Research Article - (2017) Volume 3, Issue 2
Allam A1, Alhindi H2, Al-Otaibi F3, Al-Hebshi A4, Khalil E4 and Al-Moghrabi N5*
1Medical Oncology Department, King Faisal Specialist Hospital and Research Center, Riyadh 12713, Saudi Arabia
2Department of Pathology and Laboratory Medicine, King Faisal Specialist Hospital and Research Center, Riyadh 12713, Saudi Arabia
3Department of Neurosciences, Division of Neurosurgery, King Faisal Specialist Hospital and Research Center, Riyadh 12713, Saudi Arabia
4Oncology Department, Oncology Centre, King Faisal Specialist Hospital and Research Center, Riyadh 12713, Saudi Arabia
5Department of Molecular Oncology, King Faisal Specialist Hospital and Research Center, Riyadh 12713, Saudi Arabia
*Corresponding Author:
Al-Moghrabi N
Head of Cancer Epigenetic Section
Department of Molecular Oncology
King Faisal Specialist Hospital and Research Center
P.O. Box 3354, Riyadh 12713, Saudi Arabia
Tel: 966 1 4647272
Fax: 966 1 4427858
E-mail: nisreen@kfshrc.edu.sa
Received Date: June 07, 2017; Accepted Date: June 22, 2017; Published Date: June 26, 2017
Citation: Allam A, Alhindi H, Al-Otaibi F, et al. The Response to Chemo Radiation Therapy in Unresectable Glioblastoma Multiforme Patients in Relation to MGMT Promoter Methylation Status: A Study from a Single Saudi Center. J Clin Epigenet. 2017, Vol 3:2. doi: 10.21767/2472-1158.100054
Background: The optimal therapy for Glioblastoma Multiforme (GBM) constitutes of maximal safe resection followed by adjunctive concurrent radiation and chemotherapy with Temozolomide (TMZ). However, in certain cases safe maximal resection is not practically amenable. The methylation of the MGMT promoter is being used as a prognostic and predictive factor for the GBM response to TMZ therapy. In the present study, we aimed to measure the outcome of GBM Saudi patients who underwent tumor biopsy followed by radiation therapy with or without chemotherapy based on the MGMT promoter methylation status.
Methods: The methylation-specific PCR assay was used to study the methylation status of the MGMT promoter in 77 formalin-fixed paraffin-embedded tissues of high grade glioblastoma.
Results: MGMT promoter methylation was detected in 48 samples (62%). Interestingly, significant difference (p=0.0073) was found in overall survival between patients treated with surgery, radiotherapy and temozolomide and those treated with surgery and radiotherapy only, irrespective of MGMT promoter status. The survival rate for patients with MGMT methylated tumor was significantly different favoring the plus temozolomide group (p=0.0107). However, for the patients with MGMT unmethylated tumor, there was no significant difference (p=0.1453) between the two groups. Using the Cox proportional-hazards model, the methylation status of the MGMT promoter emerged as a significant (p=0.0234) clinically relevant predictor of benefit from temozolomide.
Conclusion: Here, we have demonstrated that MGMT methylation has significantly improved the survival rate in Saudi patients with unresectable GBM who received concomitant radiation therapy and TMZ. However, MGMT methylation status did not impact the response to radiation therapy.
Keywords
Glioblastoma; Multiforme; MGMT; methylation; Survival
Introduction
Malignant brain tumors incidence is around 14.8 per 100,000 persons per year worldwide and Glioblastoma Multiforme (GBM) is the most common parenchymal brain tumor [1]. The optimal therapy for GBM constitutes of maximal safe resection followed by adjunctive concurrent radiation and chemotherapy with Temozolomide (TMZ) [2,3]. Despite the advances in diagnostic tests and treatment methods of GBM, the median survival rate remains around 15 months [4].
The use of TMZ in GBM induces alkalization into DNA to prevent its replication that causes cell death. However, the presence of DNA-repair proteins, such as O6-alkylguanine DNA Alkyl-transferase (AGT), can remove alkyl adducts from the O6 position of guanine and the O4 position of thymine, restoring these DNA bases and eventually preventing TMZ-induced cell death.
