Original Article - (2019) Volume 20, Issue 2
Oleksandr Rotar1, Igor Khomiak2, Michail Nazarchuck3, Vasyl Rotar1, Andrii Khomiak2, Krishna Taneja1, Sitlana Railianu1
1Bukovinian State Medical University, Chernivtsi, Ukraine
2O.O. Shalimov National Institute of Surgery and Transplantology, Kyiv, Ukraine
35th City Clinical Hospital, Minsk, Belarus
Received Date: December 13th, 2018; Accepted Date: February 19th, 2019
Context Purulent complications worsens prognosis in patients with acute necrotizing pancreatitis. Early diagnosis of infection development can improve results of their management. Objective Aim of our research was to establish utility of presepsin for diagnosis of local and system infected complications during acute necrotizing pancreatitis. Methods Prospective study of 70 patients with severe acute necrotizing pancreatitis in a single intensive care department of regional hospital has been performed. Levels of presepsin before any invasive treatment were determined and compared with results of bacteriological investigation of pancreatic tissue. Findings The positive results of bacteriological examination were obtained in 43 patients. The rate of presepsin was three-four times higher in the patients with infected complications than those in the individuals with sterile pancreatic necrosis. Increasing the presepsin rate above 632 pg / ml allows confirming the presence of both local and systemic infection with high sensitivity and specificity. Receiver-operating characteristic AUC of presepsin diagnostic utility for all types of infected complications of acute necrotizing pancreatitis reached 0.956, áÆ0.883-0.972, p˂0.01. Conclusion Presepsin concentration is early highly sensitive and specific marker for all kind of infected complications of acute necrotizing pancreatitis.
complications; Early Diagnosis; Pancreatitis; Acute Necrotizing; presepsin protein; human
ANP acute necrotizing pancreatitis; ROC receiveroperating characteristic
Acute necrotizing pancreatitis (ANP) is a surgical disease caused by primary aseptic necrosis of the pancreatic tissues, accompanied by the development of SIRS in both aseptic and infected necrosis. In recent years there has been a significant increase in the number of patients with infected forms of ANP and the development of septic complications, whose mortality reaches 5.5- 35.2% [1, 2, 3]. Early diagnosis of infection occurrence is crucial for choosing the optimal treatment strategy [4, 5]. At the same time, infectious complications at ANP do not have specific symptoms and clinical features that distinguish sterile from infected pancreatic necrosis. Computed tomography (CT) which is currently the imaging study of choice for diagnosis of acute pancreatic and peri-pancreatic necrotic collections can only suggests the presence of infection. The appearance of air bubbles within the necrotic pancreatic parenchyma on CT scans is considered as an almost pathognomonic finding indicating the presence of infected pancreatic necrosis, but in some cases the presence of gas indicates a connection with the gastrointestinal tract. Besides, this finding is observed in only one third of patients with infected pancreatic necrosis, and therefore its absence cannot exclude the diagnosis of infected pancreatic necrosis [6, 7]. Bacteriological examination by means of fine needle aspiration of the necrotic collection, which is considered to be the diagnostic "gold standard", needs a lot of time and costs while nearly a third of the patients will receive the negative results [8]. For differential diagnosis of SIRS and abdominal sepsis currently determination of the level of several serological markers of inflammation were applied and presepsin was recognized as most promised [9, 10]. So aim of our research was to study the diagnostic utility of the presepsin for early diagnosis of infectious complications of ANP.
