Keywords
Antidiabetic activity; Toxicity; P. floridanus; P. pulmonarius
Introduction
Diabetes is a cardiovascular risk associated with chronic hyperglycemia in the blood [1]. In 2013, IDF estimated that 382 million people, or 8.3% of adults, had diabetes worldwide of which 46% are not diagnosed. In Africa, nearly 20 million people have diabetes, thus a prevalence of 4.9% for the region. These estimates are expected to double by 2035 to reach 41.5 million, hence increasing by 109% [2]. In Cameroon, diabetes is spreading with time; according to IDF in 2013, 93 thousand people age 60- 79 years have diabetes of which 229 thousand people are not diagnosed and by 2015 10% of the population suffering from diabetes was diagnosed [3]. Indeed, hyperglycemia increases the production of free radicals that can’t be inhibited by the body. This will thus cause oxidative stress in the cell with decreased enzymatic antioxidant defenses resulting in diabetes [4]. Antioxidant supplementation is therefore essential as it helps in inhibiting the harmful action of free radicals on insulin secretion, thus balancing the glycemic index [5]. From literature review, studies on mushroom reveal that they are very good antioxidants and thus prevent from certain metabolic diseases like diabetes [6]. Cameroon is full with a diversified flora of vegetables among which the mushrooms and precisely oyster mushroom which belongs to kingdom Fungi [7]. But, in the culinary context, oyster mushrooms can be considered as vegetables. They are very good antioxidants but there is little scientific data available in our country concerning the use of oyster mushrooms in diabetes prevention hence in order to contribute in the management of diabetes, our interest was carried on their study.
Research hypothesis
The species of mushrooms P. pulmonarius and P. floridanus consumed in Cameroon would have a mechanism of action such as the hypoglycemic activity that can be exploited in the treatment of diabetes.
General objective
To evaluate the toxicity and antidiabetic activity of two commonly consumed mushroom species in Cameroon: P. pulmonarius and P. floridanus.
Materials and Methods
In vitro study: Preparation of experimental plant material
Mushroom harvest: The fresh mushrooms harvested in a myciculture in Pk 21 A suburb of the city of Douala were shade dried in large pans for 3 days and then crushed in a mixer to obtain mushroom powder.
Preparation of crude mushroom extracts: The alcoholic and hydroalcoholic extracts of Pleurotus pulmonarius (P. pulmonarius) and the aqueous extract of Pleurotus floridanus (P. floridanus) were prepared in the Biochemistry Laboratory of the University of Douala. For each extract, 100 g dried mushroom powder were macerated in 800 mL of solvent (proportion=1:8) ethanol, water/ ethanol (1:1) and distilled water, respectively. The extracts were then filtered with a filter paper and the filtrate obtained was dried in an oven (BINDER mark) for 4 days at 45°C in order to obtain the crude extracts.
In vivo study: Evaluation of the toxicity for the various extracts on rats
Physical and behavioral evaluation: The protocol of the limit test proposed by the OECD in 2008 [8] was used to evaluate the acute toxicity of mushroom extracts. This protocol recommends the administration of a single dose (2000 mg/kg body weight: BW) of extract to a first experimental animal (rodent) followed by observation of the physiological variations for 48 hours. If it survives, 4 other animals are added and are given the extract at 2000 mg/kg BW. The observation of the physiological variations of the animal in this case takes place for two weeks.
Evaluation of the state of organs: At the end of these two weeks of observations, the female rats were then sacrificed in order to better compare their organs to that of the controls negative and certain toxicity markers such as transaminases (ASAT and ALAT) were determined.