The DNA-repair protein AGT is encoded by the gene O6- Methylguanine-DNA-methyltransferase (MGMT) [4-6]. The methylation of the MGMT promoter is being used as a prognostic and predictive factor for the GBM response to TMZ therapy and other medications; however, this remains an area of debate [4,7]. In fact, the MGMT promoter methylation status in GBM is becoming a relevant factor in patient molecular profile [8,9]. In the past, the application of radiation therapy without chemotherapy for GBM was associated with poorer outcome and there is standing evidence that chemotherapy has improved the survival rate [3,10-12]. In addition, the degree of surgical resection is associated with better outcome and for newly diagnosed patients with GBM, the radiation dose of 60 Gy remains the standard care and the use of brachytherapy and radiosurgery was not supported by recent studies [12-14]. Up to date and to the best of our knowledge, the impact of MGMT methylation promoter status on the survival rate of patients with GBM was not investigated in Saudi population. In this study, we report a retrospective analysis of measuring the outcome of GBM Saudi patients who underwent tumor biopsy followed by radiation therapy with or without chemotherapy based on the MGMT promoter methylation status.
Study Samples
Thick tissue sections, 10 microns, of formalin-fixed paraffin-embedded tissues of high grade glioblastoma from Saudi patients treated between 1990 and 2009 were obtained from the Pathology Department at King Faisal Specialist Hospital and Research Centre in Riyadh, Saudi Arabia. Clinicopathological data and treatment outcome, which is represented by overall survival, were provided by the oncology department in the hospital. The study was approved by the Human Research Ethics Committee of King Faisal Specialist Hospital and Research Centre.
DNA Extraction
Genomic DNA was isolated from two to three 10 μM thick paraffin sections using Puregene kit (Gentra Systems, Minneapolis, MN, USA) following the manufacturer's instructions.
Methylation: Specific PCR
DNA methylation of the MGMT promoter region was assessed by MSP. Genomic DNA (1 μg) was treated with sodium bisul-phite and purified using the EpiTect Bisulphite Kit (Qiagen) following the manufacturer's recommendations. Modified DNA was amplified with published PCR primers that distinguish methylated and unmethylated DNA [15]. The methylated primers were: Sense, 5'-TTTCGACGTTCGTAGGTTTTCGC-3′and antisense, 5'-GCACTCTTCCGAAAACGAAACG-3′. The unmethylated primers were: Sense, 5'-TTTGTGTTTTGATGTTTGTAGGTTTTTGT-3′and antisense, 5'-AACTCCACACTCTTCCAAAAACAAAACA 3′. The PCR cycling parameters were 95˚C for 1 min; 35 cycles at 95˚C for 30 s, 59˚C for 30 s, 72˚C for 30 s, and then extension at 72˚C for 7 min (25). The PCR products were electrophoresed on 2% ethidium bromide-stained agarose gels. SssI methylase-treated and untreated bisulfite-modified lymphocyte DNA was used as the positive and negative controls, respectively. All PCR reactions were carried out in replicate.
Statistical Analysis
Survival curves were constructed using the Kaplan-Meier method. Multivariate analysis for survival was performed using the Cox proportional hazards model. The analyses were performed using the software package SAS version 9.4 (SAS Institute Inc., Cary, NC, USA).
Results
Methylated-specific PCR was performed on DNA isolated from 84 paraffin-embedded glioblastoma tumor samples. Out of those 84 DNA samples, only 77 samples gave results for MGMT methylation status (91.6%) (Figure 1). The results obtained show that 48 samples (62%) had detectable methylated MGMT promoter, whereas 29 samples (38%) had unmethylated MGMT promoter. The overall survival and outcomes were known for only 52 specimens out of the 77 (60.9% of the tumors from the overall study population) (Table 1). We have found a significant difference in overall survival between patients treated with surgery, radiotherapy and temozolomide and those treated with surgery and radiotherapy only, irrespective of MGMT promoter status (p=0.0073 T test) (Figure 2). The median overall survival among patients treated with temozolomide was 12 months (95% confidence interval, 8.6 to 15.5), as compared to 6.5 months (95% confidence 4.6 to 8.3) among those who were not treated by the drug.
