter implantation of SAPIEN 3 valve in native right ventricular outflow tract for severe pulmonary regurgitation following tetralogy of fallot repair. Catheter Cardiovasc Interv 10: 1002.
- Kenny D, Hijazi ZM, Kar S, Rhodes J, Mullen M, et al. (2011) Percutaneous implantation of the Edwards SAPIEN transcatheter heart valve for conduit failure in the pulmonary position: early phase 1 results from an international multicenter clinical trial. J Am Coll Cardiol 58: 2248-2256.
- Ewert P, Al-Halees Z, Biernacka EK, Pongiglione G, Haas N (2014) One Year Follow-Up of the PREMIER Multicenter Registry for the Edwards SAPIEN Pulmonic Transcatheter Heart Valve: An Interim Report. 49th annual meeting of the association for European paediatric and congenital cardiology, Prague, Czech Republic.
- Morray BH, McElhinney DB, Cheatham JP, Zahn EM, Berman DP, et al. (2013) Risk of coronary artery compression among patients referred for transcatheter pulmonary valve implantation: a multicenter experience. Circ Cardiovasc Interv 6: 535-542.
- Peng LF, McElhinney DB, Nugent AW, Powell AJ, Marshall AC (2006) Endovascular stenting of obstructed right ventricle to pulmonary artery conduits: a 15 year experience. Circulation 113: 2598-2605.
- Koestenberger M, Avian A, Ravekes W (2013) Reference values of the right ventricular outflow tract (RVOT) proximal diameter in 665 healthy children and calculation of Z-score values. Int J Cardiol 169: 99-101.
- Truong QA, Massaro JM, Rogers IS, Mahabadi AA, Kriegel MF, et al. (2012) Reference values for normal pulmonary artery dimensions by noncontrast cardiac computed tomography: the Framingham Heart Study. Circ Cardiovasc Imaging 5: 147-154.
- Haas NA, Moysich A, Neudorf U, Mortezaeian H, Abdel-Wahab M, et al. (2013) Percutaneous implantation of the Edwards SAPIEN pulmonic valve: initial results in the first 22 patients. Clin Res Cardiol 102:119-128.
- Demkow M, Rużyłło W, Biernacka EK, Kalińczuk Ł, Spiewak M, et al. (2014) Percutaneous Edwards SAPIEN(™) valve implantation for significant pulmonary regurgitation after previous surgical repair with a right ventricular outflow patch. Catheter Cardiovasc Interv 83:474-481.
- Arsalan M, Mack MJ (2015) Durability of devices: long-term results and clinical outcomes. EuroIntervention 11: 119-122.
- Blaiszik BJ, Kramer SLB, Olugebefola SC, Moore JS, Sottos NR, et al. (2010) Self-Healing Polymers and Composites. Annual Review of Materials Research 40: 179-211.