Evaluation of the effect of administration the mushroom extracts on body weight and fasting blood glucose of diabetic rats during 28 days of treatment
Experimental induction of diabetes by streptozotocin (STZ): This study evaluated the long-term hypoglycaemic capacity of mushroom extracts in diabetes induced adult male rats (three months of age). Their weights ranged from 210 g to 250 g. After an overnight fasting (food deprivation for 12 hours with water as libitum), diabetes was induced in rats by intraperitoneal injection of a freshly prepared solution of STZ at 55 mg/kg of body weight, i.e., a volume of 2 mL/kg (dose at which the β-cells are destroyed). The STZ was dissolved in 0.1 M sodium citrate buffer pH 4.5 [9]. Negative control received the same volume of 0.1 M sodium citrate buffer pH 4.5 through intra peritoneal route. After injection, drinking water was replaced with 5% glucose solution for 24 hours in order to overcome STZ-induced hypoglycemia following the destruction of pancreatic β cells and the massive release of insulin. This hypoglycemia can be letal for rats. [10]. After 48 hours of injection (diabetes development time), diabetes was confirmed in rats by measurement of fasting blood glucose using an SD-type glucose meter codeFree. Only rats with a blood glucose level greater than 2 g/L were considered diabetic and retained for this study.
Evaluation of the hypoglycemic potential of extracts in diabetic rats
Animal treatment: After induction of diabetes, all diabetic and non-diabetic rats were divided into six groups of five rats each and kept under the same conditions. The treatment with mushroom started 24 hours after confirmation of diabetes and was evaluated for 28 days.
Distribution of Animals
• Group I: Healthy controls or negative control (NC) that received daily gavage 5 mL/kg BW distilled water for 28 days.
• Group II: Diabetics control or positive control (PC) which received each day 5 mL/kg BW distilled water by gavage for 28 days.
• Group III: Diabetics treated with the hydroalcoholic extract of P. pulmonarius (Phal), which received 400 mg / kg BW of the extract every day by gavage for 28 days.
• Group IV: Diabetics treated with the alcoholic extract of P. pulmonarius (Pal) which received 400 mg/kg BW of the extract every day by gavage for 28 days.
• Group V: Diabetics treated with the aqueous extract of P. floridanus (Faq), which received 400 mg/kg BW of the extract daily by gavage for 28 days.
• Group VI: Standard which received 0.3 mg/kg BW glibenclamide daily by gavage.
Determination of blood glucose and weight: Blood glucose was determined by puncturing the distal tip of the tail using a lancing device. The blood collected on the glucose meter via the test strip allowed us to obtain the blood glucose for the rats. This was measured at one week interval during the 28 days of treatment more precisely on days 0 (24 h after the confirmation test for diabetes), 8, 15, 22 and 29. Glycemia was measured each time after 12 h fasting [10]. Similarly, weight measurement was conducted on a regular basis in fasting rats each week just prior to blood sampling.
Statistical analysis: Data were introduced in a Excel Worksheet (Microsoft Office 2007) then analyzed with the software Statview version 5.0. Data were presented in a percentage and average ± standard differential for qualitative variables and quantitative respectively. The significance was set at 5%.
Results
Evaluation of toxicity parameters of extracts
Observation of signs of poisoning: Two weeks after receiving various extracts at 2000 mg/kg of body weight of the different extracts, no sign of toxicity was observed (Table 1). From Table 1, we may say that no particular sign of intoxication, both behaviorally and physically, was observed in the rats given the extract. Also, during this period of toxicity assay, no deaths were recorded. This lack of pathological evidence may be due to the fact that mushrooms of the oyster species are not harmful [11]. However, other parameters such as organ observation and the determination of biochemical enzymes have been evaluated in order to confirm this.
| Sign of poisoning |
Faq |
Phal |
Pal |
Negative control |
| Locomotion |
Normal |
Normal |
Normal |
Normal |
| Activity reduced |
- |
- |
- |
- |
| Stool appearance |
Not significant |
Not significant |
Not significant |
Not significant |
| Aspect of urine |
Normal |
Normal |
Normal |
Normal |
| Sensitivity to noises |
+ |
+ |
+ |
+ |
| Appetite |
+ |
+ |
+ |
+ |
| Difficulty of breathing |
- |
- |
- |
- |
| Coat |
Normal |
Normal |
Normal |
Normal |
| Number of death |
0 |
0 |
0 |
0 |
+: Affirmative or yes; -: Negative or Not.
Table 1 Variation of the signs of toxicity depending on extracts.
Determination of biochemical enzymes: Though no signs of toxicity were detected from animals’ behavior and macroscopic organ observation, the biochemical enzyme assay was evaluated to ensure that there was no organ cytolysis (Figure 1). The values of ASAT and ALAT of our samples, compared to the negative control were not significantly different P >0.05. This means that no organ cytolysis has been observed and therefore the heart, liver, muscles, kidneys, brain and pancreas have not been significantly degraded. These results confirm those obtained by Ninkwango in 2007 [11] which showed that fungal mushrooms had no toxicity.
