- (2006) Volume 7, Issue 5
Tomas Symersky, Bas van Hoorn, Ad AM Masclee
Department of Gastroenterology-Hepatology, Leiden University Medical Center. Leiden, The Netherlands
Received: 15th, February 2006 Accepted: 8th, March 2006
Context It is generally assumed that pancreatic function recovers completely after mild but not after severe acute pancreatitis. Objective To evaluate both pancreatic function and quality of life in patients who had recovered from acute pancreatitis in a long-term follow-up study. Participants Thirty-four patients (mean age: 56 years) who had recovered from biliary (n=26) or post ERCP (n=8) acute pancreatitis. The mean time after the event was 4.6 years. Main outcome measures Pancreatic function was evaluated by fecal fat excretion, urinary 4-aminobenzoic acid (PABA) recovery, oral glucose tolerance test and pancreatic polypeptide (PP) secretion. In addition, the quality of life was measured by the gastrointestinal quality of life index (GIQLI). Results Of the 34 patients, 22 (65%) had mild and 12 (35%) had severe acute pancreatitis. Exocrine insufficiency (fecal fat greater than 7 g/24h and/or urinary PABA recovery less than 50%) was present in 22 (65%) patients: in 10 (83%) after severe and in 12 (55%) after mild acute pancreatitis, respectively (P=0.140). Endocrine insufficiency was present in 12 patients (35%): 7 (32%) mild versus 5 (42%) severe acute pancreatitis; P=0.711. the quality of life was significantly impaired after acute pancreatitis, (P=0.024). No significant relationship was found between the severity of the pancreatitis and impairment of the quality of life (P=0.604). Conclusion In a significant proportion of patients who had recovered from acute pancreatitis, exocrine and endocrine functional impairment was found. This finding is not confined only to patients after severe acute pancreatitis. Routine evaluation of pancreatic function after acute pancreatitis should be considered.
Islets of Langerhans; Pancreas, Exocrine; Pancreatic Function Tests; Pancreatitis, Acute Necrotizing; Quality of Life
GIQLI: gastrointestinal quality of life index; GTT: glucose tolerance test; PABA: 4-aminobenzoic acid; PP: pancreatic polypeptide
Patients who have recovered from acute pancreatitis are no longer from followed up if their clinical recovery is uneventful and no complications such as pseudocysts have occurred. Pancreatic function in these patients is not routinely evaluated unless typical symptoms suggesting exocrine or endocrine pancreatic insufficiency develop. It was previously assumed that the pancreas and its function would recover completely after mild (edematous) pancreatitis. Results of studies evaluating pancreatic function after acute edematous and necrotizing pancreatitis have shown contrasting results. Functional and morphological abnormalities may persist after the episode of acute pancreatitis but the prevalence of exocrine and endocrine insufficiency during a long term follow-up after acute pancreatitis varies from 10 to over 80% [1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16]. It has been suggested that the degree of functional impairment correlates with the severity of the attack, the extent of pancreatic necrosis, and the cause of the pancreatitis (alcoholic vs. biliary) [2, 3, 5].
In patients with documented severe exocrine pancreatic insufficiency, symptoms related to maldigestion of nutrients may occur. Loss of pancreatic function after recovery from acute pancreatitis, may influence the quality of life. However, few data are currently available. Broome et al. [17] studied the quality of life using the Medical Outcome Scale (MOS) Short-Form 36 in patients after recovery from acute necrotizing pancreatitis. They did not observe any significant reduction in quality of life in these patients.
The aim of the present study was to evaluate both pancreatic function and quality of life in patients who had recovered from acute pancreatitis using long term follow-up. We hypothesized that a loss of pancreatic function subsequent to acute pancreatitis would have an impact on the quality of life.
