Original Article - (2021) Volume 22, Issue 2
1Department of Surgery, Rudy Ruggles Biomedical Research Institute, Nuvance Health, USA
2Department of Surgery, University of Southern California, Keck School of Medicine, CA, USA
3Department of Medical Oncology, Northwell Health Cancer Institute, Donald and Barbara Zucker School of Medicine at Hofstra and Feinstein Institute for Medical Research, USA
4Department of Surgery, New York Medical College, Westchester Medical Center Health Network, Valhalla
Received Date: February 23rd, 2021; Accepted Date: March 05th, 2021
Background The effects of adjuvant radiation therapy on pancreatic cancer outcomes after resection are not well defined in the literature. Methods We abstracted data from the Surveillance, Epidemiology, and End Result (SEER) database to explore the impact of adjuvant radiation on cancer-specific survival in pancreatic cancer patients who received surgical resection. Results A total of 10,224 patients met our inclusion criteria with 6768 (66.2%) patients treated with surgery only and 3456 (33.8%) treated with surgery plus adjuvant radiation. Surgery followed by adjuvant radiation was associated with significantly improved survival (HR: 0.753, CI: 0.718–0.789, p<0.001). Additionally, female gender and married status were both independently associated with better survival (p<0.05), while advanced age, Caucasian race, higher TNM stage, and higher grade had worse survival outcomes (p<0.05) Asian and Spanish-Hispanic-Latino patients were less likely to receive adjuvant radiotherapy (p<0.05). Conclusion Adjuvant radiation was associated with significantly improved survival after resection for pancreatic cancer. There are significant differences in the patient populations who receive adjuvant radiation.
Pancreatic neoplasms; Pancreas cancer; Radiosurgery; Stereotactic; Stereotactic body radiation therapy (SBRT); Radiation; Radiotherapy
In the year 2020, about 57,600 people will be diagnosed with pancreatic cancer and approximately 47,050 people will die of this devastating disease. Over the last several decades, the survival rates among patients with pancreatic cancer remain dismal [1]. The overall 5-year relative survival rate for patients reported to the Surveillance, Epidemiology, and End Results (SEER) database of the National Cancer Institute from 1996 to 2004 was 5.1% [2]. Treatment strategies for early respectable and metastatic disease are relatively clear and definite, namely resection plus sequential adjuvant chemotherapy for the former and systemic chemotherapy or supportive care for the latter [3]. The role of radiation therapy in pancreatic cancer continues to be investigated. Randomized trials have yielded conflicting data in locally advanced disease. Also, as radiation techniques have improved over time, findings in older trials may not be applicable in modern practice [4]. Treatment for pancreatic cancer has been the subject of debate [5]. Despite, and often because of, the results of multiple contradictory clinical trials that have examined various management strategies, the optimal manner in which pancreatic cancer should be treated ultimately rests in the judgment and preferences of the individual oncologist [6]. At the center of much of this controversy is radiation therapy. Therefore, to further investigate the relationship between adjuvant radiation therapy and pancreatic cancer patient prognosis, we used data from the Surveillance, Epidemiology, and End Result (SEER) database to explore the impact of adjuvant radiation on cancer-specific survival in pancreatic cancer patients who received surgical resection.
Study Population
A retrospective cohort was identified using the Surveillance, Epidemiology and End Results (SEER) database, a project sponsored by the National Cancer Institute [7,8] The SEER database was established in 1973 and is a set of extensive population-based cancer registries that cover approximately 28% of the US population (Alaska Native Tumor Registry, Connecticut, Georgia Center for Cancer Statistics, San Francisco-Oakland, San Jose-Monterey, Greater California, Hawaii, Idaho, Iowa, Kentucky, Los Angeles, Louisiana, Massachusetts, New Mexico, New York, Seattle- Puget Sound, Utah). We queried the November 2018 submission from 1973 to 2015. Using the International Classification of Disease, Third Edition (ICD-O-3) codes, we queried for Primary site "Pancreas". Inclusion criteria were set as adenocarcinomas of the exocrine pancreas. Since data in the SEER database is deidentified, our study is exempt from Institutional Review Board review.
