Review Article - (2017) Volume 3, Issue 3
Simon S Chiu1*, Michel Woodbury-Farina2, Kristen Terpstra1, Vladimir Badmaev3, Zack Cernovsky1, Yves Bureau1, Jerry Jirui1, Hana Raheb1, Mariwan Husni1,4, John Copen5, Mujeeb Shad6, Amresh Srivastava1, Veronica Sanchez1,7, Marissa Williams1,8, Zahra Khazaeipool1, Autumn Carriere1,9 and Christina Chehade1
1Lawson Health Research Institute, Department of Psychiatry, University Western Ontario, London, ON, Canada
2School of Medicine, University of Puerto Rico, Puerto Rico, PR, USA
3Medical Holdings Inc., New York, NY, USA
4Northern Ontario Medical School, Thunder Bay, ON, Canada
5Victoria Medical Campus, University of British Columbia, Victoria, BC, Canada
6Oregon Health Sciences University, Portland, Oregon, USA
7Biomedical Sciences, McGill University, Montreal, PQ, Canada
8Currently PhD Program, York University, ON, Canada
9Graduate Program Kinesology, Nipissing University, North Bay, ON, Canada
*Corresponding Author:
Simon S Chiu
Lawson Health Research Institute, London Ontario
University Western Ontario, London, ON, Canada.
Tel: 1-519-631-8510
E-mail: schiu3207@rogers.com
Received date: August 07, 2017; Accepted date: August 30, 2017; Published date: September 07, 2017
Citation: Chiu SS, Woodbury-Farina M, Terpstra K, Badmaev V, Cernovsky Z, et al. (2017) Targeting Epigenetics Signaling with Curcumin: A Transformative Drug Lead in Treatment of Schizophrenia? J Clin Epigenet. 3:32. doi: 10.21767/2472-1158.100066
Despite advances in pharmacotherapy, schizophrenia continues to carry a high societal disease burden related to positive and negative symptoms, and neurocognitive deficits and severe functional impairment. Findings from epigenetics have yet to translate the epigenetics targets to efficacious drug therapy. We review the repertoire of CNS pharmacology of curcumin derived from Curcuma Longa, commonly known as “turmeric”: the well-known curry extract. We highlight the body of evidence in support of the emerging role of curcumin as a panhistone deacetylase (HDAC) inhibitor regulating the expression of genes involved in inflammation and NMDA N-methyl-aspartic acid (NMDA)-glutamate systems, as related to schizophrenia. Based on the findings from translational studies of curcumin extracts, curcumin C-3 complex formulation (Supercurcumin™) and patented liposome-based curcumin (Lipocurc™), we propose that intravenous infusion of Lipocurc™ holds promise as the novel drug lead and preferred targeted brain delivery mode in reprogramming faculty epigenetics network and in remodeling restrictive chromatin configuration in schizophrenia. Phase II/Phase III epigenetics-biomarker-based randomized placebo-controlled trials in treatment resistant schizophrenia are warranted. Our approach of intravenous infusion of Lipocurc™ behaving as pan HDAC inhibitor represents a new paradigm of drug development in combining targeting epigenetics footprints with brain-specific drug delivery system in schizophrenia. Lipocurc™ can open the Pandora box in unexplored therapeutics vistas in modifying the phenotype of treatment resistant schizophrenia.
Keywords
Epigenetics; Histone deacetylase inhibitor; Schizophrenia; Curcumin; Liposome
Epigenetics Signaling in Schizophrenia
While pharmacotherapy of second generation of atypical antipsychotics: clozapine, olanzapine, risperidol, quetiapine, ziprasidone, aripiprazole bring about significant improvement in the positive symptoms of schizophrenia [1], overall psychosocial outcomes in schizophrenia remain modest. Significant functional impairment related to negative symptoms (lack of volition, apathy, sociality, poverty of speech and affective flattening), active positive symptoms (delusions, hallucinations and bizarre behavioral disturbances) and neuro-cognition deficits, persists in 20-30% of patients with schizophrenia refractory to conventional trials of antipsychotic therapy. Treatment Resistant Schizophrenia (TRS) carries a very high disease burden on family and society at large [2]. Major breakthroughs in schizophrenia therapeutics are yet forthcoming. It becomes imperative to design molecular genetics and epigenetic tools to predict treatment responses, and to translate novel CNS drug targets for accelerating the development of new therapeutics for treatment refractory schizophrenia. Drug platforms require refinement of translational models of schizophrenia to catalyze clinical trials and to predict treatment responses. In the post-genomic era, deciphering the epigenetic code of the human genomes represents a major challenge in CNS disorders. A growing body of evidence supports dysregulation of epigenetics signaling: DNA methylation, histone modifications and microRNA, plays a significant role in schizophrenia [3-5]. Findings from studies of postmortem brains agreed largely with the lymphocytes from schizophrenia subjects and strongly suggest the shift in the epigenome towards restricted transcription in schizophrenia [6-8]. The specific epigenetic marker, H3K9me2 regulated by Histone Methyltransferase (HMT) catalyzing the insertion of two methyl groups to the 9th lysine residues of the histone 3 within the nucleosome complex, was significantly elevated. The phenotype of restrictive epigenome schizophrenia results in inactivation of transcription of multiple candidate genes related to neural plasticity and neuro-inflammation and neurotransmitter regulation. The restrictive epigenome in schizophrenia favored the male gender exhibiting higher levels of HMT and the histone protein H3K9me2 protein levels [9], which correlated with higher levels of symptom severity. The repressive and restricted nature of epigenomes with condensed chromatin configuration is localized and sensitive towards the macro- and micro-environmental milieu.