Patients
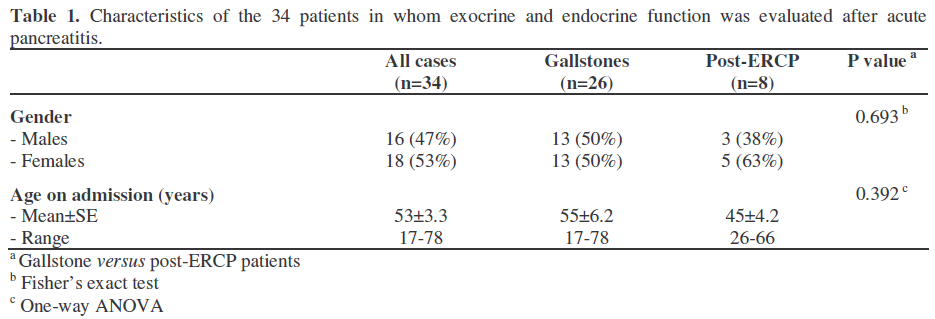
Between 1990 and 1996, 231 patients were admitted to the Leiden University Medical Center with acute pancreatitis. All medical records were reviewed. When an alcoholic etiology was suspected, the patient was excluded from the study. Progression to chronic pancreatitis and subsequent pancreatic insufficiency commonly occurs with alcoholic causes. Therefore, only patients with a documented biliary cause of the pancreatitis and patients with post-ERCP pancreatitis were invited to participate. Patients were contacted by mail. Out of the 75 patients selected, 34 (45%) patients agreed to participate in the study (Table 1). Eight patients (24%) had developed post-ERCP acute pancreatitis and 26 patients (76%) had acute biliary pancreatitis. The indication for ERCP in the 8 patients was a clinical suspicion of choledocholithiasis. During the endoscopic procedures in these 8 patients, no stones or other abnormalities were found. The severity of acute pancreatitis was based on the Atlanta criteria in the retrospective study [18]. The mean interval between the episode of acute pancreatitis (date of diagnosis) and the evaluation of pancreatic function was 4.6 years (range 1.0-7.5 years). All tests were performed within an interval of 2 months. Twenty-eight patients (82%) were treated conservatively while 6 patients (18%) required surgical intervention (drainage procedures or necrosectomy). In 2006, medical records were reviewed or the patients were contacted. The additional information confirmed that no recurrent attacks of pancreatitis had occurred during an additional follow-up of at least 4 years after the function test had been performed.
Quality of Life
The quality of life was evaluated by the gastro-intestinal quality of life index (GIQLI). The GIQLI is a validated instrument for measuring quality of life, specific for patients with gastro-intestinal symptoms and/or diseases [19, 20]. It is a questionnaire consisting of 36 questions, each with five possible responses (0 points for least desirable option up to 4 points for the most desirable option). The higher the score (scale 0-144) the better the quality of life. Eypasch et al. introduced and validated the questionnaire [19]. We have also used and validated this questionnaire previously [20].
Pancreatic Function
The pancreatic function test performed in our hospital is a combined exocrine-endocrine test: urinary bentiromide test [21] combined with a glucose tolerance test (GTT) and plasma pancreatic polypeptide (PP) secretion in response to meal ingestion [22, 23]. PP secretion is related to the amount of functioning pancreatic tissue and the degree of atrophy of the pancreas [22, 23]. After an overnight fast the patient consumed a test meal to which 1 g bentiromide and 75 g of glucose were added in order to evaluate 4- aminobenzoic acid (PABA) recovery and glucose tolerance, respectively. The test meal consisted of one slice of white bread, margarine, 2 slices of full-fat cheese, 250 mL low-fat yogurt and one hard-boiled egg. In total, the patient consumed 700 kcal (32 g fat, 29 g protein and 105 g carbohydrates). Subjects were encouraged to drink at least half a liter of either tea or water to stimulate diuresis. Urine was collected for 6 h after meal ingestion. Blood samples for measuring plasma glucose, PP and cholecystokinin were taken every 15 min, starting at 15 min before ingestion of the meal until 120 min postprandially. A peak glucose concentration of 11.1 mmol/L or higher during the test was considered to be abnormal and was considered to indicate diabetes mellitus.
Plasma PP levels were measured by a sensitive and specific radioimmunoassay previously described [24]. The cut-off values for plasma PP peak increment (reference values greater than 44 pmol/L) and PABA recovery are based on the results obtained in 35 healthy control subjects [22]. A PABA recovery of 70-80% is found in healthy volunteers; a recovery of 50% or less indicates exocrine pancreatic dysfunction [22].
Fecal Fat Analysis
Subjects collected their stools for 48 h while on a 100 g/day fat diet (which was started at least one day before starting the stool collection). Fecal fat content was analyzed using the Van de Kamer method [25]. A fecal fat excretion of more than 7 g/24 h is considered abnormal and indicates exocrine pancreatic insufficiency.
The study protocol was approved by the local Medical Ethics Committee. Written informed consent was obtained from each patient and the study protocol conformed to the ethical guidelines of the "World Medical Association Declaration of Helsinki - Ethical Principles for Medical Research Involving Human Subjects" adopted by the 18th WMA General Assembly, Helsinki, Finland, June 1964, as revised in Toyo 2004.
