- (2013) Volume 14, Issue 4
Georgios Michael Oikonomopoulos1, Konstantinos N Syrigos2, Muhammad Wasif Saif3
1Second Oncology Department, St. Savvas Anticancer Hospital; 2Oncology Unit, Third Department of Medicine, Sotiria General Hospital, Athens Medical School. Athens, Greece.
3Department of Medicine and Cancer Center, Tufts Medical Center. Boston, MA, USA
Pancreatic cancer is a frequent and lethal disease ranking fourth as a cause of cancer-related death in Western countries.There are patients, though, who respond well to chemotherapy and have a prolonged survival. There is an effort towardsidentification of specific characteristics of these tumor cells in order to identify those patients who will benefit fromchemotherapy and use them as prognostic or predictive factors. This review is an update on the 2013 American Society ofClinical Oncology (ASCO) Annual Meeting regarding the most important developments in this field for pancreatic cancer, asthey were reported in Abstracts #4006, #4016, #4046, and #4060 and a discussion is presented about their application inclinical praxis.
Antigens, CD3; Antigens, CD4; Antigens, CD8; Equilibrative Nucleoside Transporter 1; Neoplastic Cells, Circulating; Osteonectin; Pancreatic Neoplasms; Predictive Value of Tests; Prognosis; SPARC protein, human
CTC: circulating tumor cells
Pancreatic cancer remains a malignancy of poor prognosis as overall survival is still low despite recent advances in therapeutics. This is the reason why pancreatic cancer constitutes a therapeutic challenge in current oncology practice. Research is directed towards finding prognostic and predictive factors to be applied in clinical praxis.
The human equilibrative nucleoside transporter- 1 (hENT1) (also known as SLC29A1) is the major route for transporting gemcitabine through the cell membrane. Retrospective studies show that populations of cells with lower hENT1 abundance may be relatively gemcitabine resistant owing to reduced intracellular accumulation of the drug [1]. HENT1 protein expression is associated with increased overall survival and disease free survival in pancreatic cancer patients who received gemcitabine [2, 3].
A characteristic of pancreatic cancer is the formation of a dense stroma, termed a desmoplastic reaction, composed of cellular and fibrillar elements. Pancreatic stellate cells play a critical role in the formation and turnover of the stroma. The stroma is not only a mechanical barrier but also constitutes a dynamic compartment critically involved in the process of tumor formation, progression, invasion, and metastasis [4, 5]. Stromal cells express multiple proteins, one of them being the “secreted protein acidic and rich in cysteine” (SPARC), which is involved in deposition and modeling of the extracellular matrix. Expression of SPARC in the stroma of tumor is a prognostic factor [6].
Concerning the circulating tumor cells (CTC), studies show that patients who had circulating tumor cells (more than 1 in 7.5 mL) before curative surgery, or at either point after therapy initiation, trend towards poorer overall or progression free survival [7].
A high degree of infiltration of both CD4+ and CD8+ T-cells around tumor cells seems to be an independent favorable prognostic factor for overall survival in patients with pancreatic adenocarcinoma and a high prevalence of regulatory T-cells is an independent negative predictive factor for survival [8, 9].
HENT1 Tumor Levels to Predict Survival of Pancreatic Ductal Adenocarcinoma Patients Who Received Adjuvant Gemcitabine and Adjuvant 5-FU on the ESPAC Trials (Abstract #4006 [10])
Neoptolemos et al. performed tissue microarrays for hENT1 to evaluate the predictability of it in the patients who entered the ESPAC 1-3 trials. Patients were operated for resectable pancreatic cancer and received adjuvant chemotherapy with gemcitabine or 5-FU. Patients with expression above the median score (n=48) were classified as “High hENT1”. These patients had significantly longer median survival among those received gemcitabine (26.2 vs. 17.1 months; P=0.002). The expression of hENT1 was not related to any difference in the median survival among patients received 5-FU (25.6 vs. 21.9 months; P=0.362). The results show that patients with high tumor expression of hENT1 can benefit more from treatment with gemcitabine and patients with low tumor expression may be treated with 5-FU.
SPARC in Pancreatic Cancer: Results from the CONKO-001 Study (Abstract #4016 [11])
Sinn et al. have performed immunohistochemistry in the tissue sample for expression of SPARC in the stroma around the tumor, but also in the tumor cell, of patients from the Charité Onkologie (CONKO)-001 study. Patients who received gemcitabine as adjuvant treatment had a longer disease free survival and overall survival when stromal and cytoplasmic expression of SPARC was not-strong or negative, respectively (Tables 1 and 2).