Up-regulation of DNMT expression results in repressed chromatin conformation associated with altered HDAC activities. There is growing body of evidence in support of the pivotal role of altered DNA methylation processes differentially affecting the expression of several schizophrenia genes. Aberrant methylation patterns of Brain-derived Neurotrophic Factor (BDNF) gene and Glucocorticoid Receptor (GCR) gene and Glutamic acid decarboxylase67 [GAD67], reelin gene, primarily at the promoter regions in schizophrenia have been reported [8,9]. The two key enzyme pathways linked to epigenetic reprogramming and gene expression regulation: 5-hydroxymethycytosine (5-OH-CT) and the ten-eleven Translocation Protein (TET) were up-regulated, along with enhanced expression of BDNF, GCR and GAD57 genes especially in the prefrontal cortex and in the hippocampus [10]. Lymphocytes from schizophrenia patients parallel altercations changes in TET and 5-OH-CT activities in postmortem brain giving rise to striatal-limbic-cortical brain network [11]. The highly correlated patterns of DNA methylation in both the brain tissues and peripheral blood samples validate model of common epigenetic dysregulation pathway extending across brain-brain barrier to the peripheral blood elements: lymphocytes and platelets in schizophrenia.
From Curry Extract to Curcumin as the Master Epigenetic Modulator
Turmeric extracts from the rhizome of Curcuma longata, have been used orally as a condiment, commonly known as curry powder and extract, and gained respectable status as a medicinal herb for a variety of medical conditions including arthritis and gastrointestinal problems, in India and Asia for several thousand years (Figure 1). The physico-chemical properties of curcumin have been fully characterized. Curcumin belongs to the class of polyphenols and is chemically known as diferuloylmethane. The anti-carcinogenic properties of curcumin have been well studied and characterized [12]. We critically review the studies related to the anti-inflammatory property of curcumin, highlight the multi-target CNS pharmacology of curcumin, and evaluate the strength of evidence from pharmacological studies of curcumin supporting the model that epigenetics signaling orchestrates the potent central actions of curcumin in regulating NMDA and neuro-inflammatory pathways in schizophrenia.
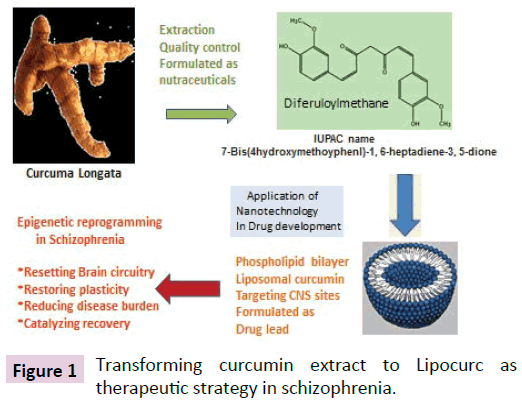
Figure 1: Transforming curcumin extract to Lipocurc as therapeutic strategy in schizophrenia.
Inflammation pathway
Curcumin has been shown to modulate multiple cell signaling pathways and interact with numerous molecular targets, including cell cycle, apoptosis, proliferation, survival, invasion, angiogenesis and inflammation (Nuclear factor: NF-κB, Tumor necrosis factor: NF, interleukins IL-6, IL-1, cyclooxygenase:COX-2 and lipoxygenase: LOX). With recent heightened interest in epigenetics signaling as cancer biomarker and therapeutics, curcumin commands a respectful edge in cancer research. In vitro and in vivo cancer models, HDAC (Histone deacetylase) modulates histone modification signaling via regulating Histone Deacetylase (HDAC) and HAT (histone acetyltransferase) activities, and DNA methylation [13]. It is noteworthy that curcumin regulates the DNA methylation dynamics through inhibiting DNA-methyltransferase (DNMT) activities in various model system [14,15]. Progress in advancing curcumin from naturally occurring phytochemicals to forefront of cancer chemotherapy has been relatively slow due to multiple factors relating to upscaling manufacturing and extraction and synthesis and the variable bioavailability. Clinical studies of various formulation of curcumin have been conducted in various cancers with promising findings. No serious adverse events are reported. We are not aware of any pipeline curcumin formulations approved by (US) FDA for therapeutic indications in cancer chemotherapy.
