- (2007) Volume 8, Issue 6
Meenal Shukla1, Muhammad F Hassan1, Vajinder Toor1, Jaspreet Kaur1, Cleo Solomon1, Henry Cohen1,2
1Department of Medicine, Kingsbrook Jewish Medical Center; 2Arnold and Marie Schwartz College of Pharmacy and Health Sciences, Long Island University. Brooklyn, NY, USA
Received April 26th, 2007 - Accepted August 7th, 2007
Context Symptomatic pancreatic involvement in sarcoidosis is extremely rare. There are 25 reported cases of detected pancreatic sarcoidosis before death. Case report We report the case of a male patient with a history of pulmonary sarcoidosis, who presented with clinical picture and CT scan findings suggestive of pancreatic cancer. Pancreatic mass biopsy showed lesions consistent with sarcoidosis. Patient had excellent clinical response to steroids. Conclusion Pancreatic involvement is usually asymptomatic and discovered on autopsy. However, clinicians should be cognizant that sarcoidosis can present with symptomatic pancreatic mass, albeit rarely.
Pancreatic Neoplasms; Sarcoidosis; Steroids
Sarcoidosis is a chronic multiorgan granulomatous disease of unknown etiology. Sarcoid granulomas are formed by infectious and non-infectious exogenous agents that stimulate auto-antigens to HLA Class II surface molecules on mononuclear cells stimulating T-helper inducer lymphocyte resulting in an exaggerated cellular immune response. Sarcoid lesions are noncaseating epithelioid cell granulomas with asteroid bodies [1, 2]. Sarcoidosis is a multiorgan disease that has a high predilection for the lymph nodes (99%), lungs (90%), myocardium (74%), liver (60%), and to a lesser extent the skin (25%) and eyes (25%) [1, 3, 4, 5]. With the exception of the liver, gastrointestinal tract involvement is uncommon. Pancreatic sarcoidosis is rare and often asymptomatic, it is often discovered on autopsy and occurs in 1-3% of cases of systemic sarcoidosis [5, 6, 7, 8]. Symptomatic pancreatic sarcoidosis has only occasionally been reported. A MEDLINE search from 1966 to 2007, using the key words pancreatic sarcoidosis revealed only 26 cases of symptomatic pancreatic sarcoidosis. We report a case of symptomatic pancreatic sarcoidosis.
A 54-year-old African American male was referred to gastroenterology clinic for the evaluation of severe fatigability, poor appetite and 3.5 kg weight loss over the past two months, and elevated alkaline phosphates and hepatic transaminases. He had no abdominal pain, nausea, vomiting, pruritus, diarrhea, yellow discoloration of skin and sclera, acholic stool or dark urine. He drank 1 to 2 cans of beer a month for 15 years, and denied smoking cigarettes. He had no history of blood transfusions, intravenous drug use or multiple sexual partners. He had not traveled outside of the United States in the past 10 years. His family history was noncontributory. His past medical history included pulmonary sarcoidosis, diabetes, hypertension, benign prostrate hypertrophy, seizure disorder, and hypercholesterolemia. His medications included levetiracetam, metformin, tolterodine, tamsulosin, glipizide, enalapril and aspirin. No allergies were reported.
