- (2010) Volume 11, Issue 6
Adam D Toll1, Thomas Kowalski2, David Loren2, Marluce Bibbo1
Departments of 1Pathology, Anatomy, and Cell Biology and 2Gastroenterology and Hepatology, Thomas Jefferson University Hospital. Philadelphia, PA, USA
Received August 25th, 2009 - Accepted September 10th, 2010
Context Recent studies have shown high amplitude K-ras gene mutation and allelic imbalances are predictive of malignancy in pancreatic cysts. Objective Our purpose is to determine the added benefit of molecular testing in diagnosing small pancreatic cysts. Design Retrospective, single-institution study. Patients Patients with pancreatic cysts (less than, or equal to, 3 cm) who presented for EUS evaluation. Intervention EUS-guided pancreatic cyst aspiration cytology, carcinoembryonic antigen (CEA) level determination, and detailed DNA analysis including K-ras gene mutation and allelic imbalance. Main outcome measurements Ability of cyst fluid DNA analysis to render a diagnosis compared with cytology and CEA level determination. Results Diagnostic agreement was seen in 55.6% (35/63) of cases. In 10 cases (15.9%), there was disagreement between cytology and molecular. Molecular testing provided a diagnosis in 20 cases (31.7%) when either cytology was unsatisfactory, or CEA not elevated (less than 192 ng/mL). Elevated CEA levels were seen in 16 cases (25.4%), each diagnosed as a mucinous lesion with molecular analysis. Conclusions Molecular analysis of pancreatic cyst fluid adds diagnostic value in scant specimens when cytology may be unsatisfactory and CEA unreliable.
Molecular Biology; Pancreatic Cyst
Pancreatic cystic neoplasms represent a relatively common pathologic entity affecting at least 1% of medical patients and represent a spectrum of lesions from inflammatory pseudocysts to malignant neoplasms [1, 2]. A significant percentage of these cysts are found incidentally during imaging work-up for unrelated conditions and require appropriate diagnostic testing to characterize the nature of the pancreatic cystic neoplasms [1]. In broad categories, they may be classified as inflammatory, serous, or mucinous. Mucinous cysts are considered premalignant lesions, and may be further subclassified as a mucinous cystic neoplasm or intraductal papillary mucinous neoplasm (IPMN) [3]. A multi- disciplinary approach to characterize pancreatic cystic neoplasms is currently used involving cytology, imaging, and cyst fluid analysis. Several tumor markers within cyst fluid have been evaluated in an attempt to identify mucinous differentiation. Among them carcinoembryonic antigen (CEA) levels above 192 ng/mL were found to demonstrate the greatest accuracy in diagnosing mucinous cysts, however could not distinguish the presence or absence of malignancy [4, 5].
Recent work utilized DNA analysis to characterize pancreatic cystic neoplasms as either mucinous or serous, and assess malignant potential [6, 7]. Focusing on K-ras gene point mutation, this group was able to detect mucinous differentiation (specificity 96%). Further, high amplitude K-ras mutations combined with allelic loss were 96% specific for malignancy [6]. Correlation of K-ras mutation/allelic imbalances with CEA, however, showed poor agreement in the diagnosis of mucinous pancreatic cystic neoplasm [7]. The management of pancreatic cystic neoplasms is still evolving, and current guidelines recommend conservative management for uncomplicated lesions less than 3 cm [8]. The aim of the current study is to determine the added benefit of molecular testing with the currently accepted diagnostic modalities of cytology, imaging, and cyst fluid analysis in the diagnosis of pancreatic cystic neoplasm.