STATISTICS
Data are expressed as mean, standard error (SE), range and frequencies. For analysis and statistical testing, Microsoft Excel 2000 and SPSS version 12 (for Windows) were used. Continuous and discrete data were analyzed by means of one-way ANOVA and the Fisher’s exact test, respectively. When statistical analyses indicated a two-tailed probability of less than 0.05 for the null hypothesis, differences were assumed significant.
Exocrine Pancreatic Function
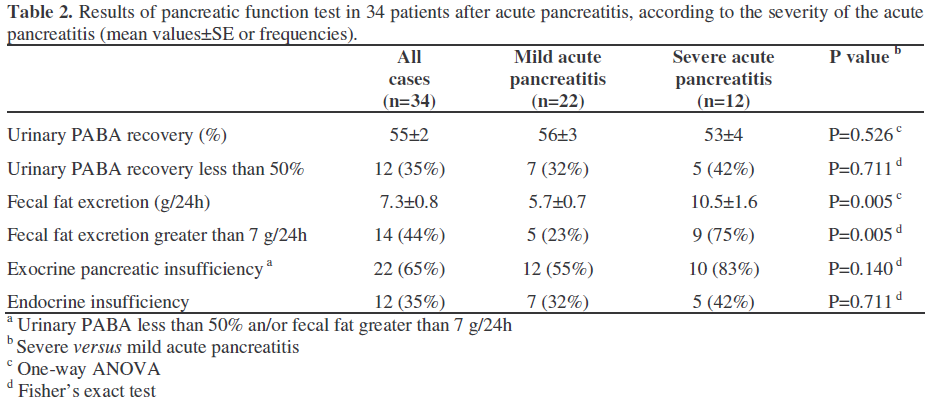
Of the 34 patients, 22 (65%) had evidence of exocrine pancreatic function impairment as shown by elevated fecal fat excretion and/or reduced urinary PABA recovery (Table 2). Fecal fat excretion was significantly (P=0.005) higher in the severe pancreatitis group as compared to the mild pancreatitis group (10.5±1.6 versus 5.7±0.7 g/24h, respectively). Consequently, there was a significantly higher number of patients with elevated fecal fat excretion (values greater than 7 g/24h) in the severe acute group (9/12, 75%) than in the mild acute group (5/22, 23%) (P=0.005). the fecal mass was also significantly higher in the severe group 263±39 g/24h versus 176±22 g/24h) than in the mild group (P=0.049). Urinary PABA recovery did not differ significantly between the two subgroups (P=0.526).
Endocrine Pancreatic Function
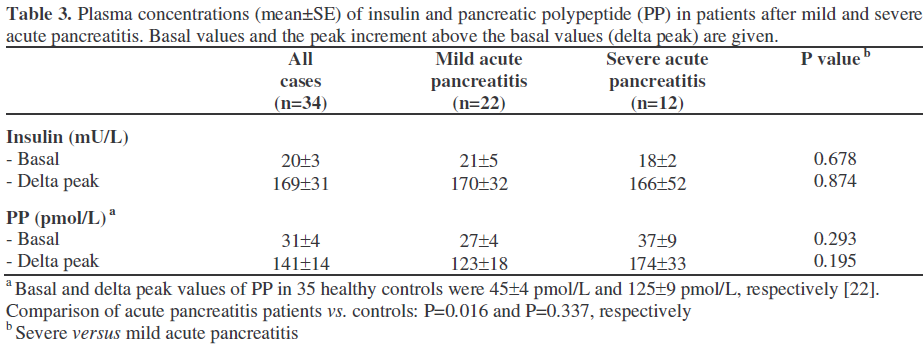
In the mild pancreatitis group, 7 patients (32%) had impaired glucose tolerance, of which 3 were insulin dependent. In the severe pancreatitis group, 5 patients (42%) had impaired glucose tolerance, of which three were insulin-dependent. Two of these three became insulin-dependent after the episode of acute pancreatitis (de novo IDDM). Basal plasma insulin levels and the peak increment in plasma insulin were not significantly different (P=0.678 and P=0.874, respectively) between patients with mild and those with severe acute pancreatitis (Table 3).
Plasma pancreatic polypeptide
Mean basal PP values significantly differed between patients and healthy controls (P=0.016, Table 3). In all groups plasma PP concentrations increased after ingestion of the test meal. Mean postprandial PP peak increments were 141±14 pmol/L and 125±9 pmol/L in patients and healthy controls, respectively (P=0.337). The postprandial plasma peak increment failed to reach the cutoff value of at least 44 pmol/L in 5 (14.7%) of the 34 patients. Four of these had experienced mild acute pancreatitis, while one had recovered from severe acute pancreatitis. There was no significant relationship between the severity of the pancreatitis and an abnormal PP response to the test meal (severe: 1/12, 8.3%; mild: 4/22, 18.2%; P=0.635).
