Research Article - (2022) Volume 30, Issue 1
Received: 04-Jan-2022, Manuscript No. IPQPC-22-12489; Editor assigned: 06-Jan-2022, Pre QC No. IPQPC-22-12489(PQ); Reviewed: 20-Jan-2022, QC No. IPQPC-22-12489; Revised: 26-Jan-2022, Manuscript No. IPQPC-22-12489(R); Published: 31-Jan-2022, DOI: 10.36648/1479-1072.22.30.06-09
Objectives: Patients with dyspepsia who are prescribed PPIs often report to their general practitioners (GPs) whether symptoms improved or not. The aim of this study is to investigate whether GPs are more likely to re-prescribe PPIs if patient feedback on the effectiveness of the drugs is positive.
Design: This is an observational study for which data was collected retrospectively from September 2016 until September 2020. Patients with un-investigated dyspepsia were selected. After taking a course of PPIs, patients either did not follow up with their GPs, or followed up stating that their symptoms either improved or did not improve. The percentage of patients who were re-prescribed PPIs was calculated in each category.
Setting: The study took place in Bildeston Health Centre, a GP surgery for approximately 6780 patients in the town of Bildeston in Suffolk, United Kingdom.
Participants: Patients were selected using convenience sampling. The search identified all patients with Read Codes relating to dyspepsia. Important eligibility criteria included patients above eighteen years of age, no previously established cause for dyspepsia, no use of non-steroidal anti-inflammatory medications or aspirin, and no suspicion of malignancy or life-threatening diseases such as gastrointestinal bleeding. 162 patients were included in the study.
Results: Patients with un-investigated dyspepsia who stated that their symptoms improved after a course of PPIs were not more likely to be re-prescribed PPIs than those whose symptoms did not improve. They were, however, 87% more likely to be re-prescribed PPIs than patients who did not follow up with their GP.
Conclusions: There is a high rate of PPI re-prescriptions in patients with un-investigated dyspepsia. This was not affected by patients’ feedback on the effectiveness of PPIs.
Definition and Epidemiology
Dyspepsia is a variety of upper gastrointestinal tract symptoms including abdominal pain, abdominal discomfort, heartburn, acid reflux, nausea, and/or vomiting [1]. It is a common presentation in primary care and has a worldwide prevalence of around 21% [2]. The prevalence of dyspepsia in the UK varies from 12% to 41% [3]. These figures are highly variable between countries and increase when a broader definition for dyspepsia is used [2,3].
Uninvestigated dyspepsia is the term used for patients with dyspepsia who have not had an upper gastrointestinal endoscopy [1]. It is diagnosed and treated differently from “dyspepsia-proven GORD”, “dyspepsia-proven functional”, and “dyspepsia- proven peptic ulcer”, all of which require endoscopy for diagnosis. When endoscopies were performed on patients with dyspepsia to investigate for underlying pathology, more than 65% had functional dyspepsia while a smaller minority had oesophagitis or peptic ulcer disease [4,5]. Less than 2% of patients had gastric or oesophageal cancer [4,5].
H. Pylori and Causes of Dyspepsia
Helicobacter pylori (H. pylori) is a well-known cause for dyspepsia and infects around 35%-40% of people in the United Kingdom (UK) [3,6]. It infects people in almost every country in the world and had an estimated global prevalence of 4.4 billion individuals in 2015 [6]. Data on the prevalence of H. Pylori in patients with dyspepsia in the UK is limited. Patients with H. Pylori carry an increased risk of gastritis, gastric and duodenal ulcers, gastric cancer, and gastric mucosa-associated lymphoid tissue B-cell lymphoma [7]. Therefore, guidelines on the management of dyspepsia have included testing for and treating H. pylori.