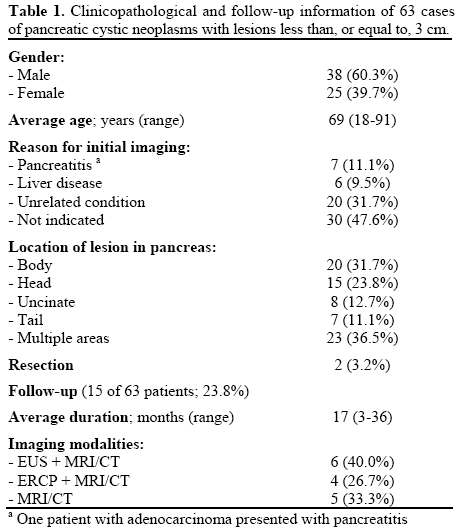
Ninety-one consecutive (from 2007-2010) cases of pancreatic cystic neoplasms that underwent fine-needle aspiration cytology, cyst fluid analysis, and molecular testing were obtained from the Department of Cytopathology. Exclusion criteria included lesions greater than 3 cm, and 63 cases (69.2%) were ultimately included in the study. The indications for the procedure varied from symptomatic to incidentally discovered lesions. Pertinent findings noted by endoscopic ultrasound (EUS) included mural nodules, papillary projections, septations, calcifications, and associated mass lesions. Diagnoses were classified as unsatisfactory, benign non-mucinous, mucinous, and suspicious/malignant. The size and location of the pancreatic cystic neoplasm was determined from imaging studies, either magnetic resonance imaging (MRI), or endoscopic ultrasound (EUS). All patients underwent the same fine needle aspiration (FNA) protocol and specimen analysis without the administration of antibiotics. Following aspiration, a portion of the specimen was sent for molecular and cyst fluid analysis of CEA and amylase. A cut-off of 192 ng/mL had been previously adopted by our laboratory to determine significant CEA elevation. Molecular analysis, however, was not routinely performed on specimens diagnosed as malignant on cytology unless specifically requested by the clinician. The remainder was used for smear slides stained both with Romanowski (Diff-Quik) and Papanicolaou methods, as well as for cell block in a process utilizing Sacomanno fixative (containing ethanol, methanol, isopropyl alcohol and carbowax) and fixation in 10% formalin for a minimum of 6 hours following centrifugation. Following this, sections of 4 μm tissue from formalin fixed, paraffin-embedded blocks were transferred to glass slides and stained with hematoxylin and eosin (H&E).
Cytologic criteria for determining mucinous lesions included the identification of mucinous epithelium that could be distinguished from gastrointestinal contamination. The presence of mucin alone was not sufficient to diagnose a mucinous pancreatic cystic neoplasm. Gastric mucosa exhibited glandular-like formations with round-oval bland nuclei and nonvacuolated cytoplasm. Duodenal mucosa appeared as strips of columnar epithelium with bland nuclei and interspersed goblet cells.
A pancreatic cystic neoplasm diagnosed as mucinous with molecular testing and cytology was considered diagnostic of a mucinous pancreatic cystic neoplasm. If there was discordance between molecular and cytology/cyst fluid analysis, the clinicians would either repeat the FNA or follow the patient closely with serial imaging studies.
Molecular analysis was performed by RedPath Integrated Pathology (PathFinder TGTM; Redpath, Pittsburgh, PA, USA) [6]. The insurer (Medicare or Medicaid; https://www.cms.gov/; https://www.medicare.gov/), or private third-party payer was billed directly for the cost associated with testing ($3,500). K-ras-2 gene point mutation analysis was performed with fluorescent-based direct sequencing of the amplified first exon of the gene. Allelic imbalance/loss of heterozygosity was measured by contrasting polymorphic microsatellite alleles of non-neoplastic specimens against each corresponding marker in pancreatic cystic neoplasm with ratios greater than 2 standard deviations from the mean considered significant. Criteria for mucinous lesions included Kras 2 gene point mutation, high DNA quantity (optical density ratio at wavelength 260/280 greater than 10) or DNA quality, or loss of heterozygosity in 2 or more genomic loci. Criteria for malignancy included K-ras-2 gene mutation, high amplitude (greater than 75%), or 2 or more genomic loci with loss of heterozygosity. Institutional criteria for resection of non-malignant lesions include: mucinous lesions greater than 3 cm or those with significant mural nodules or papillary projections, and non-mucinous lesions with significant symptomatology or patient morbidity.
The study was approved by the Institutional Review Board (IRB) of Thomas Jefferson University Hospital. This was a retrospective data analysis, and the hospital IRB did not require patient consent. The study protocol conforms to the ethical guidelines of the “World Medical Association Declaration of Helsinki - Ethical Principles for Medical Research Involving Human Subjects” adopted by the 18th WMA General Assembly, Helsinki, Finland, June 1964, as revised in Tokyo 2004.
The incremental benefit of adding molecular testing was analyzed with McNemar’s test. Two-tailed P values less than 0.05 were considered statistically significant.