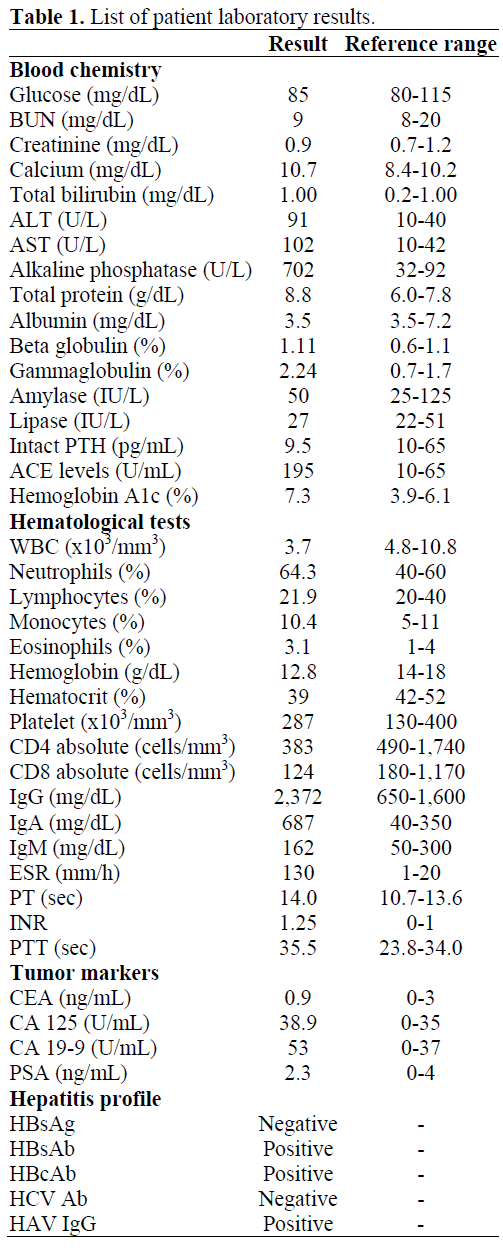
A review of systems was remarkable for a nonproductive cough and shortness of breath on exertion that waxes and wanes for the past few years. His physical exam revealed an alert and oriented male (height, 168 cm; weight 64 kg). His vitals were the following: pulse, 76/minute and regular; blood pressure, 138/76 mmHg; respiratory rate 12 breaths/minute; temperature, 36.7°C (98°F). A funduscopic examination was normal. The head and neck exam was pertinent for discrete cervical lymphadenopathy. The abdomen was soft, non-distended and non-tender with normal liver and spleen span. The neurological, cardiovascular, and chest examinations were unremarkable. He presented with leukopenia (3,700 cells/mm3; reference range: 4,800-10,800 cells/mm3), hypercalcemia (11.4 mg/dL; reference range: 8.4-10.2 U/L), elevated liver enzyme tests (AST 102 U/L and ALT 91 U/L; reference ranges: 10-42 U/L and 10-40 U/L, respectively), an elevated alkaline phosphatase (702 U/L; reference range: 32-92 U/L), and an elevated erythrocyte sedimentation rate 130 mm/h (reference range: 1-20 mm/h). His coagulation profile was slightly elevated. His renal function tests, serum glucose, and pancreatic enzymes were normal (Table 1).
Tests for pancreatic exocrine insufficiency were not performed since the patient did not exhibit signs and symptoms of malabsorption. A chest roentgenograph was normal. However, a computed tomography (CT) scan of the chest demonstrated predominant peribronchial and perivascular markings involving the upper and middle lobe of the right lung with calcified subcarinal and hilar lymph nodes suggestive of sarcoidosis. A CT scan guided biopsy of enlarged cervical lymph node was consistent with a sarcoid lesion. An abdominal CT scan demonstrated a pancreatic head mass with retroperitoneal lymph node enlargement (Figure 1). Magnetic resonance cholangiopancreatography (MRCP) and endoscopic retrograde cholangio- pancreatography (ERCP) were not performed since the biliary and pancreatic ducts were not dilated and the serum bilirubin level was normal. A CT scan guided pancreatic mass biopsy demonstrated noncaseating granulomas with Langerhan giant cells and fibrosis (Figure 2). Both an acid fast bacilli stain and gram stain for a fungal infection were negative.
Oral prednisone 40 mg daily for 7 days followed by a 7 week tapering schedule was initiated. The patient’s signs and symptoms improved: his fatigability resolved and appetite improved; liver enzyme tests and serum calcium levels normalized. The patient did develop prednisone-induced hyperglycemia that was managed by titrating upwards his glipizide regimen. A repeat abdominal CT scan 4 weeks after initiation of prednisone treatment revealed no change in the size of the head of the pancreas and peripancreatic lymph nodes. After completing his 8 week course of prednisone treatment, clinically he remains stable without any abnormal laboratory parameters.