Selection of variables
Variables of interest included the year of diagnosis, patient demographics (age at diagnosis, gender, race, and marital status), tumor characteristics (stage, grade, and tumor size), surgery technique and details, radiotherapy or chemotherapy administered, as well as survival in months and patient status at the end of follow up period. The race was recorded as white, black, Spanish-Hispanic- Latino, Asian, and others.
Survival was defined as months from the time of diagnosis to the point of last contact or the date of death in cases where the patient was deceased. Patient status at the end of the follow-up period was recorded as alive or deceased.
Statistical analysis
Data extracted from the SEER database was imported on to the statistical package for the social sciences (SPSS) version 23 IBM Corp, USA for analysis [8]. Both descriptive and inferential analysis was conducted. Characteristics of our sample population were described using frequencies, percentages, means, and medians. Survival was estimated using the Kaplan–Meier curve. Survival differences between variables were compared using the log-rank test. We subsequently computed propensity scores to assess the probability of being assigned to a treatment group (surgery only or surgery plus adjuvant radiation) based on the observed patient's demographic, and pretreatment clinical characteristics. The logistic regression model was used to calculate the propensity scores derived from these confounding preoperative parameters. These variables included age, sex, race, year of diagnosis, marital status, and preoperative tumor characteristics (size, grade, tumor location). The most appropriately matched pairs were selected based on the propensity scores using a 1:1 nearest neighbor algorithm (R-plugin 2.14.2 for SPSS vs. 21.0). A p-value of less than 0.05 was considered significant. Confidence intervals (CI) were set at 95%. Multivariate Cox-regression was used to study the relationship between various patient and disease-specific factors and survival.