Quality of Life
Patients had a mean GIQLI score of 104±3 which was significantly (P=0.024) reduced compared to an age-matched control population (mean score 126±1) [18, 19]. No significant relationship was found between the reduction in the quality of life and the severity of the acute pancreatitis; in fact, the mild group had a mean GIQLI score of 105±5, while the severe group had a mean GIQLI score of 101±4 (P=0.604).
We have shown that, even after almost five years, pancreatic functional impairment is common in patients who have recovered from a single episode of acute pancreatitis. By excluding patients with recurrent episodes of pancreatitis and patients in whom alcohol could have been an etiological factor, we enrolled those patients who were most likely to have non-progressive disease. None of the patients had been seen regularly at the outpatient department or had been followed after any previous episode of acute pancreatitis. Patients were contacted by mail and agreed to participate in the study.
It was shown, by using two conventional indirect pancreatic function tests, that up to 65% of the patients who had a mean followup of 4.6 years after the acute pancreatitis episode showed evidence of exocrine functional impairment. Our findings confirm previous observations that up to 85% of patients may have some functional impairment after acute pancreatitis [1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16]. In several of these studies, pancreatic function was evaluated shortly after the pancreatitis and the pancreas may have not completely recovered from the acute episode [3, 11]. In most studies, patients with alcohol induced pancreatitis were also included. Several of them, in fact, proved to have chronic pancreatitis [1, 2, 3]. In this group, functional impairment and morphological changes frequently develop. Marotta et al. have pointed to exocrine impairment in 30% of patients after a mild attack of acute biliary pancreatitis [16]. We excluded patients with an alcoholic origin of pancreatitis. To date, only patients with biliary or iatrogenic (post- ERCP) pancreatitis were included. We used the Atlanta classification [18] to score the overall clinical severity of the acute pancreatitis: 35% were classified as severe and 65% as mild. We found a significant correlation between the severity and the exocrine dysfunction, determined by elevated fecal fat excretion. The studies reporting a high percentage of exocrine functional impairment after acute pancreatitis primarily included patients after severe necrotizing pancreatitis: Bozkurt et al. [1] with up to 85% of patients showing functional impairment and Boreham et al. [3] with functional impairment in 86% post-severe acute pancreatitis patients but only 13% in postmild acute pancreatitis patients.
Our findings are in line with studies by Boreham et al. [3], Angelini et al. [6], and Niederau et al. [26], pointing to a relationship between the degree of exocrine functional impairment and the severity of acute pancreatitis. Seidensticker et al. [8] reported that functional impairment was more frequent in necrotizing than in edematous pancreatitis.
They also showed that 34 months after recovery, 50% of the patients had morphological abnormalities, indicating that progression to chronic pancreatitis occurs in a considerable percentage of patients with alcoholic origin.
With a follow-up of at least 4 years after the pancreatic function test, none of the patients we studied had experienced another attack of pancreatitis or had had symptoms pointing to chronic pancreatitis. Ten patients were taking enzyme supplementation initiated on the basis of the result of the pancreatic function test.
Endocrine impairment was observed in 35% of the patients. This percentage was not different among the subgroups with mild and severe acute pancreatitis. The percentage of patients with impaired glucose tolerance before the episode of acute pancreatitis is not known. As for insulin treatment, none vs. 2 patients became insulin-dependent after mild and severe acute pancreatitis, respectively.
Quality of life was markedly impaired in the post-acute pancreatitis group. No data on the quality of life were available from the period before the acute pancreatitis. Therefore, we cannot conclude that impaired quality of life results from the acute pancreatitis episode per se. On the other hand, the group of patients after acute pancreatitis had no comorbidity which could have interfered with the quality of life measurement.
We conclude that a significant proportion of patients having had a single attack of nonalcoholic acute pancreatitis shows evidence of exocrine functional impairment (65%) and endocrine impairment (35%), even after a long follow-up period. This finding is not confined to the severe acute pancreatitis patients, but is also present in the patients who have recovered from mild acute pancreatitis. In addition, the quality of life is significantly impaired. Due to the high prevalence of impaired pancreatic function following acute pancreatitis, a routine evaluation of the exocrine function should be undertaken since any dysfunction can be adequately managed by pancreatic enzyme supplementation.