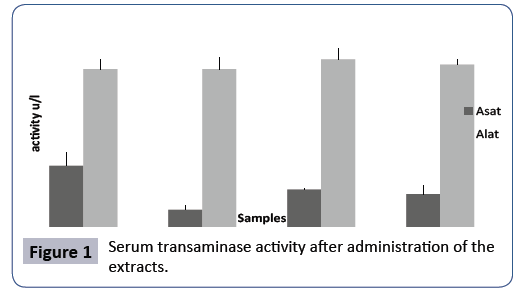
Figure 1: Serum transaminase activity after administration of the extracts.
Evaluation of the effect of mushroom extracts on fasting blood glucose of diabetic rats
Evaluation of the weight evolution during the 28 days treatment of the diabetes with mushrooms extracts: Figure 2 shows that there is an increase in weight for healthy rats or normal control (NC) time, from 250.3 ± 21.70 g to 272.3 ± 21.36 g (thus, an increase of 22 g) at the end of the 28 days. All the other groups, with the exception of the Faq extract, show a slight decrease in weight over time. Except for the DC, there was no significant difference (p<0.05) between the various tests groups and the NC. There was a significant difference in the untreated diabetic control, also referred to as DC, in the third weeks (22 days) and in the fourth weeks (29 days). On day 22, there was a significant difference of 195.775 ± 17.49 g in the DC compared to 270.25 ± 20.02 g in the NC (a difference of 74.475 g). Also, on day 29, there was a significant difference of 185.125 ± 14.06 g in the DC compared 272.3 ± 21.36 g in the NC (A difference of 87.175 g). The NC curve thus appears to be the most significant curve over time, with the DC curve showing a lower weight over time and the control curve representing the batch of diabetic rats treated with the reference, it shows a slight decrease over time, as do extracts, but it does have a greater weight than extracts over time. With, the curve of the extract Pal being merged with the curve of the extract Phal. As for DC, our results are in accordance with those reported by Pari and Latha [12] who found that in male rats of Wistar albino strain, injection of STZ caused a significant weight loss within three weeks. They indicated that these conditions are due to the excessive loss of tissue proteins. Similarly, Sathishsekar and Subramanian [13] and Taleb-Senoucia and et al. [14] also reported similar results and suggest that body weight loss in the untreated diabetic group can be explained by lipids and structural proteins catabolism due to lack of hydrated carbons that are used as energy source. For the Phal and pal extracts, although they do not show a significant decrease for p<0.05, the slight decrease observed could also be due to a partial catabolism of lipids and structural proteins. Thus, these extracts do not exhibit a high activity, such as the Faq extract, where weight increase is observed after the third weeks. The strong activity of the Faq extract to limit weight loss better than the other extracts could be due to the fact that it would exhibit a better antioxidant activity than the other extracts. The ability of our extracts to protect diabetic rats from the massive loss of body weight could thus be due to their ability to limit the catabolism of lipids and proteins [15-17] and thus their ability to reverse gluconeogenesis and control protein loss [18,19].
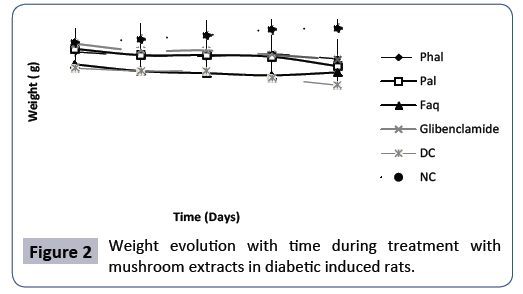
Figure 2: Weight evolution with time during treatment with mushroom extracts in diabetic induced rats.