This study was performed at Regional Emergency Hospital, Chernivtsi, Ukraine, between February 2014 and December 2017. During this period 408 patients with acute pancreatitis were admitted to clinic. Acute pancreatitis was defined according to the 2012 revision of the Atlanta classification as an association of two of the three following features: typical abdominal pain (acute onset of a persistent, severe, epigastric pain often radiating to the back), serum lipase or amylase activity at least three times greater than the upper limit of normal, and characteristic findings of acute pancreatitis on abdominal cross-sectional imaging studies [11]. Necrotizing pancreatitis was characterized by inflammation and associated pancreatic parenchymal necrosis and/or peripancreatic necrosis, as shown by a lack of pancreatic parenchymal enhancement and/or the presence of findings of acute necrotic collection and walled-off necrosis on contrast-enhanced computed tomography (CT). The CT-protocol for pancreatic evaluation consisted in a retarded venous phase after 35 s of venous contrast administration. A CT scans were performed in all patients for establishing diagnosis of ANP between 72-96 h after the onset of abdominal pain and were repeated if indications for intervention appeared. Only patients to whom interventional treatment was applied were included to current study. Besides, persons were excluded if any of the following criteria were present: a) age < 18 and > 80 years; b) recent surgical interventions; c) psychoses; d) pregnancy; e) previously history of chronic pancreatitis. After exclusion of abovementioned cases we enrolled to current study 70 patients with ANP.
In line with international guidelines, intervention was generally only performed in case of suspected or confirmed infection of pancreatic necrosis or peri-pancreatic necrosis alone accompanying an aggravated general condition [12, 13]. The decision to intervene was based on clinical grounds (presence of SIRS and ongoing organ failure) and results of CT scans. Whenever possible, intervention was postponed until approximately four weeks after the onset of disease. Microbiological investigation of biological material from necrotic collections was performed at time of intervention and every 3rd day in case draining was applied until removing of drains. Blood culture was performed in all persons at time of intervention and repeated in case deterioration of patient’s condition. Before interventional treatment EDTA plasma samples were collected for presepsin determination. The plasma samples were categorized into 4 groups according to results of microbiological assay at time of intervention and clinical conditions of patients: SIRS without infection, local infection, sepsis and septic shock. Besides, plasma samples were collected from 10 healthy persons as control values for presepsin.
Severity of ANP was determined in line with the recently revised Atlanta Classification [11, 12, 13] by presence and duration of organ failure as well as by APACHE II score. Organ failure was defined by modified Marshall scoring system. The diagnoses of SIRS, sepsis and septic shock were made according to the criteria described by the American College of Chest Physicians/ Society of Critical Care Medicine (ACCP/ SCCM) [14]. All patients received fluid resuscitation, and full laboratory investigations were performed on the first 3 days and in the 24 hours before any intervention. Nasojejunal enteral feeding was initiated if an oral diet was not tolerated after 48-72 hours after admission. If there were problems with nasojejunal intubation nasogastric tube feeding was started. Parenteral nutrition was only initiated when oral route was not tolerated or sufficient, but at least small amount of enteral feeding was present in all patients.
The plasma samples were stored at −70°C until measurement. Presepsin was determined using (Mitsubishi Chemical Medience Co., Tokyo, Japan), a chemiluminescent enzyme immunoassay for the quantitative measurement of presepsin concentration in whole blood or plasma. The test principle is based on the non-competitive chemiluminescence enzyme immunoassay [10]. During incubation of the sample with alkaline phosphatase-labeled anti-presepsin polyclonal antibody and anti- presepsin monoclonal antibody-coated magnetic particles, presepsin of the sample binds to the anti-presepsin antibodies, forming an immune complex with enzyme-labeled antibody and antibody-coated magnetic particles. After removal of the unbound substances a chemiluminescent substrate is added. After a short incubation period, the luminescence intensity generated by the enzyme reaction is measured. The luminescence intensity is related to the presepsin concentration of the sample, which is calculated by means of a standard curve (measurement range: 20 –20,000 ng/l, functional sensitivity: 57.1 ng/l, reference interval: 60 –365 ng/l).
Ethics
The study was conducted in accordance with the principles of the Declaration of Helsinki. The ethics review board of each participating hospital approved the study. Patients or their legal representatives gave written informed consent.
Statistical Analysis
Results of presepsin determination were presented as median [inter-quartile range (IQR)]. Two-group comparisons were performed using the Mann–Whitney U-test with the Bonferroni correction, and multiple comparisons were performed using the Kruskal–Wallis H test or one-way repeated-measures analysis of variance (ANOVA) with Dunnett’s multiple comparison tests. We used the cut-off value obtained by a receiver-operating characteristic (ROC) analysis. Values of P less than 0.05 were considered to indicate statistical significance. Group comparisons and ROC analyses were performed using JMP software (SAS Institute, Cary, North Carolina, USA). Analyses of Spearman correlation coefficients and interactions were performed using SPSS 18 software (SPSS, Chicago, Illinois, USA).