Another common cause of dyspepsia is the use of non-steroidal anti-inflammatory drugs (NSAIDs). NSAIDs are effective antipyretic, analgesic, and anti-inflammatory medications. They are easily accessible over the counter and are the most prescribed medications in the world [8]. The mechanisms by which NSAIDs cause dyspepsia is complex and not fully understood [8]. They inhibit cyclooxygenase-1 (COX-1) and cyclooxygensase- 2 (COX-2) enzymes, leading to reduced production of prostaglandins. This increases gastric motility and mucosal permeability leading neutrophil infiltration, oxyradical production, and gastric damage [9]. Management of NSAID induced dyspepsia includes the cessation of the drug, using the lowest effective dose, changing to an NSAID with lower gastrointestinal side effects, or co-prescribing proton pump inhibitors (PPIs) [8].
NICE Guidelines
The National Institute for Health and Care Excellence (NICE) published guidelines on the management of un-investigated dyspepsia in primary care. Recommendations state that patients should either be managed with a full dose of PPIs for one month or tested for H. Pylori and treated with eradication therapy if the patient tests positive [1]. In either case, if symptoms persist or recur, then the alternative approach should be tried [1]. If both methods fail to control symptoms, then endoscopy or long-term acid suppression with PPIs or H2-receptor antagonists should be considered [1]. Re-prescribing PPIs is not part of the pathway for management of un-investigated dyspepsia.
PPIs and Overprescribing
Proton pump inhibitors (PPIs) are prescribed frequently due to their efficacy and low side effect profile. They are often overprescribed leading to increased spending by the healthcare services and avoidable medication side effects [10]. Side effects of the long-term use of PPIs include an increased risk of fractures, gastric polyps, gastric cancer, hypomagnesaemia, and Clostridium difficile infections [11,12]. The factors that influence prescribing decisions which could lead to overprescribing are numerous and interrelated. They commonly include clinical experience, physician’s specialty, continuous professional development, cost of the medicine, and pharmaceutical companies’ marketing and promotion strategies [13]. Patient preferences expectations, and involvement in their treatment also influenced prescriptions [13].
The purpose of this study is to investigate whether patient feedback on the effectiveness of PPIs in uninvestigated dyspepsia increases the likelihood of PPIs being re-prescribed.
This is an observational study. The sample was selected from Bildeston Health Centre, a GP surgery in Suffolk to which around 6780 patients are registered. Patient records from September 2016 until September 2020 were accessed through System One. No specific study size was targeted, and convenience sampling selected for patients over the four years. The selection process was based on searching for the following Read Codes: dyspepsia, non-ulcer dyspepsia, flatulent dyspepsia, gastro-oesophageal reflux disease, indigestion, indigestion symptoms, gastritis, acute gastritis, gastritis unspecified, H. Pylori gastritis, gastritis and duodenitis, and acid reflux and/or oesophagitis. 262 patients were identified using this method (Figure 1). Of those, 180 were included in the study after the following exclusion criteria were applied: age <18 years, dyspepsia with proven underlying cause, patients taking aspirin or NSAIDs, patients at risk of gastrointestinal bleeding, patients who cannot or do not want to take PPIs, suspicion of upper gastrointestinal cancer or bleeding, information not documented clearly, and doctors unsure of diagnosis.
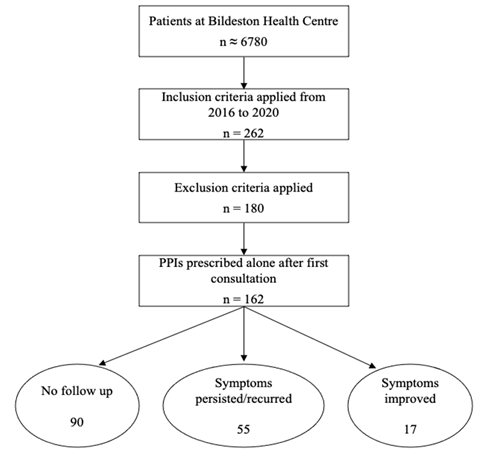
Figure 1:Flowchart of Patients treatment at Bildeston Health centre
Out of the 180 patients in the study, 162 were treated with PPIs alone after being diagnosed with un-investigated dyspepsia. Most of these patients were above 43 years of age. Following a course of PPIs, patients often contacted their GP with feedback regarding the effectiveness of the PPIs in reducing their symptoms. There were three outcomes following a course of PPIs (Figure 1). The first is that patients did not follow up with their GPs. The second is that patients followed up stating that their symptoms did not improve (persisted/worsened). The third is patients stating their symptoms improved. The GP then set a new management plan following patients’ feedback or lack thereof. This plan included re-prescribing PPIs, testing for H.pylori, or referring patients for an upper gastrointestinal endoscopy.