A total of 10,224 patients met our inclusion criteria with 6768 (66.2%) patients treated with surgery only and 3456 (33.8%) treated with surgery plus adjuvant radiation. Table 1 compares the baseline characteristics of the two groups. Significant differences between patient demographics and tumor characteristics were demonstrated.
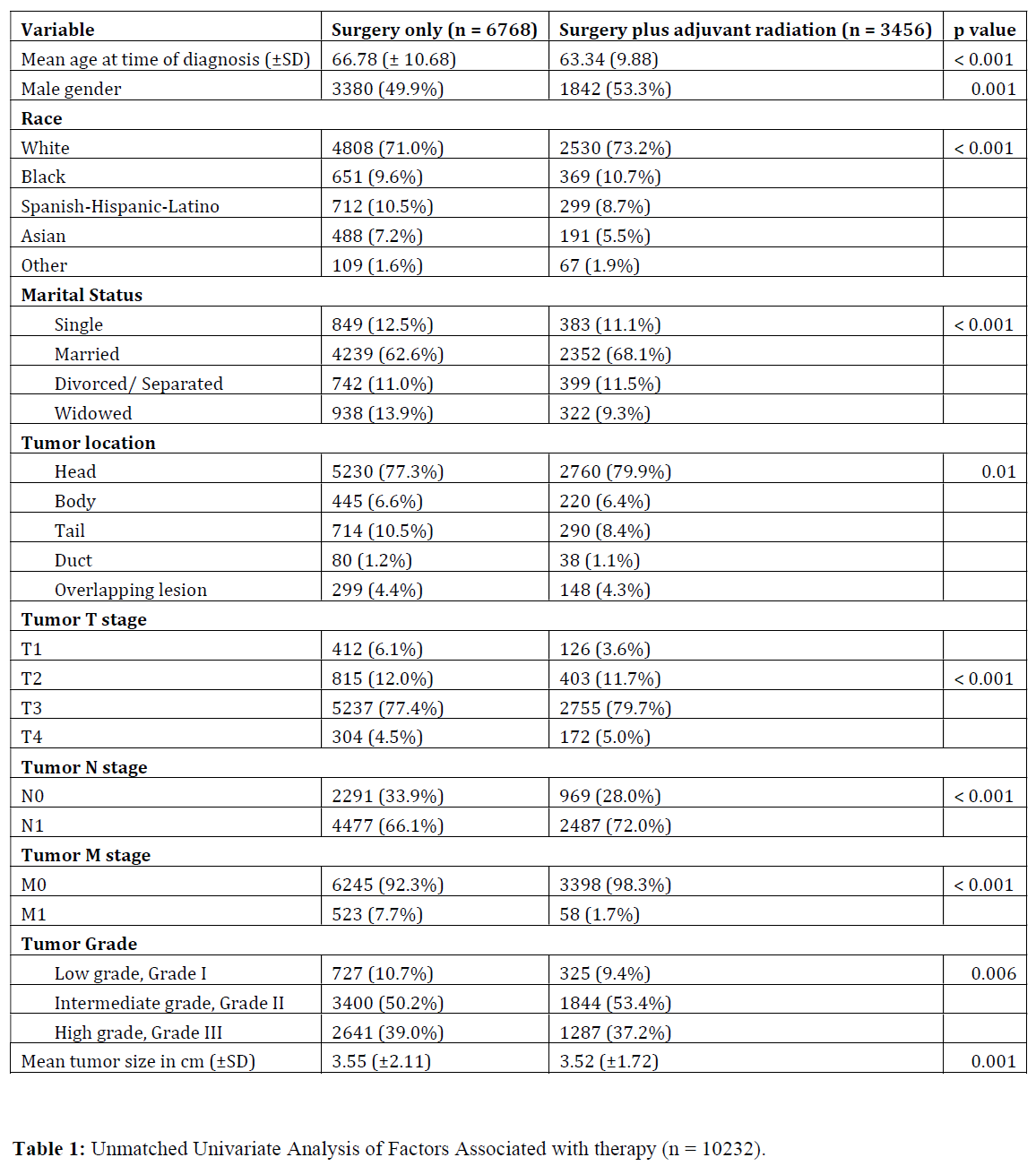
Unmatched survival analysis
Patients who underwent surgery plus adjuvant radiation had a significantly higher overall survival (OS) than patients who underwent surgery only (p <0.001). The median OS values were 21 and 16 months for surgery plus adjuvant radiation and surgery only respectively.
Propensity Score Matching and Multi-variate Survival Analysis
Propensity matching yielded 5766 patients (2883 matched pairs) from surgery plus adjuvant radiation and surgery only groups. There were no significant differences in baseline characteristics between the two groups after matching (Table 2).
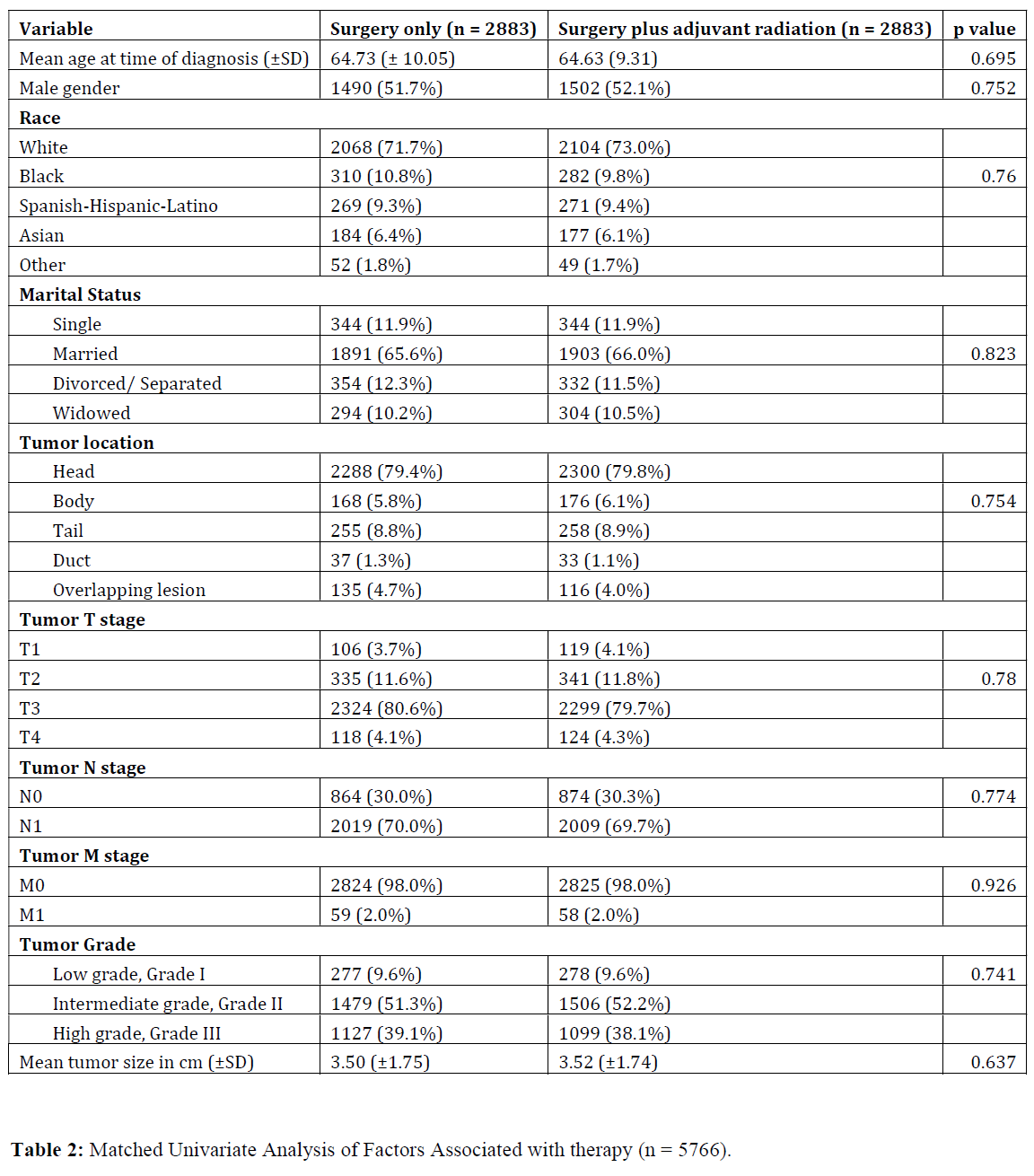
Patients following surgery plus adjuvant radiation had significantly higher OS when compared to patients undergoing surgery only (p < 0.001). The median OS values were 21 and 16 months for surgery plus adjuvant radiation and surgery only respectively.