Assessment of blood glucose changes during the 28 days of treatment of diabetes with mushroom extracts: In order to better evaluate the evolution of blood glucose during the treatment period, it should be noted that at the beginning of experimental period, there was no significant difference in blood glucose for all groups. Also, after induction of chronic hyperglycaemia, precisely at time 0, with the exception of the normal control (NC), there was no significant difference between the other batches of rats. Thus, the analysis in Figure 3 below present six curves showing the evolution of blood glucose levels in controls and trials during the treatment of diabetes. This figure shows that the NC groups have the lowest values for blood glucose throughout the experimental period, ranging from 0.70 ± 0.05 to 0.71 ± 0.05 g/L, which is normal because in fasting healthy subjects, blood glucose according to the standard should be between 0.7 and 1.1 g/L. From the figure, the mushroom extracts show a better hypoglycemic activity than standard during the first 2 weeks and this effect continued to be observed for the next two weeks (3ᶦᵈand 4áµÂâ€â€Âᵈ) only with the Faq extract. However, there was a significant decrease in blood glucose levels in weeks 1 and 3 in rats receiving the extracts and standard group. On the other hand, the DC showed a significant increase in blood glucose levels during the first two weeks of treatment 2.8 ± 0.39 to 4.03 ± 0.54 g/L. From the third weeks, there is a decrease in blood glucose up to the fourth weeks. Figure 3 shows that at the end of the 28 days of treatments, the Faq, Pal extracts and the reference glibenclamide, showed hypoglycemic activity with glycemia variation being respectively from 3.49 ± 0.32 to 1.59 ± 0, 67 g/L, 3.24 ± 0.24 ± 2.5 ± 0.55 g/L and 3.77 ± 0.22 ± 2.5 ± 0.68 g/L. The Phal extract, on the other hand, at the end of the 28 days shows a slight rise in blood glucose from 2.99 ± 0.72 to 3.15 ± 0.27 g/L. The Faq extract is thus the best hypoglycemic extract. The hypoglycaemic activity of the Faq and Pal extracts could be explained by the fact that these extracts could act in the same way as some oral antidiabetics by restoring the damage caused by streptozotocin. Indeed, the hypoglycemic effect observed in these extracts could be due to the fact that the latter inhibited the release of the reactive species of oxygen caused by streptozotocin. This led to a closure of the K+/ATP channels followed by membrane depolarization of the beta cells with stimulation of the Ca2+ inflow, which represents the first key step for insulin secretion [12]. Thus, the hypoglycemic effect of the Faq and Pal extracts can be assimilated to both the organic and the inorganic constituents. It is important to note that the inorganic constituents that medicinal plants contain sometimes play an important role in improving their medicinal properties, including hypoglycaemic activity [10]. Bhaskar and et al. [19], who studied the hypoglycemic effect of aqueous mucuna prurien extract (200 mg/kg) in rats rendered diabetic by STZ, reported that a number of essential minerals such as Na, K, Ca, Zn, Mg, Fe, Cu and Mn may be associated with an insulin release mechanism and its activity. Since the floridanus species is richer in minerals than the pulmonarius species according to Bhaskar et al. [19], this may explain why the Faq extract exhibits the best hypoglycaemic activity.
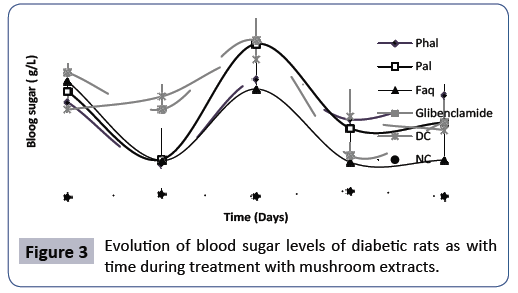
Figure 3: Evolution of blood sugar levels of diabetic rats as with time during treatment with mushroom extracts.
Conclusion
At the end of our study which consisted in evaluating the toxicity and antidiabetic activity of two species of mushroom commonly consumed in Cameroon, namely P. pulmonarius and P. floridanus, it was found that the alcoholic and hydroalcoholic extracts of P. pulmonarius and the aqueous extract of P. floridanus revealed no significant signs of toxicity. The antidiabetic test carried out after injection of the streptozotocin shows that the Faq and Pal extracts exhibit good antidiabetic activity with the Faq extract as the best hypoglycemic extract.