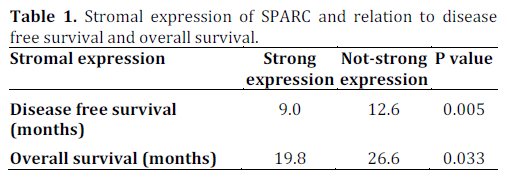
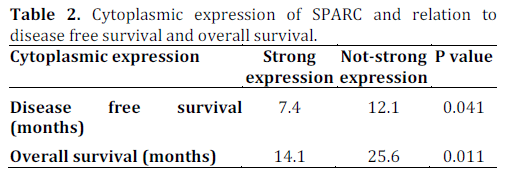
So SPARC expression estimation, both in the tumor or its stroma, seems to be a valuable prognostic factor in patients receiving gemcitabine as adjuvant therapy.
Circulating Tumor Cells in Locally Advanced Pancreatic Adenocarcinoma. The Ancillary Study Circe-07 of the LAP-07 trial (Abstract #4046 [12])
Huhuet et al. prospectively screened for circulating tumor cells patients with locally advanced unresectable pancreatic cancer before and after two months of chemotherapy. More than one tumor cell in 7.5 mL was considered as positive.
Before treatment 5% of patients had positive detection of CTC and 9% at the end of 2 months of therapy. This positivity is associated with poor tumor differentiation (P=0.04) and the overall survival was shorter (P=0.01). So based on the hypothesis that CTC are an index of metastatic potential of the tumor, evaluation of CTC may help to distinguish between patients who need local or systematic treatment.
Prognostic Value of an Immune Score Combining Intra- and Peri-Tumoral T-Cell Subset Density in Patients with Pancreatic Adenocarcinoma (Abstract #4060) [13]
Turcotte et al. evaluated the power of an immune score based on intra- and peri-tumoral Tcell subsets (CD3, CD4, CD8, CD45RO, FOXP3) and density. This immune score is based on the intratumoral CD4 T-cell count (greater than the median value), peritumoral CD8 T-cell count (less, or equal to, the median value) and intratumoral/ peritmoral CD3 T-cell ratio (greater than 1). Results were grouped in 4 categories (none, 1, 2, and 3), according to how many of these features were present. The difference between the groups were significant in relation to overall survival (P=0.01), disease free survival (P=0.01) and time to response (P=0.02). FOXP3 and CD45RO T-cell counts do not seem to have any prognostic value, either as alone or in the immune score. The immune score seems to be an independent prognostic factor that adds information to the TNM staging system.
Even though the results of combinations and newer therapeutic agents become more and more encouraging, pancreatic cancer is still a disease with a very poor prognosis. Effort is given in finding prognostic and predictive factors for these patients.
HENT1 expression seems a useful predictive factor of response to chemotherapy with gemcitabine in the adjuvant setting. The results of the study of Neoptolemos et al. [9] are in accordance with the results of the study of Farell et al. in the population of the Radiation Therapy Oncology Group (RTOG) 9704 study [2]. Patients with hENT1 expression benefit from chemotherapy with gemcitabine, and patients with low or no expression should avoid gemcitabine, as hENT1 is a transporter of it to the intracellular space.
SPARC, also known as osteonectin, is an extracellular matrix protein that has been associated with poor prognosis in pancreatic cancer patients as well as with invasion and metastasis in preclinical models [14, 15]. Recent studies suggest that SPARC plays a critical role in collagen turnover in pancreatic cancer. SPARC is a target of albuminbound nab-paclitaxel. In a phase I/II study of this agent in patients with pancreatic ductal adenocarcinoma, a combination with gemcitabine showed promising results with a median survival of 12.8 months in patients with advanced disease. In parallel mechanistic studies nab-paclitaxel, in addition to its cancer cell-directed effects, has been shown to increase the delivery of gemcitabine. This might be because of stromal depletion and/or modification of the catabolism of gemcitabine [16].
The detection of CTC in patients with pancreatic cancer seems to have a negative prognostic value [17]. Also in resectable pancreatic cancer it seems to be of significant value, because it can determine the patients who may need chemotherapy, besides local treatment, due to micrometastatic disease [12]. Another study by Negin et al. shows that not only the presence of CTC is a poor prognostic factor, but also the type of those cells. Not all CTC are equal. In detail the MUC1 cells seem particularly bad, suggesting that there is a difference in the biology of these tumors and providing some insight into how these tumors function [7].
Infiltration of CD4+ and CD8+ T-cells around tumor cells seems to affect the course of pancreatic cancer. There is a positive relation of the amount of peritumoral T-cells and pancreatic cancer. It is an independent favorable prognostic factor for overall survival (P=0.01) [8]. High prevalence of regulatory T-cells seems to be an independent negative predictive factor for survival (P=0.001) [9]. An immune score, as the one presented by Turcotte et al., is a useful tool [13]. Immune response to the tumor seems to control better the course of the malignancy.
Predictive factors are available now to help doctors decide the treatment that fits their patients. Prognostic factors are also important to find those patients that are candidates for an aggressive therapeutic approach.
The authors have no conflicts to disclose