Case Report - (2023) Volume 23, Issue 3
Received: 29-Oct-2021 Editor assigned: 03-Nov-2021 Reviewed: 17-Nov-2021 Revised: 27-May-2022 Published: 18-Jun-2022
Pancreatic infiltration with lymphoma/leukemic cells is a rare manifestation of ALL or NHL, especially for adults. Leukemia/lymphoma of the pancreas should be considered if there are multiple or diffusing pancreatic nodules on imaging findings and MRI possesses higher sensitivity than CT scan in identifying tumor lesions. Patients with ALL/NHL pancreas infiltration usually displayed unspecific symptoms. EUS-FNA is helpful to make the pathological diagnosis. FISH can better help in diagnosing and categorizing lymphoma.
Diffuse large B-cell lymphoma, Breast lymphom, Acute lymphoblastic leukemia, Endoscopic ultrasound guided, Fine-needle aspiration, non-Hodgkin lymphoma, Primary pancreatic lymphoma
Extraneous non-Hodgkin lymphomas represent up to 30% of all cases of non-Hodgkin lymphoma, and the gastrointestinal tract is the most commonly involved extramural site, accounting for 50% cases. PPL is a rare clinical entity most likely to be misdirected as pancreatic carcinoma in presentation. There is a paucity of cases described in the literature, with 150 cases reported in the English language medical literature. The most common histological type of PPL is non-Hodgkin’s lymphoma. Less than 2% of all extramural malignant lymphomas and 0.5% of all pancreatic masses are PPLs [1].
Diagnostic criteria for primary pancreatic lymphoma include: No lymph node enlargement of superficial or mediastinal lymph nodes, a normal leukocyte count in the peripheral blood, main mass in the pancreas with lymph nodal involvement confined to the peripancreatic region, and no hepatic or splenic involvement. Breast Lymphoma (BL) is a rare Non-Hodgkin Lymphomas (NHL), accounting for only 0.04%-0.7% of breast tumors and about 2% of extrinsic lymphoma.
A 33-year-old female patient, first presented with lumbago lasting for half a year, and occasionally pain in her upper abdomen, radiating toward her vest. The symptoms went unnoticed until she developed pain in her lower abdomen and groin. She was first checked at a local hospital on June 2018. Initial laboratory tests after admission showed anemia HGB: 81 g/L, WBC: 3.45 × 109/L and slightly elevated Ca125 (36.7 IU/ml, reference range 35 IU/ml. The percentage of neutrophile and lymphocyte, blood platelet, prothrombin time, hepatic and renal function tests, and other tumor markers was all in normal range. Chest imaging showed normal results. Abdominal ultrasound revealed an enlarged pancreas with uneven echo and splenomegaly. Abdominal CT scan (Figure 1) suspected pancreatitis, due to swelling of the pancreas, perspicacious complicated seepage and dispersion in the effusion peritonitis; the main pancreatic duct expansion, on the right side of the attachment multiple lymph node enlargement and fusion; splenomegaly; So his local doctor gave him a blood amylase test, but the results were normal. Subsequently, abdominal enhanced CT was completed, indicating that the pancreas was enlarged, the lymph nodes around the mesentery, retroperitoneum and bilateral iliac vessels were enlarged, and the pelvis suffered multiple bone destruction. Therefore, her doctor suspected that she has pancreatic cancer and transferred to the gastroenterology department of our hospital for further diagnosis. In addition to her history of hepatitis B virus and cesarean section, she was allergic to cephalosporin (the specific drug was unknown). Her other past medical history was unremarkable, and she denied smoking and alcohol intake. She has no special findings except for her appearance of anemia. However, the patient started to have fever with the temperature fluctuating from 37.5°C to 38.5°C. We were once again improving blood test. This time we found something different (Table 1) [2].
Figure 1: Enhanced CT was completed, indicating that the pancreas was enlarged, the lymph nodes around the mesentery, retroperitoneum and bilateral iliac vessels were enlarged, and the pelvis suffered multiple bone destruction.
| Complete blood count | Liver function |
|---|---|
| WBC: 3.17 × 109/L (3.5-9.5 × 109/L) HGB: 72 g/L (115-150 g/L) PLT: 79 × 109/L (125-350 × 109/L) |
LDH: 5338.6 IU/L (109-245 IU/L) B2 microglobulin: 4.52 mg/L (0.8-2.4 mg/L) α-HBDH: 4298.1 U/L (90-250 U/L) ALP: 196.9 IU/L (40-150 IU/l) ADA: 67.5 U/L (4-24 U/L) |
| Differential count | Coagulation analysis |
| BYLBXB: 1% (<0%) RDW-CV: 22.7% (11.5%-14.5%) RDW-SD: 58.1% (35.0%-56%) DHXB: 32% (40%-75%) GZXB: 18% (1%-5%) ESR: 93 mm/H (0-34 mm/H) |
APTT-TIME: 43.8 S (24.0-43.0 s) FBG: 4.77 g/L (2.0-4.0 g/L) DD.P: 30.71 mg/L (0-5 mg/L) |
| Count | Serum protein electrophoresis |
| CRP: 204.6 mg/l (<10 mg/L) HCPR: 188.58 mg/L (0-6 mg/L) PCT: 0.21 ng/ml (0-0.05 ng/ml) IL-6: 23.76 pg/ml (0.00-7.00 pg/ml) CA125: 53.07 u/ml (<35 U/ml) |
ALPHA1: 9.1% (2.9%-4.9%) |
Table 1. Laboratory investigations.
We completed the gastroscope to indicate duodenal bulbus ulcer (Figure 2). EUS examination revealed obvious hyperplasia and swelling of the entire pancreas, uneven reduction of echo, no obvious space-occupying lesions in the parenchyma, no dilatation of the biliopancreatic duct, no calculous shadow, thin film formation around the pancreas, and no obvious enlarged lymph nodes around the probe (Figure 3).