The results of the study are summarized in Tables 1 and 2. There were no major (e.g. pancreatitis, hemorrhage), or minor (e.g. fever) peri- or postprocedural complications. The final study group was 60.3% male and 39.7% female. Diagnostic agreement was seen in 55.6% (35/63) of cases; 20 (57.1%) were mucinous, 12 (34.3%) benign non-mucinous, and 3 (8.6%) suspicious/malignant. In 10 cases (15.9%), there was disagreement between cytology and molecular analysis. Among cases with disagreement, CEA levels supported the molecular diagnosis in 4 cases, and the cytologic diagnosis in 2 cases (4 cases did not have sufficient material for cyst fluid analysis). Cytology was unsatisfactory in 16 cases (25.4%) where molecular analysis was able to render a diagnosis. In 2 cases (3.2%), both cytology and molecular analysis were deemed unsatisfactory.
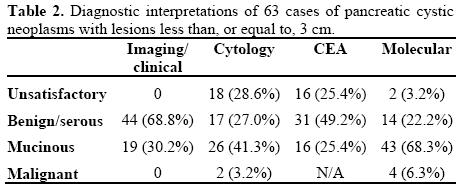
Elevated CEA levels (greater than 192 ng/mL) were seen in 16 cases (25.4%), each diagnosed as a mucinous lesion with molecular analysis. In 4 cases (6.3%) CEA was elevated when cytology was unsatisfactory, each diagnosed as mucinous cyst with molecular. Amylase levels averaged 42,102 U/L (range: 1-189,600 U/L; reference range: 0-132 U/L) in non-mucinous pancreatic cystic neoplasms, and 16,541 U/L (range 2-141,324 U/L) in mucinous pancreatic cystic neoplasms.
Imaging studies along with clinical impression favored a diagnosis of mucinous pancreatic cystic neoplasm in 19/63 (30.2%) cases (15 IPMN, 4 mucinous cystic neoplasms). Among these, 16/19 (84.2%) showed agreement with molecular analysis. Three cases showed the presence of mural nodules, cytology and molecular diagnosed two cases as benign/serous, and one case as mucinous. Branch-duct dilatation was noted in one case (not resected). No cases demonstrated main duct dilatation or mass lesions. In 10 cases (15.9%), molecular diagnosed a mucinous lesion while EUS favored a benign serous/ inflammatory cyst.
Two patients underwent pylorus-preserving pancreaticoduodenectomy, one diagnosed histologically as chronic pancreatitis, the other as adenocarcinoma arising in association with IPMN (surgical margins were free of tumor). In both cases molecular analysis correctly identified the lesion, while cytology was unsatisfactory in the case of chronic pancreatitis, and initially diagnosed a benign mucinous lesion in the case of adenocarcinoma (repeat FNA was performed following the diagnosis of malignancy by molecular analysis, and repeat cytology diagnosed the lesion as suspicious for malignancy).
Molecular analysis diagnosed a mucinous lesion in 8 cases (12.7%) when CEA levels were not elevated. In 3 cases (4.8%), a mucinous lesion diagnosis was made by molecular analysis when both cytology was unsatisfactory and CEA not elevated. In 20 cases (31.7%), a diagnosis was made by molecular analysis when either cytology was unsatisfactory, or CEA not elevated.
Analysis of McNemar’s test demonstrated a statistically significant (P=0.001) benefit with regard to the ability of molecular analysis to aid in providing a diagnosis when compared to cytology (61/63, 96.8% vs. 44/63, 69.8%). This value was also significant when applying the criteria of elevated CEA to identify a mucinous pancreatic cystic neoplasm (P=0.010; 24/63, 38.1% vs. 16/63, 25.4%).
The results of our study demonstrate the addition of molecular analysis significantly increases the diagnostic yield of pancreatic cystic neoplasms less then, or equal to, 3 cm when used in conjunction with cytology and cyst fluid CEA levels. A significant advantage molecular analysis holds over other testing modalities is the amount of specimen required for diagnosis. Although variable, fine-needle aspiration cytology requires several mL of specimen, and cyst fluid analysis requires at least 1 mL, while molecular testing requires only 200 μL [6]. This becomes important in scant specimens when cytology is likely to be unsatisfactory (29% of our cases), and CEA unreliable.
The rationale for utilizing elevated CEA levels to identify mucinous epithelium derives from the embryologic origin of endoderm-derived columnar epithelium being able to secrete CEA [8]. Our results showed poor agreement between CEA and molecular analysis, consistent with previous work with regard to correlating these diagnostic modalities [8]. This finding was previously attributed to the requirement for lining cells to secrete CEA, while molecular analysis depends on these same lining cells to acquire specific mutations. Amylase levels were also examined in the current study, and although they averaged higher values in non-mucinous pancreatic cystic neoplasms, the results were not significant. Cyst fluid analysis may be helpful, however must be interpreted in the context of multiple diagnostic modalities.