With regard to the above results, we can say that the extracts of P. pulmonarius and P. floridanus exhibit antidiabetic activity. This suggests the use of these fungi as functional food. We thus envisage for a future research to carry out a phytochemical screening of these extracts in order to know the compounds which would be at the origin of the antidiabetic activity of these extracts.
Acknowledgments
The authors would like to thank the Laboratory of Biochemistry of the Faculty of Sciences of the University of Douala where most of this work has been done.
References
- Harris M, Zimmet P (1997) Classification of diabetes mellitus and other categories of glucose intolerance. Alberti K, Zimmet P, Defronzo R, editors. International Textbook of Diabetes Mellitus,(2nd edn.), Chichester: John Wiley and Sons Ltd; 1997,pp: 9-23.
- IDF (2013) Diabetes Atlas, Sixth edition.
- Camerpost (2015) Cameroon tribune by Yvette MBASSI-BIKELE; 37% of the population have high blood pressure in Cameroon.
- Mohora M, Greabu M, Muscurel C, DuŃă C, Totan A (2007) The sources and the targets of oxidative stress in the etiology of diabetic complications. Romanian J Biophys 17: 63-84.
- Sharma A, Kharb S, Chungh SN, Kakkar R, Singh GP (2000) Evaluation of Oxidative Stress Behavior and After Control of Glycemia and After Vitamin E Supplementation in Diabetic Patients. Metabolism 49: 160-162.
- Dubost NJ, Or B, Beelman RB (2007) Quantification of polyphenols and ergothioneine in total antioxidant capacity. Food Chemistry.
- Courtecuisse, Duhem B (2011) Guide to mushrooms of France and Europe. 1752 species described and illustrated. Paris: Delachaux and Niestle.
- Organization for Economic Cooperation and Development: OECD (2009) Emission scenario document on plastics additives. Paris (FR): OECD Environment Directorate. Series on Emission Scenario Documents.
- Crouch R, Kimsey G, Priest DG, Sarda A, Buse MG (1978) Effect of streptozotocin on erythrocyte and retinal superoxide dismutase. Diabetologia 3:53-57.
- Bouldjadj (2009) Study of the antidiabetic and antioxidant effect of lyophilized aqueous extract of Artemisia herba alba Asso in healthy rats and rats rendered diabetic by streptozotocin. Memory of the Magisterium in Cellular and Molecular Biology. University Mentouri Constantine, p: 36.
- Ninkwango Temoka Antoine (2007) The cultivation of oyster mushrooms. Cameroon (2nd edn.), p: 47.
- Pari L, Latha M (2005) Antidiabetic effect of Scoparia dulcis: effect on lipid peroxidation in streptozotocin diabetes. Gen Physiol Biophys 24: 13-26.
- Sathishsekar D, Subramanian S (2005) Antioxidant properties of Momordica Charantia (bitter gourd) seeds on Streptozotocin induced diabetic rats. Asia Pac J Clin Nutr 14: 153-158.
- Taleb-Senoucia D, Ghomaria H, Kroufa D, Bouderbalaa S, Prostb J, et al. (2009) Antioxidant effect of Ajuga iva aqueous extract in streptozotocin-induced diabetic rats. Phytomedicine 25: 221-235.
- Chen YQ (1998) Sp1 sites mediate activation of the plasminogen activator inhibitor-1 promoter by glucose in vascular smooth muscle cells. J Biol Chem 273: 8225-8231.
- Al-Shamaony L, Al Khazraji MS, Twaij HA (1994) Hypoglycemic effects of Artemisia herba-alba. II. Effect of a valuable extract on some blood parameters in diabeticanimals. J Ethnopharmacol43: 167-171.
- Tastekin D, Atasever M, Adigüzel G, Keles M, Tastekin A (2006) Hypoglycaemic effect of Artemisia herba-alba in experimental hyperglycaemic rats. Bull Vet Inst Pulawy 50: 235-238.
- Rajagopal K, Sasikala K (2008) Antihyperglycaemic and antihyperlipidaemic effects of Nymphaea stellata in alloxan-induced diabetic rats. Singapore Med J 49: 137-142.
- Bhaskar A, Vidhya VG, Ramya M (2008) Hypoglycaemic effect of Mucuna pruriens seed extract on normal and streptozotocin-diabetic rats. Fitoterapia 79: 539-543.