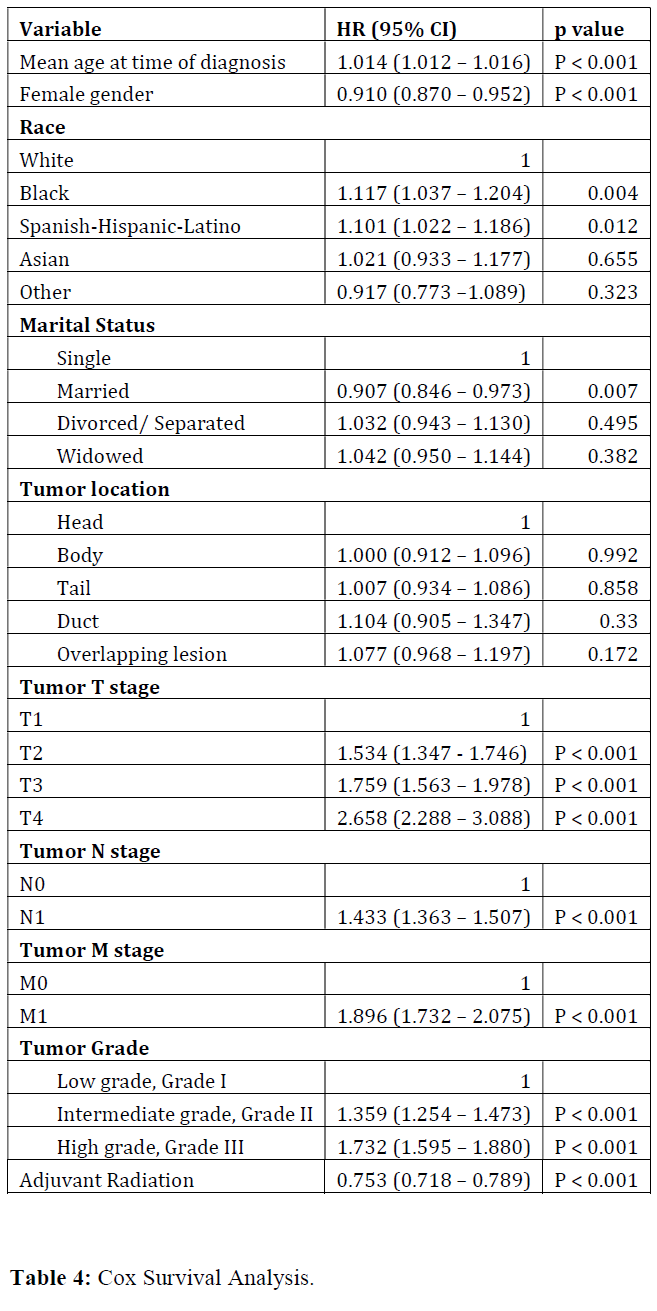
We further investigated the correlation between OS and other covariates in a Cox Regression Analysis. Multivariate analysis demonstrated that surgery plus adjuvant radiation was associated with significantly improved survival (HR: 0.753, CI: 0.718 – 0.789, p < 0.001). Additionally, female gender and married status were associated with better survival (p < 0.05) while advanced age, Caucasian race, higher TNM stage, and higher grade had worse survival outcomes (p < 0.05) (Table 3).

Predictors of adjuvant radiation
In a logistic regression model evaluating factors associated with receipt of adjuvant radiation (Table 4), older age, prior or current marital status, and higher T and N stages were associated with receipt of adjuvant radiation. Spanish-Hispanic-Latino and Asian races and higher M stages were associated with adjuvant radiation not being administered.
Our study showed a survival benefit in patients who received adjuvant radiation therapy for pancreatic cancer. This study adds to the growing literature of retrospective studies on the role of radiation therapy following pancreatic resection and highlights the need for a large multicenter randomized trial.
Radiation therapy represents a highly complex and technical treatment that is rapidly evolving. The modality has undergone and continues to undergo, dramatic transformations with advances in computational modeling and medical imaging [9,10]. These advances will only continue to accelerate in the coming years and will likely follow a "double exponential" growth pattern leveraging advances in both hardware and software capabilities. While radiation therapy has historically been described as a singular entity in oncologic management it represents a broad treatment class, inclusive of a wide range of treatment procedures and methods. Despite the spectrum of potential treatments under the category of radiation therapy, the allencompassing nomenclature may obscure the heterogeneity that exists within the term radiation therapy.11
The GITSG trial randomized patients with pancreatic adenocarcinoma resected to negative margins to 40 Gy delivered with 2 Gy daily fractions with a split course (2-week break after the first 20 Gy were delivered) with concurrent 5-FU chemotherapy versus observation alone [11]. Five-year OS was 15% vs. 5% and median diseasefree survival (DFS) was 11 vs. 9 months, both favoring the adjuvant chemoradiotherapy arm. The trial was closed early due to poor accrual. However, this trial helped establish adjuvant chemoradiotherapy (CRT) as the standard of care in the United States.
