- (2016) Volume 17, Issue 1
Marcos Vinicius Perini1,2, Fabiana Roberto Lima3, Nathan Elie Frenk3, Hilton Muniz Leão Filho3, Fabricio Ferreira Coelho1, Jaime Arthur Kruger1, Manoel de Souza Rocha3, Paulo Herman1
Department of 1Gastroenterology, Surgical Division, University of Sao Paulo Medical School, Sao Paulo, Brazil
Department of 2Surgery, University of Melbourne, Australia
Departments of 3Anatomical Pathology and 4Radiology, University of Sao Paulo Medical School, Sao Paulo, Brazil
Received June 18th, 2015-Accepted August 31st, 2015
Serous cystic neoplasms of the pancreas are usually benign. Malignant serous cystic neoplasm is a rare clinical entity. We report a case of a 45-year-old man presenting with metastatic liver lesions on ultrasound. Computed tomography scan showed a hypervascular solid lesion arising in the pancreatic body and invading the celiac axis and portal vein with liver metastases. Ultrasound-guided liver biopsy diagnosed a serous cystic neoplasm, indistinguishable from serous cystadenoma. Based on clinical, radiological and pathological correlation, the diagnosis of cystadenocarcinoma was established. There are few reported cases of malignant serous cystic neoplasms in which malignancy was confirmed. Although rare in males, cystadenocarcinoma should be considered in the differential diagnosis of patients presenting with a pancreatic mass and liver metastasis.
Cystadenocarcinoma, Serous; Cystic Fibrosis; Liver; Neoplasm Metastasis; Pancreas
With the advances in imaging technology, incidental pancreatic cystic lesions can be found in up to 13,5% of adult patients [1]. The majority of neoplastic cysts are classified as serous and mucinous [2]. Serous cystic adenomas are usually benign whereas mucinous cystadenomas have the potential of malignant transformation [3-5]. In 1989, George et al. described serous microcystic adenocarcinoma as a new entity that was pathologically indistinguishable from serous cystadenoma but that displayed malignant behavior [6].
After this preliminary report, others investigators have reported patients with malignant serous cystadenocarcinomas of the pancreas and in a recent retrospective multinational study including 2622 patients, only three serous cystadenocarcinomas (0.1%) were recorded [7].
Until now thirty one cases of this very rare entity have been described (Table 1). We report here the clinical, imaging and pathological findings of a patient with a serous cystic neoplasm of the pancreas with malignant behavior, presenting with a pancreatic mass and liver metastases.
A forty-five year-old male was referred to our hospital with a 3-month history of weakness and weight loss. Physical examination revealed tenderness on liver palpation and no signs of jaundice.
Laboratory evaluation revealed normal bilirubin levels (1.0 mg/dL – normal <1.5 mg/dL), elevated gamma-GT (261 U/L) and alkaline phosphatase (124 U/l), and normal levels of tumor markers as follows: CA19-9 of 8.1 ku/L (<37), CEA of 0.8 ug/L (<5.0), and AFP of 1.3 ku/L (<10), chromogranin A of 4.1 u/L (0-17.2).
Contrast-enhanced computed tomography (CECT, Figure 1) identified a solid, heterogeneous and predominantly hypervascular mass involving and replacing most of the pancreatic body and tail, measuring 9.5 cm, associated with atrophy and ductal dilatation of the remaining pancreatic tail. The lesion showed strong enhancement in the arterial phase and washout in the portal and delayed phases. Thrombosis of the splenic and superior mesenteric veins and tapering of the celiac trunk branches were also observed. Several other hypervascular masses with the same enhancement pattern were seen throughout the liver, measuring up to 9.3 cm, as well as peripancreatic lymphadenopathy and minimal ascites.
Figure 1. (a.). Arterial phase CECT image shows an infiltrative, heterogeneous, predominantly hypervascular mass involving the pancreatic body (white arrows) and several hypervascular lesions in the liver. (b.). Portal phase image shows ductal dilation and parenchymal atrophy in the pancreatic tail (black arrow), as well as peripancreatic lymphadenopathy (black arrowhead) and collateral circulation (black arrow) secondary to splenic vein thrombosis. (c.). Portal phase image shows splenic vein (white arrow) thrombosis secondary to tumor infiltration by the pancreatic mass (black arrowhead); some of the liver lesions are shown to have necrotic center (black arrow). (d.). Delayed phase shows contrast washout of the liver lesions (black arrows).
Imaging findings were strongly suggestive of a pancreatic primary neoplasm with multiple liver hypervascular metastases, most likely a neuroendocrine tumor. Since the findings were nevertheless inconclusive, an ultrasound-guided percutaneous liver lesion biopsy was performed.