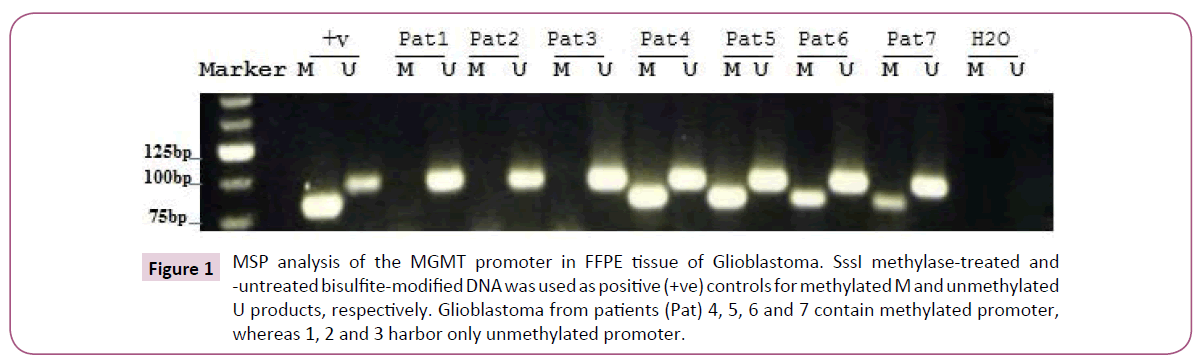
Figure 1: MSP analysis of the MGMT promoter in FFPE tissue of Glioblastoma. SssI methylase-treated and -untreated bisulfite-modified DNA was used as positive (+ve) controls for methylated M and unmethylated U products, respectively. Glioblastoma from patients (Pat) 4, 5, 6 and 7 contain methylated promoter, whereas 1, 2 and 3 harbor only unmethylated promoter.
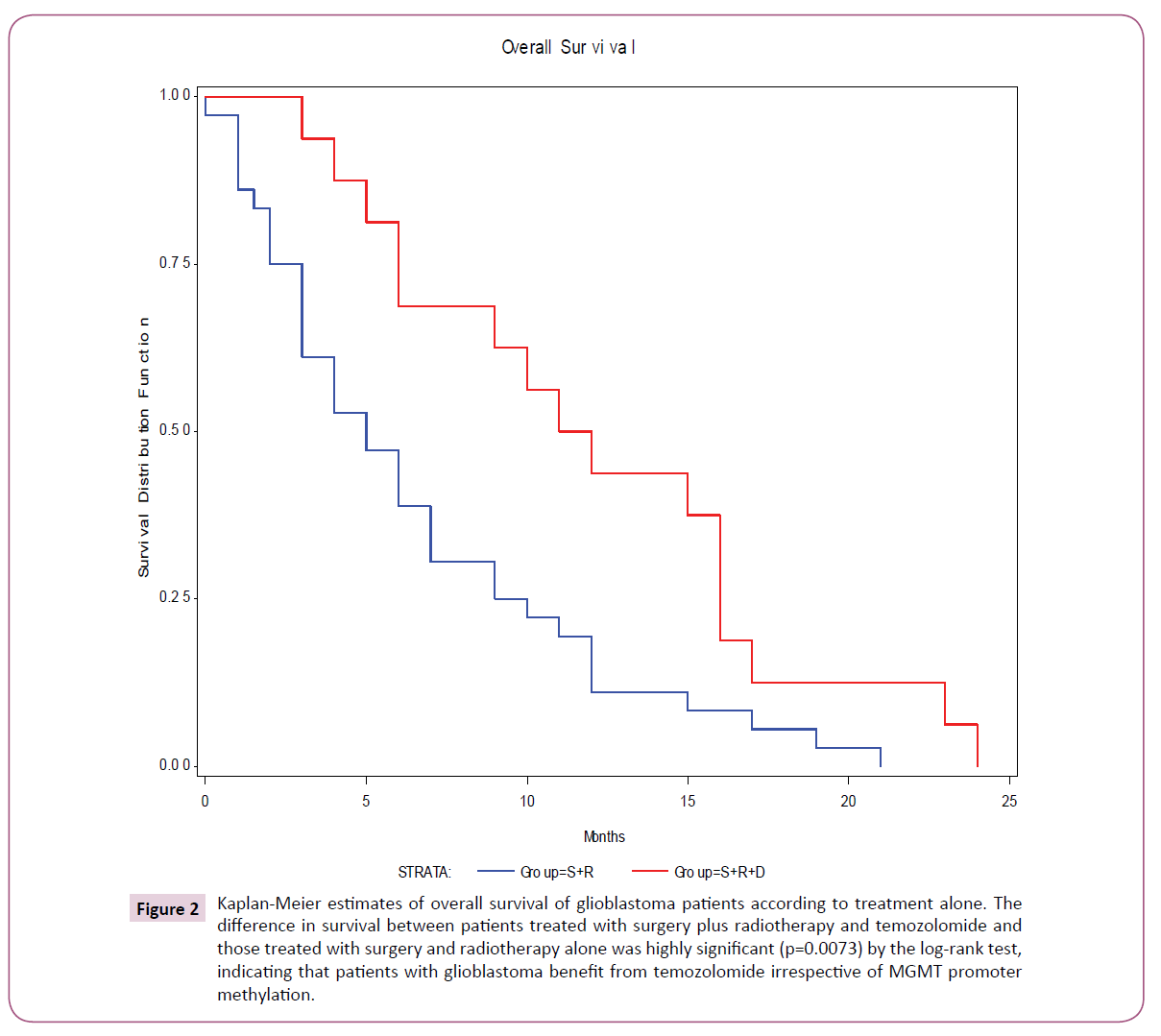
Figure 2: Kaplan-Meier estimates of overall survival of glioblastoma patients according to treatment alone. The difference in survival between patients treated with surgery plus radiotherapy and temozolomide and those treated with surgery and radiotherapy alone was highly significant (p=0.0073) by the log-rank test, indicating that patients with glioblastoma benefit from temozolomide irrespective of MGMT promoter methylation.
| Total n=52 | % | Methylated n=33 | 63.5% | Unmethylated n=19 | 36.5% | |
|---|---|---|---|---|---|---|
| Age | ||||||
| Median | - | - | 46 (13-79) | - | 54 (14-76) | - |
| Gender | ||||||
| Female | 23 | 44 | 17 | 73.9 | 6 | 26 |
| Male | 29 | 55 | 16 | 55.2 | 13 | 44.8 |
| Treatment | ||||||
| S+R | 36 | 65 | 23 | 63.8 | 13 | 36.1 |
| S+R+D | 16 | 35 | 10 | 62.5 | 6 | 37.5 |
| S: Surgery; R: Radiotherapy; D: Drug (Temozolomide) | ||||||
Table 1: Characteristics of patients and methylation status of the MGMT promoter.
When both treatment assignment and MGMT promoter methylation status were considered, the Kaplan-Meier estimates of overall survival for the patients with MGMT methylated tumor was significantly different favoring the plus temozolomide group (p=0.0107 by the Log-rank test) (Figure 3A). By contrast, for the patients with MGMT unmethylated tumor, there was no significant difference in overall survival between the two groups (p=0.1453 by the Log-rank test) (Figure 3B).