Molecular testing has been examined as a potential adjunct diagnostic test due in part to the poor sensitivity/specificity of cytology and CEA for mucinous pancreatic cystic neoplasms. Elevated CEA levels were considered the most accurate test for mucinous differentiation in pancreatic cystic neoplasms, however show only 79% accuracy for these lesions [4]. Fine-needle aspiration cytology is less reliable as a result of the low cellularity inherent in pancreatic cystic neoplasms, however may be improved with intracystic cytobrushing [9]. Molecular analysis provides information regarding several factors pertaining to malignancy. Most notably they examine K-ras mutational status, recognized as the most commonly mutated gene in pancreatic cancer, as well as identifying the allelic imbalances responsible for inactivating tumor suppressor genes [7, 10]. Our results are consistent with these observations, lesions with a K-ras point mutation were only found in mucinous pancreatic cystic neoplasms. Through interrogation of multiple genetic factors associated with malignant transformation, molecular analysis has shown great promise as an aid to the current diagnostic regimens of pancreatic cystic neoplasms. Importantly, molecular analysis cannot be used as a singular diagnostic modality in pancreatic cystic neoplasms. A recent study found significant numbers of false positive and negative results when comparing molecular analysis to CEA with histology [11]. Another limitation of molecular analysis is the inability to subclassify mucinous lesions as either IPMN or mucinous cystic neoplasms. Correlation with clinical history, cytology, and endoscopic findings are critical in this regard. A recently published smaller study examined pancreatic cystic neoplasms of all sizes (range: 0.3-7.6 cm) and found diagnostic concordance between molecular analysis and currently used diagnostic tests [12]. Our study expanded on this work by utilizing a larger sample size, and focusing on smaller pancreatic cystic neoplasms where molecular analysis should have the greatest benefit.
An example of the diagnostic sensitivity of molecular analysis is illustrated by a case in our study initially diagnosed as a mucinous lesion on cytology, while molecular analysis diagnosed malignancy (imaging studies favored a branch-duct IPMN). In view of the molecular findings, a repeat FNA was performed, and cytology now interpreted the lesion as suspicious for adenocarcinoma. A subsequent surgical resection revealed adenocarcinoma arising in association with an IPMN. In this case appropriate clinical management occurred as a direct result of molecular analysis.
The management of pancreatic cystic neoplasms is evolving, and while current guidelines suggest conservative management for uncomplicated serous and branch-duct IPMN less than 3 cm, debate continues regarding optimizing care for these patients [1, 9, 13]. The size cut-off of 3 cm is based on analysis of several studies, one of the largest coming from Memorial Sloan-Kettering Cancer Center demonstrating a malignancy risk of 3% in mucinous lesions less than 3 cm, a figure only marginally greater than the risk of significant morbidity/mortality from undergoing pancreatic resection [1, 13, 14, 15]. In practice, the decision to pursue surgical resection is multi-factorial, and studies have stressed the importance of life expectancy along with the surgical risk of the patient as a way to guide management [16]. A newer approach to the clinical management of pancreatic cystic neoplasms involves non-surgical ablation with 80% ethanol lavage [17, 18]. The theory behind this procedure is based on speculation the ethanol can sclerose the cyst lining, preventing further accumulation of fluid. Some pertinent issues need to be addressed before ethanol lavage becomes widely utilized and include inability to treat multiloculated cysts, and post-ablation complication rates between 12-16% [17]. Ideally, the use of molecular analysis will provide clinicians with a better idea of which patients are at greatest risk for malignant transformation, and who would therefore benefit the most from early surgical intervention or ablation. An important limitation of our study is the lack of surgical resections to correlate with diagnostic testing. This was expected given the conservative management currently recommended for a significant number of these lesions (Figure 1). Without histologic correlation, it is not possible to determine whether the addition of molecular testing improves the diagnostic accuracy of pancreatic cystic neoplasms. The results of the current study show the addition of molecular testing improves the diagnostic yield of pancreatic FNA in scant specimens. Further, specimens diagnosed as malignant by cytology at our institution are not routinely sent for molecular analysis. Long-term follow-up studies correlating molecular analysis with the most recent consensus guidelines in regard to malignant transformation and overall prognosis in pancreatic cystic neoplasm are required to more completely define the role of this new diagnostic modality.
In summary, we have presented data demonstrating molecular analysis adds to the diagnostic sensitivity of pancreatic FNA. This benefit becomes even more pronounced in scant specimens when cytology may be unsatisfactory and CEA unreliable.
Conflicts of interest and funding None