Recent interest has been drawn towards neuro-inflammation as the mechanisms underlying changes in brain abnormalities in white and grey matter volume loss and impaired regional connectivity in parallel with the neuro-progressive course in the earliest stage of the disease marked with relapse episodes [16]. At the cellular level, imaging and neuropathological studies find evidence for the emerging role of microglia, the resident immune cells of the brain: increased density and activation of microglia [17,18]. In this respect, microglia modulates the inflammatory response through alerting the immune sensors of the family of cytokines [19]. Hence, microglia stands at the cross-roads of epigenetics-mediated neuro-inflammation and immunity regulation, and redefines schizophrenia with respect to aberrant release of cytokines as a result of dysregulation of immunity and neuro-inflammation. Both positive and negative findings have been reported on up-regulation of the translocator protein 18 kDa (TSPO), a marker of activated glia, in schizophrenia [19-22]. Studies on the neurobiology of inflammation have unraveled neuro-inflammation as the potential target in the treatment of schizophrenia. The celecoxib families of anti-inflammatory drugs have been shown to produce promising positive results in early stage of schizophrenia. It appears highly likely that antipsychotics exert their therapeutic effects through the neuroinflammatory signaling pathway [23]. It is perhaps fortuitous that the majority of phytochemicals exert their multiple actions: anti-inflammatory, anti-oxidant and neuroprotective actions through reprogramming synaptic plasticity [24]. Several lines of evidence suggest that curcumin possesses anti-inflammatory properties in translational models of CNS dysfunction. In the lipopolysaccharide-induced model of inflammation, rodents exhibited anxiety and depressive phenotypes which can be correlated directly with increased levels of pro-inflammatory cytokines: IL- IL-1β and TNF-α and oxidative stress indices of glutathione depletion in the hippocampus [25]. A recent review summarized the potent effects of curcumin on the balance between pro-inflammatory and anti-inflammatory phenotypes of cytokines in various biological systems [26]. In the primary rodent glial cultures, curcumin exerted significant protection against glial viability under conditions of severe pro-inflammatory and oxidative stress [27,28]. In an in vitro study using the Liposaccharide (LPS)-induced culture milieu, curcumin reduced the microglial pro-inflammatory biomarkers; inducible nitric oxide synthetase: iNOS and tumor necrosis factor: TNF and increased the anti-inflammatory cytokine IL-4. The property of curcumin in restoring the polarization of inflammatory phenotypes towards anti-inflammatory mode makes it an appealing lead compound in managing psychosis episodes in schizophrenia. Taken together, curcumin as epigenetic modulator is well positioned to maintain a fine balance of the polarization phenomenon between the proand anti-inflammatory phenotypes of cytokines at the neuroimmune interface in schizophrenia.
NMDA-glutamate signaling and curcumin
Recently, aberrant signaling of N-methyl-d-aspartate (NMDA)- glutamate has been associated with positive, negative and cognitive deficits in schizophrenia [29]. Postmortem brain studies have identified altered expression of the cluster of structural and signaling molecules of the postsynaptic density (PSD) which bind directly of the NR2 subunits of NMDA system [30], with adverse consequences on the NDMD-mediated trafficking, membrane expression and downstream signaling pathways. There is preliminary evidence suggesting that curcumin-mediated neuroprotection and modulation of neuro-inflammation target the NMDA system. Stress is known to be a major determinant in relapse in schizophrenia. A recent study showed that curcumin can buffer against the deleterious effects of stress. In restraint stress paradigm in rodents, curcumin rescued the adverse changes in the dendritic morphology of CA3 pyramidal neurons in the hippocampus, and protected hippocampal neurons against NMDA-NR-2B upregulation in the presence of corticosterone [31]. In the primary retinal and hippocampal neuronal culture using whole- call patch-clamp, curcumin treatment protected against NMDA cell death through reduction of NMDA- induced [Ca(2+)] (i) current rise: the kinetics was time- and dose-dependent. Furthermore, curcumin neuroprotection correlated significantly with increased activity of NMDA-NR2A level.
In schizophrenia, the reduced life expectancy carries higher cardio-metabolic risks compared to the general population [32]. Pharmacotherapy focusing on the cross-talks of metabolic pathways and neural plasticity has met with marginal progress in improving the general well-being of patients with schizophrenia. Glycemic control, for example, has been found to influence cognitive outcome in schizophrenia. In the streptotocin induced diabetes model, curcumin on protection against NMDA neurotoxicity were found in the streptotocin induced diabetes model [33]. Curcumin reversed glutamate accumulation and the enhanced density of NMDA receptor density and the decreased glutamate aspartate transporter expression in the cerebral cortex. There is a growing body of evidence of the beneficial effects of curcumin in metabolic syndrome in both preclinical and clinical studies [34]. Hence, the neuroprotective actions of curcumin are synergistic with its beneficial effects in counteracting the negative CNS consequences of metabolic syndrome which is often related to the current antipsychotic therapy in schizophrenia.
Recently, NMDA-glutamate system has drawn much attention in the treatment of refractory depression: a series of controlled studies of ketamine therapy delivered through the intravenous route in treatment resistant depression have shown highly promising results in the anti-depressant efficacy [35]. We consider it highly relevant to interpret the known interactions of curcumin’s interaction with NMDA- NR1 and NMDA-NR2 [36]. NMDA receptor complex is under dual regulation and control through diversity and interaction among subunits: NR1 and NR2 [36-38]. At the NMDA-NR2 complex, ketamine functions as NMDA antagonist. In contrast, curcumin behaves as a bidirectional allosteric modulator of NMDA and antagonizes the CNS effects of ketamine as shown in the ketamine-induced mania model [39]. In rodents, curcumin pretreatment at 20 mg/kg and 50 mg/kg for 2 weeks blocked the hyperactivity and oxidant stress in the ketamine mania model [40]. In the forced swimming test of rodent “depression-hopelessness”, curcumin behaves as an antidepressant whose anti-depressant property is mediated through NMDA-NR2 [41]. Blockade of NMDA-NR2 by the specific antagonist, CPP reversed curcumin’s behavioral effects. Pre-treatment with curcumin blocked LPS-induced microglial activation and the excessive release of interleukin-1β and tumor necrosis factor-α, along with the levels of inducible nitric oxide synthase and cyclooxygenase-2 mRNA in the hippocampus and Prefrontal Cortex (PFC) [26].