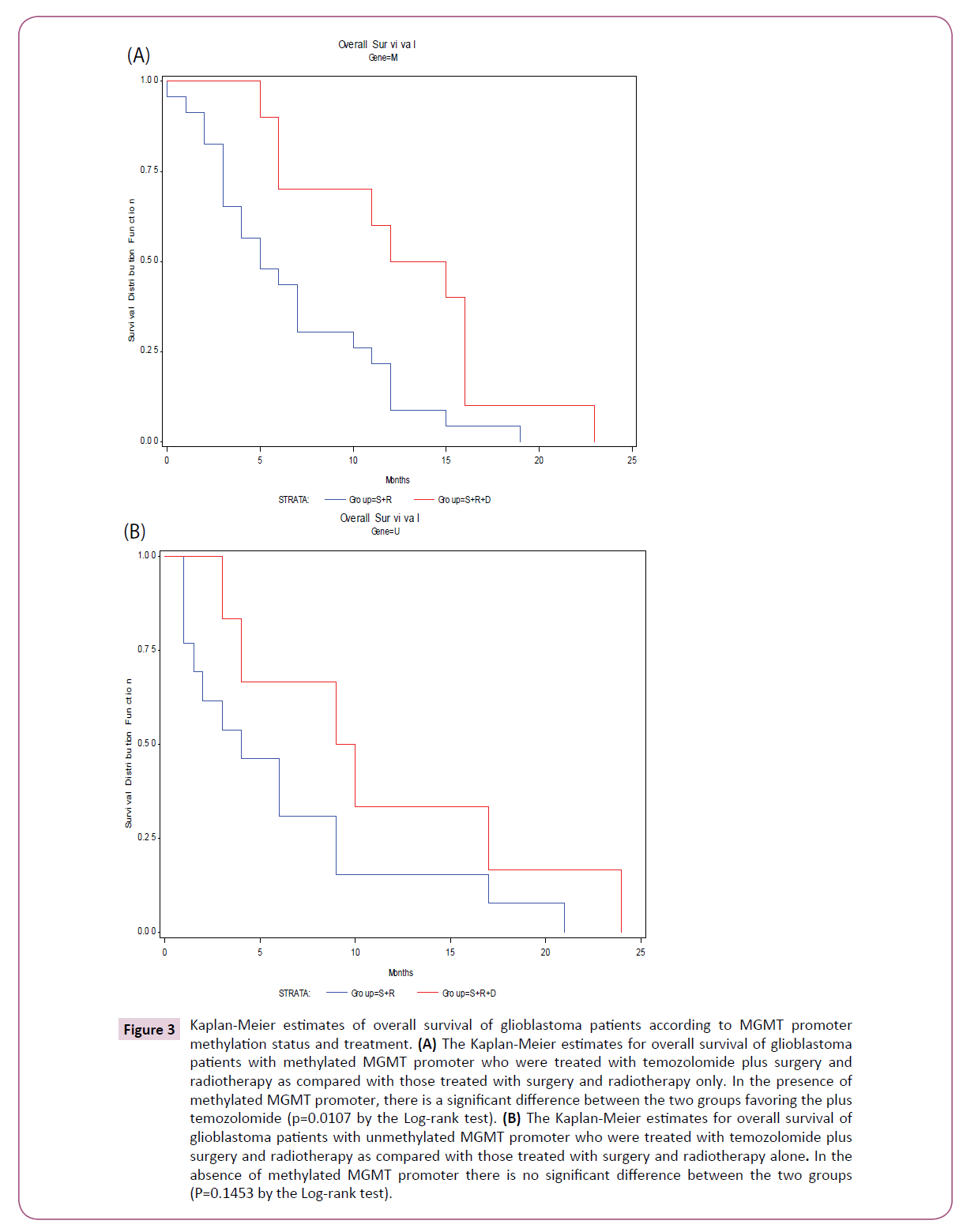
Figure 3: Kaplan-Meier estimates of overall survival of glioblastoma patients according to MGMT promoter methylation status and treatment. (A) The Kaplan-Meier estimates for overall survival of glioblastoma patients with methylated MGMT promoter who were treated with temozolomide plus surgery and radiotherapy as compared with those treated with surgery and radiotherapy only. In the presence of methylated MGMT promoter, there is a significant difference between the two groups favoring the plus temozolomide (p=0.0107 by the Log-rank test). (B) The Kaplan-Meier estimates for overall survival of glioblastoma patients with unmethylated MGMT promoter who were treated with temozolomide plus surgery and radiotherapy as compared with those treated with surgery and radiotherapy alone. In the absence of methylated MGMT promoter there is no significant difference between the two groups (P=0.1453 by the Log-rank test).