Patients who reported improvements following a course of PPIs were re-prescribed the medications in 88.2% (15/17) of cases (Figure 2). Those who reported no improvements were re-prescribed PPIs in 83.6% (46/55) of cases. Reporting improvements in symptoms to the GP was associated with a 4.6% increase in PPI re-prescriptions (95% CI [-18%, +18%]) in comparison to reporting persistence/recurrence of symptoms, which was not statistically significant.
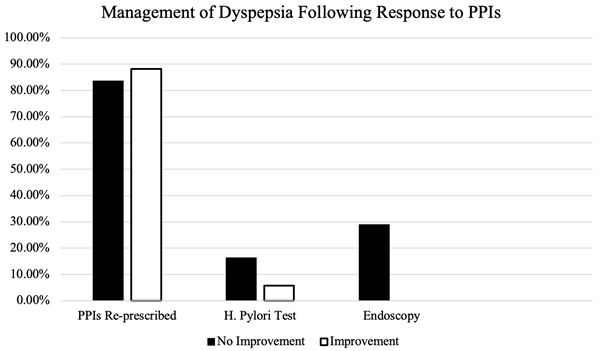
Figure 2: Management of Dyspepsia following response to PPIs
Of the patients who did not improve on PPIs, 29% (16/55) were referred for endoscopy and 16.4% (9/55) were tested for H. pylori. Of those who improved on PPIs, 5.8% (1/17) were tested for H. Pylori and 0% (0/17) was referred for endoscopy.
The total number of patients with un-investigated dyspepsia who were re-prescribed PPIs was 62. 26% (16/62) of them were also referred to either endoscopy or H. Pylori testing.
Summary
The results of this study show that PPI re-prescriptions are high for un-investigated dyspepsia, and that they are not significantly higher in patients who report improvements to PPIs compared to those who do not. These results consolidate the problem of overprescribing PPIs and contribute to narrowing down the causes of over prescription in a GP setting. Of the many factors that lead to overprescribing, patients’ expression of their response to PPIs has no influence on the problem.
Interpretation
Both groups of patients, those who improved on PPIs and those who did not, had high rates of PPI re-prescriptions. The pathway that NICE provides for the management of un-investigated dyspepsia does not advocate the re-prescription of PPIs regardless of a patient’s response to treatment. NICE states that patients who improve should be monitored for symptoms of recurrence, and those who do not should be tested for H. Pylori infection [1]. This study shows that over 80% of patients were re-prescribed PPIs regardless of whether they reported improvements or not. The underlying reasons for these findings are multifactorial.
Firstly, some of these PPI re-prescriptions are justifiable and offer an improvement to the health of patients. For example, 26% of patients who were re-prescribed PPIs were also referred for endoscopy or H. Pylori testing. The likely purpose of these PPI re-prescriptions was to control symptoms until the cause of dyspepsia was identified and treated. GPs may justify the use of PPIs to control dyspeptic symptoms while awaiting further investigations.
Furthermore, GPs might more readily re-prescribe PPIs when under the impression that H. Pylori is not very prevalent in the village of Bildeston. Data suggests that H. Pylori infections are less common in industrialised countries of the western world and more common in low-income countries, indigenous communities, and communities of low socioeconomic status and poor hygiene [6]. Given these facts and the absence of data on the prevalence of H. Pylori in Bildeston, GPs might more readily prescribe PPIs with the assumption that patients are less likely to have an underlying H. Pylori infection. Further evidence to this point is that only 16.4% of patients who did not improve on PPIs were tested for H. pylori. Despite the lack of data on H. Pylori in Bildeston, there is strong evidence suggesting an infection prevalence of above 35% in the UK [3,6].