Our results are in concordance with prior studies of adjuvant radiation therapy for pancreatic cancer. Cox analysis conducted by Blain et al. found that receiving EBRT was an independent prognostic factor for an improved hazard ratio of dying from pancreatic cancer (HR=0.65, p<0.0001) [12]. Sugawara and Kunieda analyzed 2,532 patients from the SEER database treated with surgery plus/minus adjuvant radiotherapy from 2004 to 2009 [13]. They also found a survival advantage in the radiotherapy group. Overall survival (OS) was 20 months vs. 16 months in the adjuvant radiotherapy and observation groups, respectively (p<0.0001). Disease-specific survival was 22 months vs. 18 months in the adjuvant radiotherapy and observation arms, respectively. Opfermann et al. analyzed 3,314 patients from the SEER database treated with surgery plus/minus adjuvant radiotherapy from 1998 to 2006 [14]. They also found a survival advantage in the radiotherapy group. OS was 19 months vs. 14 months favoring the radiotherapy arm (p<0.001). The expanded timeframe of our study is promising for the continued use of this adjuvant therapy. Moreover, we found that the type of surgery performed, and the location of the tumor does not influence the benefit of adjuvant radiation. Alternatively, the relatively short lifespan of these patients could mean that survival differences are potentially attributable to the type of surgery performed and the location of the tumor, both of which do not appear within the available follow-up period.
Concerning trends regarding potential disparities in the utilization of radiation therapy emerged from our analysis. Namely, those white patients were more likely to receive adjuvant radiation than Asian and Spanish-Hispanic- Latino patients. Shavers et al similarly found that Hispanic patients were less likely to receive radiation therapy for pancreatic cancer than non-Hispanic whites (OR 0.50, 95% CI, 0.27–0.95) [15] Gad et al found an increasing trend of pancreatic adenocarcinoma incidence-based mortality among Asian-American patients, with an annual increase of 4.535% (95%CI 3.538-5.541; P<0.001) from 1992 to 2015 [16]. Their SEER analysis did not include trends in radiation therapy. In concordance with our data, Eloubeidi et al. identified no difference in radiation therapy by race when comparing black and white patients (15.0% vs. 14.2% for white's vs. blacks, P=.65) on analysis of the Alabama Statewide Cancer Registry from 1996 to 2000, however, they did identify that black patients were less likely to receive both chemotherapy (26.78% vs. 32.3%, P=.02) and surgery (14.0% vs. 17.0% P=.09), which has also been found on SEER analysis [17].
Healthcare inequities in the incidence and mortality of pancreatic cancer are multifaceted. Given the survival advantage identified among patients who receive surgery and adjuvant radiotherapy, ensuring access to such treatment modalities is an important step in improving pancreatic cancer outcomes. Noel and Fiscella identified that black and Hispanic patients with pancreatic cancer had lower rates of surgical resection and were more likely to be treated at low-volume hospitals, proposing that differences in pancreatic cancer outcomes are driven by race and socioeconomic status [18]. Continued analysis of differential utilization of radiation therapy, both on a population and individual patient level, is needed to determine steps towards more equitable treatment.
It is important to note some important disadvantages of using the SEER database. This database does not report chemotherapy data and does not report the dose or duration of radiotherapy. Also, margin status was not available in the SEER database for this group of patients [19-22]. However, previous meta-analyses conducted have suggested that the benefit of adjuvant radiotherapy may be limited to patients with positive margins, so perhaps these patients were more likely to have gotten this adjuvant treatment in the SEER database and thus would be over-represented in the irradiation arm. Therefore, incorporating more information on chemotherapy, radiotherapy, and margin status would be useful in investigating the effects of these factors on survival in pancreatic cancer patients. Data from randomized clinical trials may also aid in the investigation of the impact of various treatment approaches.