To analyze further the influence of the methylation status of the MGMT promoter, a multivariate analysis with the use of the Cox proportional-hazards model stratified according to treatment group was used (Table 2). The methylation status of the MGMT promoter emerged as a significant (p=0.0234) clinically relevant predictor of benefit from temozolomide.
| Un-methylation | MGMT promoter methylation | MGMT promoter | ||
|---|---|---|---|---|
| P Value | Hazard Ratio (95% CI) | P Value | Hazard Ratio (95% CI) | |
| Drug plus S plus Rad (vs. S plus Rad) | 0.0234 | 0.383 (0.167-0.878) | 0.1897 | 0.478 (0.159-1.44) |
| Age | 0.6254 | 1.006 (0.983-1.028) | 0.9266 | 0.999 (0.975-1.024) |
| Male vs. Female | 0.1588 | 0.586 (0.278-1.233) | 0.2154 | 0.483 (0.153-1.527) |
| S: Surgery; Rad: Radiotherapy; CI: Confidence Interval; Drug (Temozolomide) | ||||
Table 2: Results of analysis with the cox proportional-hazards model.
Discussion and Conclusion
Glioblastoma multiforme is the most malignant astrocytic tumor of the nervous system that can arise as primary or secondary tumors [16]. The overall prognosis remains poor despite many clinical and laboratory trials to control such highly malignant brain cancer. The concomitant use of radiation therapy and TMZ was found to prolong the survival rate as compared to radiation therapy only [17]. More recent, the MGMT promoter methylation status was reported to be associated with better response to therapy [8,9]. Several studies were mainly focused on the degree of chemotherapy effectiveness in MGMT promoter methylated and non-methylated GBM [9,18-20]. In contrast, a study on Chinese patients showed no correlation between MGMT promoter methylation status and survival rate in patients with GBM [21]. Moreover, surgical maximal resection is a known factor that prolongs survival as compared to biopsy only [14]. However, there are many factors that can limit surgical resectability of GBM such as deep seated tumor, bilateral disease with corpus callosum involvements, and patient’s related comorbidities and age. In this study, we evaluated the survival rate of patients with GBM that is not amenable for resection in relation to MGMT promoter methylation status and the response to radiation therapy alone and chemo-radiation therapy. We used MGMT promoter methylation determined by methylation specific PCR from paraffin-embedded tissues, which is considered a cost effective and ease to apply method [19,22]. The patients’ groups have no differences in terms of characteristics and all underwent tumor surgical biopsy. Irrespective of the MGMT promoter methylation status, the patients group who underwent tumor biopsy with no chemotherapy showed poorer survival rate as compared to the group who received TMZ in addition to other treatment modalities (p value: 0.0023). In the analyzed groups, TMZ therapy had improved the median survival rate from 6.5 months to 12 months. In our institution, this finding was observed after the introduction of TMZ and the positive impact of TMZ on the survival rate is known for more than a decade [3,6,8,17].
The MGMT promoter methylation status did not improve the response to radiation therapy in this study. In contrast, we demonstrated that MGMT methylation has significantly improved the survival rate in patients with GBM who received concomitant radiation therapy and TMZ. This finding was supported by the Cox proportional-hazards model. Taking in consideration the overall survival rate results; the patients with MGMT unmethylated tumor showed poorer outcome regardless of the type of delivered therapy. These results support the use of MGMT as a prognostic factor in Saudi patients with GBM. This is in concordance with other studies [9,23] in contrast to the Chinese study. The divergence in the results based on different population sample worth further investigations to reveal the exact reason of the different response to therapy.
In conclusion, we have demonstrated that MGMT promoter methylation status increases the response to TMZ in Saudi patients with unresectable GBM but not to radiation therapy. Unmethylated GBM showed poorer outcome regardless of the delivered treatment methods. Hence, MGMT methylation status is an important prognostic factor that needs to be considered during patients counselling.
Acknowledgements
We would like to thank the research center administration for its continuous encouragement and support. The King Faisal Specialist Hospital and Research Center supported this study under the RAC proposal no, 2080 024.