Although suspension of infected ANP with typical clinical presentation (SIRS, ongoing organ failure, leukocytosis, etc.) was the only indication for interventional treatment in current study bacteriological investigation of necrotic collections confirmed infection presence in 49 (70.0%) patients. There were no septicemia and organ dysfunction at time of invasive procedure in 15 of them (Table 1). Positive blood cultures and/or SOFA score over 2 were estimated in 37 patients with infected ANP and 12 cases among them were presented with septic shock.
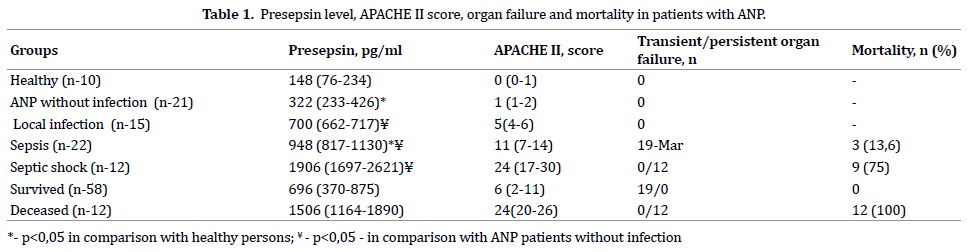
Examination of healthy individuals (control group) determined presepsin concentration in the blood plasma on an average level of 148 (76-234) pg/ml. The signs of SIRS were presented in all patients without infection of necrotic collection and their presepsin concentration blood increased on an average to 322 (233-426) pg/ml, p=0.02. Infection occurrence in patients with ANP was associated with reliable increase of presepsin level an average to 880 (714-1247) pg/ml (p=0,001), which is 2.7 times as high as in patients without infection. At early stages of infected complications when they were local (without signs of generalization) presepsin level increased to 700 (662-717) pg/ml, p=0.001. Presepsin concentration increases progressively with sepsis to 948 (817-1130) pg/ml and in case of septic shock – to 1906 (1697-2621) pg/lm, p=0,001. ROC-analysis found a high dependence of presepsin level on infectious complications available with acute necrotic pancreatitis (AUC ROC 0.956, СІ 0.883- 0.972, p=0.001, Figure 1). According to the data of the study critical presepsin concentration which is indicative of occurrence of infected ANP was 632 pg/ml (sensitivity – 90. 11%, specificity – 92.86%).
Generalization of infected complications was followed by worsening of patients’ condition: the majority of persons with sepsis developed transient organ failure, in 3 patients occurred persistent organ failure (in all cases - renal). All the 12 patients with septic shock had got multiple organ failure producing unfavorable effect on the results of treatment. Besides vascular, there were 7 cases of pulmonary, 4 – renal, 3 – neurological, 2 – hepatic failures. In patients with renal failure concentration of presepsin increased not significantly: 1084 (1061-1164) pg/ml, p=0.254 - in case of sepsis and 2033 (1851-2398), p=0.722 – in persons with septic shock, respectively. Direct correlation between presepsin level and the score evidencing severity of patients’ condition by APACHE II scale was found (Figure 2), correlation coefficient between presepsin concentration and the score according to APACHE II scale was 0,798 (p=0.001).
12 patients out of 70 being examined died (17.1%): 3 patients out of 22 with sepsis, and 9 out of 12 – with septic shock. Presepsin level and the APACHE II score in those who died at time of intervention were significantly higher than in those patients who survived (Table 1). High presepsin level and the APACHE II score were equally indicative of the lethal outcome development: AUC ROC for presepsin was 0.896 ± 0.37 and 0.962 ± 0.28 – for APACHE ІІ scale, p=0.01. Presepsin level higher than 1032 pg/ml was considered to have unfavourable prognosis for patients to survive, and it corresponded to 16 points by APACHE II scale (sensitivity 82.24 %, specificity 88.12 %).