Converging pharmacological evidence supports the role of curcumin as an allosteric modulator of NMDA via resetting the set-point of the functional level of NMDA-R (NMDA-receptor). Curcumin as an allosteric NMDA-R modulator is active in the ketamine model of schizophrenia reflecting NMDA-R hypofunction [42]. Studies to examine the augmenting effect of the NMDA-R non-competitive antagonist, meantime originally indicated for Alzheimer’s dementia, have met with discrepant findings in schizophrenia [43-45].
The neuroprotective action of curcumin extends beyond NMDA to the substantia nigra-striatal dopamine system. In preclinical models of Parkinson disease: DJ-1 gene knockout, 6-hydroxydopamine- lesion and MPTP model, curcumin has consistently been demonstrated to rescue the loss of tyrosine hydroxylase-immunoreactivity fibers through its antiinflammatory and antioxidant action [46,47]. In vitro, curcumin inhibits aggregation and toxicity of alpha-syncline [48]. It is noteworthy none of the 1st and 2nd generation of antipsychotics exhibit dual antipsychotic, anti- Parkinsonian and anti-dyskinesia properties. Taken together, curcumin’s pharmacological profile differs from meantime in modulating the sensitivity of NMDA-R subunits: NR-1/NR-2 interaction through the allosteric site and hence rescuing the vulnerable brains in schizophrenia from the devastating consequences of neuro-inflammation through modifying the cytokine network. The multi-faceted CNS pharmacology of curcumin finds a favorable niche in interacting with the key targets in the brain and in the periphery in schizophrenia.
Epigenetics footprints and Curcumin
In the post-genomic era, there is growing body of evidence in support of epigenetics signaling regulating expression of genes involved inflammation in schizophrenia [15,49-51]. Curcumin behaves as an epigenetic modulator in targeting epigenetic signal pathways of inhibiting HDAC (Histone deacetylase) and modifying the expression of non-coding miRNA. A recent study found that two miRNAs, miR-566 and miR-346, target more schizophrenia-associated genes than would be expected by chance [52]. The emerging role of HDAC in shaping the spinoffs of neuroinflammation on brain and behavior has becoming a matter of heightened interest. In models of vascular injury, HDAC inhibitors confer protection against ischemia-induced oligodendrocyte loss through interacting with multiple pathways including oligodendrogenesis, anti-inflammation, and caspase-3 down- regulation [53]. White matter loss and dysregulation of myelin modelling has been shown to contribute towards schizophrenia. Curcumin has been identified as a potent modifier of epigenetics signal pathways at multiple sites: HDAC (Histone Deacetylase) isoforms 1, 3, 8, HAT (histone acetyltransferase), non-coding RNA miRNA-22, miRNA186a and miRNA-199a (Figure 2) [54,55]. In high throughput epigenetic screening assay using HeLa nuclear extract, curcumin was found to be more potent in inhibiting HDAC than valproic acid and sodium butyrate [56]. The inhibition constant Ki of curcumin (539 nM) was comparable to Ki of Trichostatin A (504 nM). Curcumin is more potent than valproic acid (Ki 564 nM) and sodium butyrate (Ki 365 nM). Cross talks of HDAC with miRNA most likely exert synergistic effects in orchestrating and coordinating multiple gene expression. HDAC inhibitors control epigenetic programming involved in may alter the course of schizophrenia through remodeling chromatin, histone-related modifications. Discrepant findings- have been reported on the effects of valproic acid in preventing relapse and in improving the negative symptoms in schizophrenia [57]. In considering the therapeutic relevance of HDAC and HAT in schizophrenia, four main classes of HDAC have been characterized: the zinc-dependent HDAC I, II, IV and the NADdependent class III HDAC [58]. The structural diversity of HDACs most likely makes HDAC a highly attractive drug target for schizophrenia. Isoform-specific HDAC inhibitors: HDAC class I/ II inhibitors have drawn much interest in drug development. Studies in epigenetics have spinoffs in facilitating new drugs in treatment of schizophrenia. Very few investigators are aware of the mechanisms of action of the benzamide series of HDAC: amilosulpiride. As Class I HDAC inhibitor, amilosulpride is currently approved in Europe, United Kingdom and China for treatment of schizophrenia [59]. Amilosulpride exhibits dual activities in exhibiting potent antagonist activity at striatal dopamine: DA receptor subtypes D2/D3, with selectivity directed towards the short-arm of D2 receptors in the thalamus [60]. Furthermore, amisulpride behaves as a selective antagonist at serotonin-receptor subtype 7: (5HT-7) mediating the likely pro-cognitive action of amisulpride [61]. Amilosulpride exhibits dual activities in interacting with both dopamine and serotonin receptor subtypes. Amilosulpride possesses potent antagonist activity at striatal DA subtypes D2/D3, with selectivity directed towards the short-arm of D2 receptors in the thalamus. On the other hand, amisulpride behaves as a selective antagonist at the serotonin-receptor subtype 7: (5HT-7) mediating its likely procognitive action [62]. The pharmacological profile of amisulpride raises the intriguing hypothesis whether complex interactions at the D2/D3 and 5-HT-7 receptors reflect altered expression of D2/ D3 and 5HT (7) genes because of potent HDAC inhibition.