Histology revealed a metastatic tumor composed of sparse fibrous septa and numerous small cysts lined by cuboidal cells with clear cytoplasm and round compact nuclei. The adjacent non-neoplastic hepatic tissue exhibited sinusoidal dilatation due to compression. On immunohistochemical analysis, the neoplastic cells showed diffuse positive staining for 35BH11 (keratin 8) and EMA (epithelial membrane antigen), whereas only rare cells demonstrated immunoreactivity for CEA (carcinoembryonic antigen). Other immunohistochemical markers such as keratins 7 and 20, CD10, TTF-1, chromogranin A, PSA and CDX-2 were negative (Figure 2). Such findings were suggestive of metastatic pancreatic serous cystadenocarcinoma.
The patient was referred to oncology and no chemotherapy regimen was offered after case discussion. CECT performed five months later showed disease progression with increased dimensions of the pancreatic mass and of the liver metastases, as well as the presence of ascites. The patient was referred to palliative care and died 18 months after diagnosis.
According to the World Health Organization Classification of Tumors, serous cystic neoplasms represent cystic epithelial lesions composed of glycogenrich cells that produce a watery fluid similar to serum. While the vast majority is benign (serous cystadenoma), in rare cases these tumors may be malignant (serous cystadenocarcinoma). Serous cystic neoplasms include serous microcystic adenoma, serous oligocystic and illdemarcated adenoma, von Hippel–Lindau-associated cystic neoplasm, the solid variant of serous cystic adenoma, and serous cystadenocarcinoma. The term serous cystadenoma refers to the serous microcystic type (the most frequent) of these neoplasms [2]. Although the vast majority of these lesions are benign, some of them can invade surrounding tissues and metastasize. In this rare situation it is named cystadenocarcinoma. Until now, 31 cases of serous cystadenocarcinoma have been reported in the English literature as shown in Table 1.
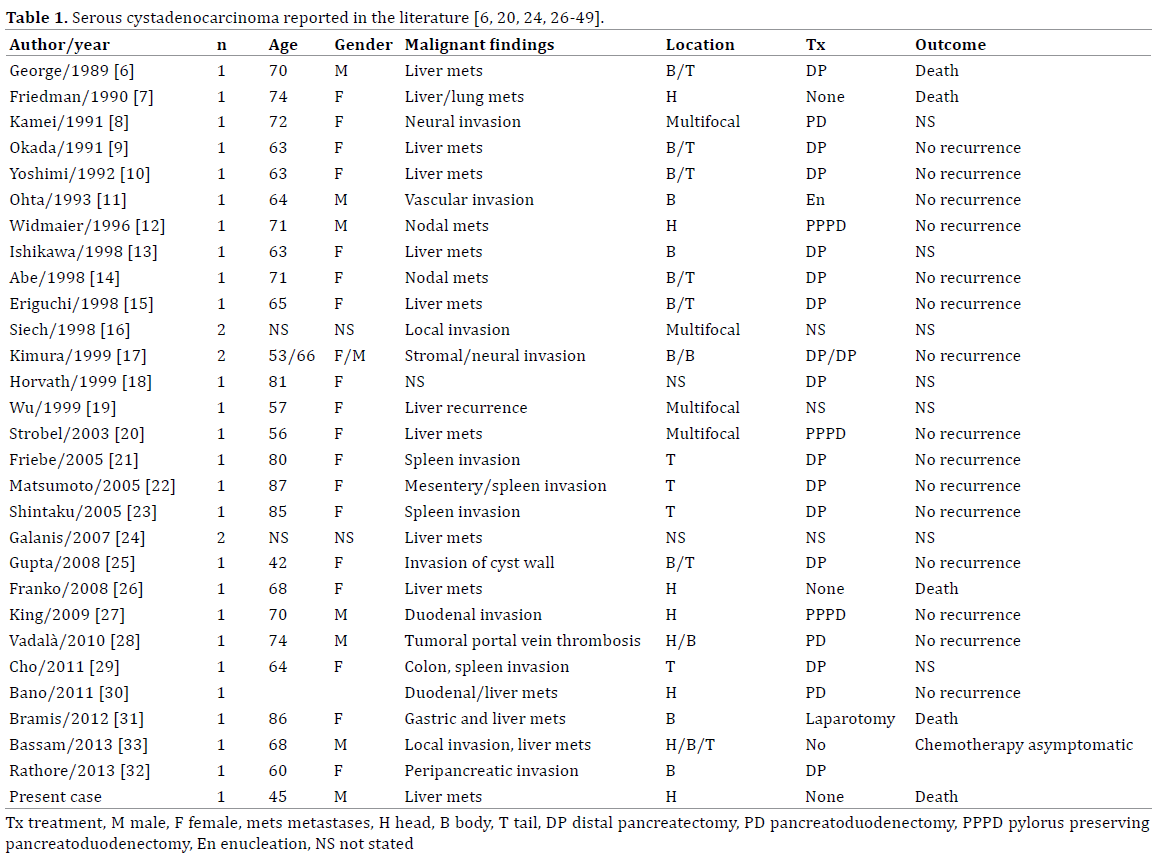
Although some authors refer no gender predilection others report a higher incidence in women. Serous cystadenocarcinoma are mainly located in the pancreatic head, as does its benign counterpart [8, 9].