Asymptomatic Pancreatic Sarcoidosis Prevalence
In 1937, Nickerson published the first autopsy case of pancreatic involvement with noncaseating granulomas in a patient with systemic sarcoidosis [9]. Mayock et al., reported three cases of pancreatic involvement (3%) from 287 autopsies of patients known to have sarcoidosis [7]. A large autopsy series in Japan accumulated over 28 years of patients who died from sarcoidosis reported a higher incidence of pancreatic involvement at 2.1% [10]. In this Japanese autopsy series of case reports, half of those found to have pancreatic sarcoidosis were asymptomatic and died of other causes. In 2006, Caceres et al. reported 25 patients with surgically proven pancreatic sarcoidosis of which 12 cases presented with a pancreatic mass localized to the head of pancreas, and 13 cases presented with a diffusely firm nodular pancreas [4].
Symptomatic Pancreatic Sarcoidosis Prevalence
In 1950, Curran and Curran published the first surgically diagnosed case of pancreatic sarcoidosis via an exploratory laparotomy of a patient with persistent diffuse abdominal pain [11]. Noguchi et al. in 1993 reported 14 patients with pancreatic sarcoidosis [12]. Biopsy of the pancreas established the diagnosis in 7 cases; 4 were diagnosed by biopsy of the peripancreatic lymph nodes and 3 by clinical findings only. At laparotomy 6 patients showed swollen pancreas with diffuse nodular changes, 4 had enlargement of the head and only one patient had a combination of pancreatic head mass and diffuse enlargement. Peripancreatic lymphadenopathy was observed in majority of these patients. In 2006, Cacreas et al. reported 4 cases of symptomatic pancreatic sarcoidosis out of 25 patients with surgically proven pancreatic sarcoidosis [4].
Symptomatic pancreatic sarcoidosis most commonly presents with abdominal pain, weight loss, obstructive jaundice and emesis [4, 13]. Three cases of pancreatic sarcoidosis presented as acute pancreatitis [14, 15]. Pancreatic symptoms are due to organ infiltration and compression by enlarged lymph nodes [2]. Sarcoidosis presenting as a mass in the pancreas is less in common but does occur [13, 16, 17, 18, 19].
Our patient with a known history of pulmonary sarcoidosis presented with constitutional symptoms and a pancreatic mass on CT scan of abdomen. Our patient’s clinical condition and liver enzyme tests improved with corticosteroid treatment (prednisone). A follow up CT scan of the abdomen 8 weeks after presentation and during prednisone treatment showed no change in the size of pancreatic head. In this setting, pancreatic sarcoidosis is likely and a laparotomy with standard pancreaticoduodenectomy may not be necessary. However, since our patient only had pancreatic head involvement, a biopsy of the pancreatic head is necessary to rule out a malignancy and to establish the diagnosis of pancreatic sarcoidosis. If the pancreatic head mass caused significant obstruction of the common bile duct, ERCP with brushing of the pancreatic and biliary duct with possible pancreatic and biliary stent placement could be considered in addition to medical treatment. Our patient is being followed clinically with monitoring of liver enzyme tests every 6 months; cancer antigen 19-9 (CA 19-9), serum angiotensin converting enzyme (ACE) levels, and CT scan of abdomen to detect any change in size of pancreatic head, involvement of body and tail of pancreas and other abdominal visceras by sarcoidosis or malignancies are being evaluated annually.
Pharmacotherapy
There are no standard guidelines for treating pancreatic sarcoidosis and the prognosis of the disease is variable. Corticosteroids is the pharmacotherapy of choice for the management of sarcoidosis [3, 20, 21, 22]. Corticosteroids are indicated for ocular, cardiac and central nervous system involvement, stage 2 pulmonary disease, and patients presenting with malignant hypercalcemia or constitutional symptoms. Corticosteroids therapy has been effective in many cases, however, occasionally achieving only spontaneous remission [15, 16]. Chronic corticosteroid use can be associated with hypertension, cushingoid effects, psychosis, osteoporosis, glaucoma, cataracts, hypokalemia, glucose intolerance, telangiectasis, acne, and gastropathy. Due to the deleterious effects of chronic corticosteroid pharmacotherapy, its use should be employed only after careful considering its risks and benefits. After discontinuing corticosteroids, the recurrence rate of pancreatic sarcoidosis in severe symptomatic cases is 100% [22].
Sarcoidosis is a chronic multiorgan disease that uncommonly involves the pancreas. Pancreatic involvement is usually asymptomatic and discovered on autopsy. However, clinicians should be cognizant that sarcoidosis can present with symptomatic pancreatic mass, albeit rarely.