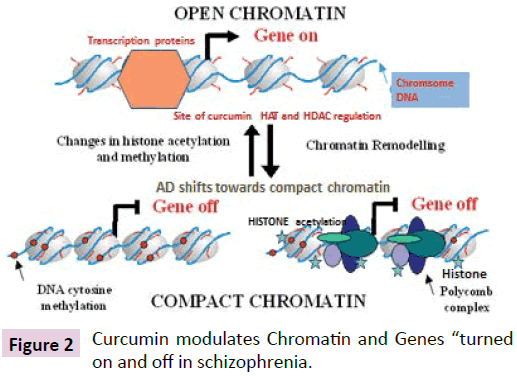
Figure 2: Curcumin modulates Chromatin and Genes “turned on and off in schizophrenia.
As an atypical antipsychotic, amisulpride treatment results in larger effect size than first and second generation of antipsychotics with efficacy equal to olanzapine and risperidone in large scale RCT in schizophrenia [63]. A recent meta-analysis of randomized controlled trials to compare 15 antipsychotic drugs versus placebo in acute treatment of schizophrenia showed that amilosulpride ranked second to clozapine in the efficacy measure of standardized mean differences with 95% credible intervals clozapine 0.88, 0.73–1.03; amilosulpride 0.66, 0.53– 0.78. Amisulpride treatment results in larger effect size than 1st and 2nd antipsychotics with efficacy equal to olanzapine and risperidone in large scale RCT in schizophrenia. No adverse events are reported when amisulpride is combined with clozapine in treatment resistant schizophrenia. We note that none of the studies employed pharmacodynamics engagement biomarkers derived from epigenetics molecular footprints to correlate or to predict treatment responses Pharmacy- genomics embracing epigenomics has become a rapidly growing field in predicting treatment response in schizophrenia. Since schizophrenia is a complex, neuro-psychiatric disorder embedded with dysregulation in multiple epigenetics-regulated signal pathways reflecting the interface of nature-nurture diathesis at the cellular and molecular levels: isoform-selective HDAC inhibitor: HDAC-6 inhibitor may not offer superior therapeutic advantages over pan-HDAC inhibitors. Hitherto, there is insufficient evidence to conclude whether isoform- selective HDAC inhibitors or multitarget HDAC inhibitors translate to better clinical efficacy and safety and tolerability, especially in the CNS disorders [64].
Translational studies with Curcumin C-3 complex and Lipocurc™
Our approach is to repurpose Lipocurc™ (patented formulation of liposome-based curcumin: Sign Path Pharm. PA USA as originally approved by FDA: USA for Phase II clinical trials in cancer) earmarked for cancer chemotherapy for adjunct treatment in schizophrenia. The variable and limited systemic bioavailability of oral curcumin formulations has been the major barrier in advancing studies to examine critically the likely therapeutic benefits of curcumin. Combining curcumin with piperine extracted from pepper has been found to greatly enhance the oral bioavailability of curcumin. We have recently completed a clinical study of a standardized formulation of curcumin: curcumin C-3 complex combined with piperine in schizophrenia [65]. In the study, we investigated the efficacy of standardized Curcumin C-3 complex combined with Bioperine™ (Supercurcumin™) SC in modifying core symptoms and cognitive deficits in schizophrenia. We randomized patients diagnosed as schizophrenia with persistent negative symptoms into Group I and Group 2 treated with 1 g and 4 g of Supercurcumin™ daily oral dosages respectively for 16 weeks. In our small cohort, 17 subjects with mean age: 39·9 years, male/female: 13/4):15/17 subjects completed the study. We found that Group 1(1 g-SC) and Group II (4 g-SC) significantly (P<0.05) improved total and general psychopathology sub-scales pf PANSS: (Positive and Negative Symptoms scale). Within group pre- and post-treatment comparison showed that the mean changes in total PANSS score and PANSS: general psychopathology were significant for both Group 1 and Group 2 (p< 0.05). Cohen d' effect size favored SC in PANSS-positive and negative subscales. Both groups exhibited neurocognitive improvement in selected cognitive domains. SC was well tolerated with no serious adverse events. Our findings highlight targeting histone-mediated epigenetics signaling offers novel roadmap in treatment resistant schizophrenia. Recent interest has been drawn towards exploring novel methods of drug delivery to specifically target CNS sites for CNS Drug development. Nanotechnology-based drug development platform has gradually found its niche in drug development in CNS disorders. Phospholipid-derived liposomes have been found to be safe and highly tolerated in translational models of neurodegenerative and neuropsychiatric disorders [66]. Sign Path Pharm has succeeded to formulate a liposome-based curcumin through contracting with accredited laboratories in Austria. We have demonstrated in the rodent and canine species that intravenous Lipocurc passed through the blood brain barrier (68, 69, and 70). Lipocurc™ treatment significantly improved motor impairment compared with saline treatment. Lipocurc™ significantly blocked neuronal apoptosis: the apoptotic index of DJ-1-KO-Lipocurc™ group was markedly reduced compared with the placebo group in the DJ-1 gene knockout rat model of Parkinson’s disease (3.3 vs 25.0, p<0.001) [47]. We interpret the findings in the light of the capacity of curcumin in stimulating DA neurons in the substantia nigra [47]. Very few antipsychotics exhibit concomitant anti- Parkinsonian properties. In addition, we have shown that in the DJ-1 gene knockout rat model of Parkinson disease. Lipocurc™ partially restored the motor deficits and significantly improved cell survival in the striatum with reduced apoptosis.