Another theory arises from how behavior is affected by operant conditioning, specifically through positive and negative reinforcement. Operant conditioning explains behavior as being driven by the consequences of actions. In positive reinforcement, an action is more likely to recur if it is followed by a favorable event. This could explain why doctors re-prescribe PPIs to patients who describe improvements in their symptoms. Furthermore, negative reinforcement describes how an action is more likely to recur if it is followed by the removal of an unfavorable event. If doctors feel burdened by their patients’ suffering, albeit the unfavorable event, then the relief of this burden with a course of PPIs will motivate doctors to repeat this prescription through negative reinforcement. This theory only applies to patients who report improvements on initial PPI therapy and does not help explain the high re-prescription rates in patients who do not improve.
Finally, the high numbers of PPI re-prescriptions could be partly explained by the lack of awareness of the NICE guidelines for the management of un-investigated dyspepsia. Many of the re-prescribed PPIs are of higher dose than the original course. This suggests an attempt to further suppress acid excretion to reduce symptoms without investigating for underlying pathology.
Identifying the reasons for the high PPI re-prescriptions in un-investigated dyspepsia is important for preventing avoidable medication side effects and complications of undiagnosed H. Pylori infection. A review exploring factors that influence prescribing decisions found thirty-three factors. It was shown that “clinical experience”, “cost of treatment”, and “patient preference”, were all more common factors that influence prescribing than “practice guidelines and policies”. Furthermore, “clinical effectiveness and safety” of the medications was as common as “practice guidelines and policies” in influencing prescribing [13]. Therefore, being cheap, effective, and safe are three characteristics of PPIs that will highly favor their prescription.
PPIs could also mask underlying pathology. They effectively reduce symptoms of dyspepsia in patients with H. Pylori [14]. In a study looking at patients on long term PPIs, 31.2% had underlying H. Pylori infections [15]. Given that PPIs control dyspeptic symptoms, and that dyspepsia is often the only symptom of H. Pylori infection, these patients are less likely to seek medical advice to identify the underlying cause of their problems. Not screening for H. Pylori and keeping patients on PPIs instead will result in increased medication dependence and the increased likelihood of developing complications from chronic H. Pylori infection such as gastritis, peptic ulcers, gastric cancer, and lymphoma [7]. Furthermore, treatment of H. Pylori in patients on long term PPIs has shown to reduce PPI prescriptions, PPI dosages, and symptoms of dyspepsia [15].
Strengths and Limitations
One of the strengths of this study is that it explored the effects of a single variable on overprescribing in a very specific setting. Sampling was done over a four-year period which included the COVID-19 pandemic. Limitations include the use of convenience sampling where data was collected from a single GP surgery. It does not allow for generalisability to other surgeries around the United Kingdom. Furthermore, despite successfully identifying the high rate of PPI re-prescriptions, it fails to scientifically explain the reasons behind this phenomenon.
Prescribing is influenced by various factors, not all of which have been explored. This study identifies a specific situation in overprescribing and rules out patient feedback as being a driving cause. While the problem of overprescribing is well documented in the literature, this study highlights its occurrence where guidelines do not encourage it. This raises questions on whether refresher courses on guidelines are indicated and uncovers a new area for quality improvement projects to target.
There was no funding for this research.
members at Bildeston Health Centre who were of great support during this research project.
Ethical risks were considered, and no ethical approval was sought.
Christer Sawaya contributed to the whole of this research project
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
Citation: Sawaya C (2022) The Effect of Patientsâ?? Response to Proton Pump Inhibitors on Medication Re-prescription in Un-investigated Dyspepsia: An Observational Study in General Practice. Qual Prim Care. 30.41792.
Copyright: �© Sawaya C. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.