Infection of pancreatic necrosis is a common occurrence with overall rates of infection ranging from 30% to 70%. Early diagnosis and early treatment are essential to improving the results of treatment of infected ANP [4, 5]. Compared with patients with sterile pancreatic necrosis, those with infected necrosis had significantly higher morbidity and mortality [1, 2, 3]. At the same time, infectious complications at ANP do not have specific symptoms and clinical features that distinguish sterile from infected pancreatic necrosis whereas modern imaging methods can only suggest the presence of infection [6, 7].
Taking in account insufficient specificity of imaging methods for early diagnosis of infected ANP some laboratorial markers were evaluated. C-reactive protein was one of the first studied biomarker but its elevation was related mostly to grade of SIRS than infection alone [15]. Same problem were found with other promising markers – interleukin-6 and procalcitonin. Both of them showed good specificity for sepsis diagnosis, especially procalcitonin, but their cut-off levels were too high for discrimination infected necrosis cases from ANP patients presented by SIRS without infection thus lowering their accuracy [16, 17, 18].
In our research we evaluated efficacy of new sepsis biomarker presepsin for early diagnosis of infected ANP. Presepsin was identified as a protein whose levels are elevated specifically in the blood of patients with sepsis. One of the production mechanisms of presepsin is related to bacterial phagocytosis, and the cleavage of membrane CD14 by lysosomal enzymes of granulocytes are involved in its secretion. The measurement of presepsin levels is useful for the diagnosis of sepsis, evaluating the severity of sepsis, and monitoring clinical responses to therapeutic interventions.
In our study presepsin level was considerably higher both in patients with infected ANP comparing those having SIRS without infection. A clear dependence of presepsin concentration on the activity and severity of infectious process was found. At the same time, in addition to increased presepsin level only 11 out of 15 patients with local infection developed the signs of infection such as leukocytosis (>12000) and immature forms of granulocytes >10%. Although, further bacteriological examination found pathological microorganisms in all the cases. It is explained by the fact that the mechanism of presepsin increase differs radically from other proinflammatory markers. Presepsin is formed by means of enzymatic splitting of the circulating soluble receptor СD- 14 by proteases with bacterial phagocytosis at the earliest stage of inflammation before the increase of interleukin-6 level and laboratory signs of systemic inflammatory response syndrome. Contrary to other markers of sepsis the time of presepsin half-life in the blood plasma is very short – from 30 minutes to 4 hours, and for procalcitonin – 25-30 hours. Presepsin concentration increased progressively with sepsis to 948 (817-1130) pg/ml and in case of septic shock – to 1906 (1697-2621) pg/ml, which is almost three and six times as much respectively from the parameters of patients with SIRS without infection. Elevation of presepsin concentration in patients with renal failure was not significant in comparison with appropriate group, but amount of persons with it was nonsufficient in our study to make any conclusion.
Severity of condition of the examined patients with sepsis and severe sepsis was determined by the development of organ failure. An expected relation was found between presepsin level and the score evidencing severity of patients’ condition by APACHE II scale (Figure 2). Presepsin level and the score according to APACHE II scale in those who died were twice and 1, 8 times as much as respectively than in those patients who survived. High presepsin level and the score according to APACHE II scale reliably (p<0.01) were equally indicative of the lethal outcome development when patients with acute necrotic pancreatitis were admitted.
Determination of presepsin in the blood allows confirming with high sensitivity and clinical specificity both local and systemic infected complications during acute necrotizing pancreatitis. High levels of presepsin (over 1032 pg/ml) have negative prognostic utility for patients surviving.
The authors are grateful to all contributors to this research especially the director of the private unitary enterprise “ARGOMEDA”, Minsk, Belarus, Dr. Yaraslau Latushka and staff of surgical and intensive care departments of Chernivtsi Emergency Regional Hospital, Ukraine, for helping of performing research.
There are no competing and conflicting interests.