Our research group completed both pharmacokinetic studies and the 4-week toxicity studies in healthy control rodents and in the canine species [67-70]. According to the protocol, rats were given intravenous bolus injections three times a week for 4 weeks (empty liposomes, and 10, 20 and 40 mg/kg Lipocurc™) The dogs were given I hour intravenous infusions at 10 mL/kg/h three times a week for 4 weeks (5% dextrose in water, empty liposomes, and 5 and 20 mg/kg Lipocurc™). For the rodent species, there were no deaths on the study, and no changes in the clinical signs, body weight, food consumption, clinical chemistries or organ weights, and no treatment-related effects on gross or microscopic pathology. Red blood cell parameters began to decrease for all groups, including the empty liposomes group, with a nadir during Week 2 and bounced back and normalized by 96 hrs. Treatment with empty liposomes had the least hematological effect, with a dose-response effect of the Lipocurc.
In view of the wide safety margin, the NOAEL (no-observedadverse- effect level) defined as "the highest experimental point that is without adverse effect", has been determined for the rodent species to be greater than 40 mg/kg. For the beagle dogs treated with 5 mg/kg Lipocurc™, the analysis of all generated aggregated data: clinical observations, ophthalmology, ECGs, clinical pathology, gross pathology and histopathology likewise concluded no treatment-related toxicity. In the canine species, hypersensitivity reaction occurred in all dogs infused with empty liposomes and in only one animal dosed with Lipocurc™ at 20 mg/kg. At5 mg/kg none of animals in the group infused with Lipocurc™ was affected. The trajectories from combining the standards of nanotechnology-based mode of drug delivery, and the requirements of US-FDA in meeting the pharmaceutical-grade standards, have shaped new directions in drug development. The smooth transition from extraction of Curcuma Longata to synthesis and the multi-step in the preclinical toxicological and safety evaluation of Lipocurc™, has set the ideal stage for seeking formal FDA approval for embarking upon Phase I study. The creative trajectories from extraction of curry extract to exploiting nanotechnology to formulating pharmaceutical grade of Lipocurc are summarized in a schematically in the Figure 1.
First-in-Human Phase I Trial of Lipocurc™
Recently Phase 1 clinical study with Lipocurc™ in normal healthy subjects was completed: the results showed highly favorable pharmacokinetics and tolerability of Lipocurc™ with very few adverse events [71]. This was a dose escalation study of single dosage infusion of Lipocurc™ over 2 hrs in healthy volunteers over 9 IV dosage levels: 10, 20, 40, 80, 120, 180, 240, 320 and 400 mg/m² were investigated. For the investigation of the total curcumin and tetrahydrocurcumin concentration in plasma, blood was collected at baseline, during the infusion at 15, 30, 90 min, and at sequential intervals at the end of infusion, and 1, 2, 4, 8, 24 and 48 hrs. Dose escalation was performed until the highest planned dose (400 mg/m²) was reached. At this dose level, an unexpected increase of the Mean Cell Volume (MCV) was detected in both subjects that were dosed. The effect of curcumin in hematological indices may be related to the binding of curcumin with red blood cell membrane as a potential drug carrier to the target tissues. Blood smears were prepared revealing transient echinocyte formation with no long lasting adverse effects observed. No clinically significant 2 changes were recorded for vital signs or physical examinations. One subject (120 mg/m) experienced a mild QT prolongation on ECG that resolved on the same day. The infusion of Lipocurc™ resulted in rapid and dose-dependent development of plasma levels of curcumin with Tmax values ranging from 0.9-1.7 hr. The values of Cmax ranged from 42 ± 22 ng/mL to 2359 ± 412 ng/ mL, corresponding to Lipocurc™ dosages between 10 mg/m2and 400 mg/m2. At the dose of 120 mg/m2, the Cmax reached 402.05 ± 211.39 ng/ml. The Tmax values of tetrahydrocurcumin (THC) plasma levels ranged between 0.5–1.8 hr. The plasma levels of curcumin during infusion were 7-16-fold higher than those of THC. The clearance of curcumin was similar across doses and ranged from 1.42–5.29 L/kg/hr. Urinary excretion of curcumin and THC was analyzed for subjects 25–50. Urinary clearance calculated from urine curcumin levels and urine volumes account for only a very small percentage (0.12%) of the total systemic clearance of curcumin in humans, consistent with studies in other species. In conclusion, the systemic safety of Lipocurc™ was good over the dose range between 10 and 320 mg/m2. In subjects dosed at 400 mg/m2, we observed an increase in MCV and erythrocyte echinocyte formation. This finding required dose de-escalation and additional safety analyses, even though stopping criteria were not reached. Minor decrease of erythrocytes and hemoglobin was observed in two subjects, but was judged as not clinically significant by the principal investigator. Markers of hemolysis (hydroxybutyrate dehydrogenase, potassium, haptoglobin, LDH) did not change significantly. Continuous infusion of Lipocurc™ achieved relevant plasma levels (ng/mL) during the infusion period. Upon termination of infusion, the plasma levels dropped very rapidly for curcumin and THC. The post-infusion plasma exposure of curcumin was 4–10% of exposure during infusion, while the post-infusion plasma exposure of THC was 26–37% of the exposure during infusion. We tentatively conclude that long time infusion (at least 2 hrs and preferably 8 hrs) seems to be an appropriate tool for achieving sustained exposure to curcumin and THC. We have no pilot data to determine the threshold infusion duration and dosage of Lipocurc™ in schizophrenia.