CECT and magnetic resonance usually are capable to distinguish serous cystic neoplasms from pancreatic pseudocysts and from mucinous lesions if they show a typical microcystic appearance [9, 10]. Although imaging is useful for the differential diagnosis between serous and mucinous tumors, the assessment of tumor malignancy grade is not possible. On the other hand, hypervascular pancreatic lesions composed of a solid stroma and some small cystic lesions without signs of metastases or local invasion may be diagnosed as a benign lesion with an accuracy of 95% [11]. Moreover, imaging of cystic neuroendocrine tumors typically demonstrates a septated cyst with at least a rim of arterially enhancing tissue [12]. These tumors are generally well-circumscribed, and they may demonstrate variable levels of uptake on an indium 111 (111In) pentetreotide scan[13].
There are no radiologic criteria that allow the diagnosis of a malignant cystic lesion, unless tumor infiltration or metastases are present [2]. One study addressing pancreatic cystic lesion using 18-FDG PET found that a positive result strongly suggests malignancy and, therefore, radical resection should be proposed; a negative result indicates a benign tumor that may be treated with limited resection or, in selected high-risk patients, followup [14].
Endoscopic ultrasound-guided cyst fine needle aspiration may help determine the nature of pancreatic cystic lesions [15]. Some reports have shown that cystic fluid CEA levels have a sensitivity and specificity of 87.5% and 44%, respectively, and that high levels of this marker (>250 ng/mL) reliably identify a mucinous neoplasm, whereas values lower than 5 ng/mL can exclude it. High CA 19-9 levels in cyst fluid have been shown to be indicative of mucinous tumors [16]
Moreover, positive mucin staining identifies mucinsecreting lesions. Expression of mucin-like carcinoma associated antigen in the fluid can also be used to differentiate cystadenomas from cystadenocarcinomas and pseudocysts. Mucin staining in conjunction with CEA levels in the cyst fluid appears to be the best approach for the differential diagnosis of pancreatic cystic lesions [17-19].
Immunohistochemical study of serous cystadenoma shows expression of cytokeratins 7, 8, 18 and 19, with the predominance of cytokeratin 7 and 19 [20], and lack of p53 [21] and K-ras mutations [22]. Staining for CEA, trypsin, desmin, actin and factor XIII is also negative [22]. In about 40% of the cases, cells are positive for neuron-specific enolase, but immunostaining for neuroendocrine or islet cell differentiation, such as chromogranin, synaptophysin, insulin, glucagon, somatostatin andVIP is negative [23].
The potential for malignant degeneration of the serous cystic pancreatic lesion is low. Contrary to pancreatic adenocarcinoma that is considered highly aggressive, cystic neoplasms are slow growing and can reach a large size before diagnosis, yet they still have a favorable prognosis. However, there have been an increasing number of case reports describing serous cystadenomas that clinically behave in a malignant fashion. A serous cystadenoma of the pancreas is diagnosed as cystadenocarcinoma when invasion of the surrounding tissue or metastatic spread are present [6]. Since the first description of this malignant behavior, some authors raised concerns regarding “watch and wait” policies for cystic pancreatic lesions. While Hovarth et al. proposed resection for every cystic pancreatic lesion [24], others advocate that the chance of malignancy is lower than the morbidity rate after a pancreatic surgery [25]. Adopting this point of view, those who propose resection agree that close surveillance should be employed and in cases of doubt, resection should be indicated [8, 9, 25].
Although surgical treatment of pancreatic serous cystic lesions is driven by tumor location, growth rate, local complications (such as biliary obstruction), suspicion of malignancy and clinical conditions, the treatment of patients with cystadenocarcinoma is resection whenever possible. On the other hand, some surgeons argue that due to its slow rate of growing, suspicious lesions could be followed and resection offered in selected cases in which lesions increase more than 0.4 cm per year [26].
When dealing with pancreatic cystadenocarcinomas with limited and resectable liver metastases, surgical treatment could be an option in well selected cases. In these cases, where the complete tumor removal is possible, long term survival can be achieved. However in those patients with disseminated disease, as the case described, survival is dismal and depending mostly in empiric treatment and loco-regional complications.
Pancreatic cystadenocarcinoma is a rare entity and few cases have been described in the literature. Aggressive surgical treatment is still considered the main treatment option for this rare disease. More importantly, its existence accounts for controversy regarding management of its benign counterpart, the much more common and often incidentally discovered serous cystadenoma of the pancreas.