Lipocurc™ Infusion: Lead Drug on the Horizon for Schizophrenia?
We are encouraged by the overall safety profile of Lipocurc in control subjects and the promising findings with oral curcumin in schizophrenia to propose an exploratory dosefinding randomized placebo-controlled study of intravenous infusion of Lipocurc™ in treatment resistant schizophrenia. Sign Path Pharm PA USA has prioritized cancer treatment for Lipocurc in the research and development (R/D) platform; however, we consider it worthwhile to repurpose Lipocurc for likely therapeutic effects in schizophrenia. A recent RCT study found infusion of nitroprusside in modulating NMDA receptor, significantly improved the positive, negative, depressive and anxiety symptoms: the changes in Brief Psychiatric Rating scale: BPRS scores persisted for 4 weeks post-infusion [72]. No adverse events were reported. This study highlights the heuristic merits of exploring formulations of antipsychotics beyond the conventional oral and long-acting intra-muscular route and the relevance of novel neurotransmitter and signal transduction pathways related to neural repair and plasticity. It remains to be seen whether the roadblocks in therapeutic advances in schizophrenia can be markedly overcome through leverage of recent advances in epigenomics and genomics and in epigenetics-driven phenotype characterization in vitro and in vivo [73].
To sum up, converging evidence provides the strong rationale for adopting the two-step paradigm shift in curcumin pharmaceutical development: 1) Transforming curcumin from crude spice and botanical extract to novel drug leads ; 2) in repurposing the liposome-based formulation: Lipocurc™ from oncology to CNS disorders including schizophrenia. In this respect, our interest in intravenous infusion mode of delivery of potential drug lead in schizophrenia resembles the series of studies of intravenous infusion of ketamine in treatment resistant depression with comorbid suicidality [74,75]. Curcumin, however, distinguishes from ketamine and likely esketamine with its potential antipsychotic effects as shown in our study with oral formulation of curcumin as curcumin C-3 complex (66). Furthermore, the benefits of curcumin in reducing the depressive symptoms have been reported in 6 randomized controlled studies in patients diagnosed as depressive disorders [76].
These considerations lead us to entertain seriously that organizing and conducting Phase II of Lipocurc™ in schizophrenia open a new chapter translating recent advances in epigenetics –based, NMDA- and inflammation pathways, coupled with nanotechnology in drug delivery, towards developing and optimizing novel therapeutics in managing the devastating consequences of schizophrenia. It is somewhat disconcerting that a recent meta-analysis of TRS schizophrenia shows that the response rate to clozapine is about 40 % with 25 % reduction in the PANSS (Positive and Negative Syndrome Scale [77]. We have reason to hold that intravenous infusion of epigenetics-based drug leads adds to the growing list of therapeutic paradigms of CNS drugs while awaiting vigorous clinical trials to validate the efficacy, effectiveness and safety and tolerability in treatment resistant schizophrenia.
There is preliminary evidence supporting the construct that treatment resistant schizophrenia differs from treatment responsive schizophrenia schizophrenia demonstrating abnormal glutamate system and reduced gray matter [78]. None of the comparative studies have examined epigenetics signatures in relation to correlates in immune function, neurotransmission, and neural development. It is highly likely that in TRS, deranged epigenetics marks regulating NMDA and inflammatory pathways, providing a highly restrictive milieu for transcription and posttranscription of gene expression. Chromatin modelling in TRS may become more compact and restricted, selectively out proinflammatory phenotype and shifting the epigenetics network towards a compact transcription program. The consequences can be seen clinically in patients diagnosed with schizophrenia as highly rigid incapable of adapting socially, complicated by the impaired neurocognition and high prevalence of active positive and negative symptoms.
Conclusion and Future Directions
We conclude that accumulating evidence highly favors further refinement of an innovative epigenetics- driven drug platform conferring unprecedented degree of freshness and vitality to the challenging field of schizophrenia. Lipocurc™ may well be the promising drug lead for modifying the devastating course of schizophrenia. In the brief synopsis, we focus primarily in characterizing the multi-faceted pharmacology of curcumin and review critically the evidence for the theme of epigenetics regulation as the underlying mode of action of curcumin. We have shown the ambitious roadmap in utilizing liposome nanotechnology in formulating curcumin as a novel promising drug lead in schizophrenia and other neuropsychiatric and neurodegenerative disorders. However, we do not dismiss the importance of epigenetics-based nutraceuticals as likely adjunct treatment of schizophrenia [79,80]. We hold it is beyond the limited scope of our brief therapeutic perspectives on curcumin to review all the previous studies on nutraceuticals in schizophrenia, let alone all the other psychiatric disorders as entered in the Diagnostic Statistical Manual DSM-V for psychiatric disorders. It is perhaps fortuitous that the majority of phytochemicals: omega 3- fatty acids, vitamin D, B vitamins (B6, folate, B12), vitamin E, and carotenoids in different stages of schizophrenia exert beneficial effects in schizophrenia [81,82]. The results are it is perhaps fortuitous that the nutraceuticals exert anti-inflammatory and antioxidant effects via hitting epigenetics targets similar, albeit non-identical, to curcumin: DNA methyltransferase (DNMT), as well as Histone Acetyl-Transferase (HAT) and HDAC activities [83]. The overall therapeutic objective of epigenetics-based nutraceuticals is to activate the repressed epigenome in schizophrenia and to shift gradually the conformation of chromatin from the “closed state” to the “open, relaxed state” favoring active transcription of key genes relevant to schizophrenia. In view of the finding of repressed epigenomes in schizophrenia, nutraceuticals capable of restoring the phenotype of relaxed epigenomes will activate the transcription of genes which have been silenced and rendered inactive in schizophrenia.
Different classes of phytochemicals have been shown to selectively blocking DNA methylation via inhibiting DNMT, or restoring the balance between HDAC and HAT activities. Omega- 3-fatty acid is perhaps the best studied nutraceutical: the bioactive components: Eicosapenaenoic Acid (EPA) and Docosahexaenoic Acid (DHA), when formulated in optimal ratios, have been shown to improve depressive symptoms in mood disorders [84,85]. The positive effects of omega 3-fatty acid are likely to be mediated through the NAD+ class of HDAC: the Sirtuin signaling pathway [86]. Subsyndromal depression is quite common in schizophrenia. Evidence will soon be forthcoming for consensus opinion to formally recommend Polyunsaturated Fatty Acid (PUFA) as adjunct therapy in schizophrenia. The properties of nutraceuticals in reducing the negative symptoms and in enhancing cognition in schizophrenia are of no less importance. Baseline level of Omega- 3-fatty acid has recently been shown to correlate with cognitive impairment and functional level [87]. The family of dammarane triterpenes isolated from the traditional “tonic” medicine in Asia: Panax Ginseng, and in North America Panax quinquefolius, exert differential epigenetic modulatory effects in activating NADH+ HDAC: Sirtuin-1 [88]. Our group has demonstrated that standardized formulated Panax Ginseng: Ginsana-115, improved negative and depressive symptoms in schizophrenia [89]. An independent group found HT1001, a proprietary North American ginseng extract (Panax quinquefolius), improved cognition in the different cohort of schizophrenia.
Single nutraceutical may not suffice to alter the devastating course of treatment resistant schizophrenia. Combination of epigenetics-driven nutraceuticals with fruits and vegetables regulating epigenetics may be yet another promising approach in improving the outcome of schizophrenia. The epigenetics diet derived from phytochemicals exhibiting epigenetics modifying activities has been proposed for cancer chemoprevention, but has yet to be tested in schizophrenia (Figure 3). Recently, there has been paradoxical resurgence of interest in examining the neuroprotective effects of ketogenic diet emphasizing low carbohydrate and high protein nutrients, in schizophrenia [89].
Figure 3: Epigenetics signaling in schizophrenia: Epigenetics diet and nutraceuticals.
It is intriguing that beta- hydroxybutyrate, the main ketone body, has been shown to be the endogenous HDAC inhibitor in counteracting the deleterious effects of excesses of oxidative stress. In this respect the ketogenic diet can be considered as a variant of epigenetics diet. Further clinical trials are required to establish the efficacy and safety in schizophrenia and other neuropsychiatric disorders.
In conclusion, converging evidence suggests that curcumin shares with certain classes of nutraceuticals, herbal and dietary supplements, in behaving as epigenetics modulator displaying multiple interrelated pharmacological actions in resetting the resetting the aberrant plasticity in crucial brain regions: hippocampus, prefrontal cortex, limbic cortex in schizophrenia. While current levels of evidence from controlled studies are not vigorous or conclusive for publishing guidelines in mental health practice, the future is very bright provided that more randomized controlled studies of prolonged duration using brain imaging and peripheral biomarkers with larger sample sizes, can establish the efficacy and safety of nutraceuticals in schizophrenia. Transforming epigenetics-based health products to promising drug leads is a bold step in catalyzing CNS drug development, especially combined with nanotechnology and may confer marked therapeutic benefits in improving the positive and negative symptoms and enhance neuro-cognition in schizophrenia. Personalized medicine encompassing “oomics”: epigenomics, nutrigenomics, metabolomics and microbiota profiling, can clarify and resolve some of the discrepant findings and open the door towards personalized nutrition approach in translating epigenetics advances to enhancing outcome in schizophrenia.
Acknowledgment
We gratefully acknowledge the financial support from Michel J Fox Parkinson foundation and the Stanley Medical Research Institute MD USA for studies on curcumin and Lipocurc™.