Figure 2: We completed the gastroscope to indicate duodenal bulbus ulcer.
Figure 3: EUS revealed obvious hyperplasia and swelling of the entire pancreas, uneven reduction of echo, no obvious space-occupying lesions in the parenchyma, no dilatation of the biliopancreatic duct, no calculous shadow, thin film formation around the pancreas, and no obvious enlarged lymph nodes around the probe. EUS-FNA (22G, negative pressure puncture method) was used to puncture the pancreatic body for 1 time.
The pathological results of EUS-FNA revealed B and T lymphocyte hybrid hyperplasia, with definite malignant change. The results of no treatment group were CEA-, CD20+(B lymphocytes), CD3+(T lymphocytes), CK19+(focal), ki-67+(30%), CgA-Syn+(focal)ck-CD138-Bone penetration results: Acute leukemia bone marrow (original cell 49%). The patient was transferred to the Department of hematology. Flow cytometry show abnormal cells account ed for 79% of nuclear cells, and the boundary between them and mature lymphocytes was not clear. They mainly expressed HLA-DR CD10 CD19 CD20 CD22 CD38 FMC 7 ki 67 cCD79a and the proliferation of myeloid system was obviously inhibited, indicating the proliferation of abnormal B lymphocytes and negative fusion gene. Cytogenic analysis of BM aspirate, indicated 46,x,dic(X,20)(P22.3; p13),-1, t(13 )(p36;q21),+der (12) add (12) (p13)add (12) (q24),add (13)q (32),der(15)t(1;1)(p31;p15), 22,+mar,inc[19]/46, XX. Direct anti human globulin experiments suggest positive multi-specific antibodies. The diagnosis of Acute B lymph ocytic leukemia was established.
The patient told the hematologist that she found discomfort in both breasts and could feel the lump herself, but there was no pain or tenderness. Physical examination revealed that the right mammary gland can be palpated with a hard and non-smooth surface mass, which was up to 3 cm, but no tenderness. The left mammary gland was normal. CT scan suggests hypoechoic nodules on the right breast (Figure 4). Mammography also found hypoechoic nodules on the right breast, classified as BI-RADS III. Then her doctor biopsied her breast nodules [3,4].
Pathological diagnosis: Lymphocytic hyperplasia, with diffuse infiltration large atypical lymphocy tes. Immuno histochemical staining suggests atypical lympho cytes: CD20(+), CD3(-), bcl-6(+), mum-1(+), c-myc(+,40%), bcl-2(-), p53(+,60-70%), CD5(-), CD30(-), CyclinD1(-), CD21(-), ki-6 7:80%; In situ hybridization: EBER (-); IgH gene rearrangeme nt monitoring clonal amplification peaks within the target band. The diagnosis of Diffuse large B cell lymphoma (WHO: Aggressive) also established too (Figure 4).
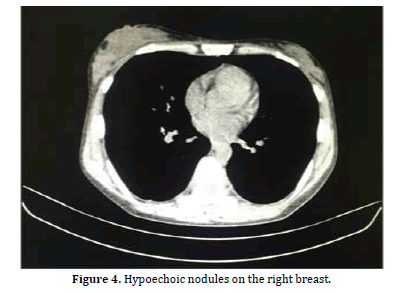
Figure 4: Hypoechoic nodules on the right breast.
Treatment
Before receiving chemotherapy, the doctor gave anti-hepatitis B virus (entecavir) and antiacid drugs. She received chemotherapy with R-hyperCVAD (Mabthera+Cytoxan+Vindesine+pirarubicin+Dexamethasone).
Outcome and follow-up
The patient obtained hematologic remission 3 weeks after onset of chemotherapy and lesions in pancreas and breast disappeared. Her doctor suggested a hematopoietic stem cell transplant or CAR-T, but she refused for personal reasons. She underwent 8 chemotherapy treatments succes sively, and completed the last one in April 2020. Bone penetration suggested complete remission of leukemia. PET showed no abnormal FDG metabolism in the whole body. She is in good condition without any discomfort. SPL (Secondary Pancreatic Lymphoma): Malignant lymphoma infrequently involves the pancreas.
The estimated frequency of primary NHL of the pancreas is about 2.2%. Secondary involvement of pancreas by NHL is rare [5]. Approximately 0.2%-2% of patients with NHL have pancreatic involvement at presentation. The clinical manifestation and radiological find PPL should be differentiated from those of pancreatitis, including, acute or chronic pancreatitis 4, autoimmune pancreatitis and occupying lesions, such as pancreatic carcinoma.
Pancreatic lymphoma is a disease exhibiting a male predominance (male: female ratio: 7:1) [6].
The age of the patients and duration of the symptoms are usually similar to those of pancreatic adenocarcinoma. In pancreatic lymphoma, symptoms are frequently unspecific. The most common presenting symptom reported is abdominal pain (83%), follow by abdominal mass (58%), weight loss (50%), jaundice (37%), acute pancreatitis (12%), small-bowel obstruction (12%) and diarrhea (12%). Frequent symptoms of NHL, such as fever, chills and night sweats are rare in PPL. All case of PPL reported to date in western countries are of the B-cell type, but some of T-Cell pancreatic lymphoma have been described in Japanese series.
The laboratory tests for PPL are non-specific, Lactate dehydrogenase and beta-2-microgolbulin are the helpful markers for the diagnosis of lymphoma. Tumor burde, β2-microglobulin levels >2 mg/l and high LDH levels are poor prognostic markers9. Anecdotal cases of elevated Ca19-9 levels in PPL have been reported. Bone marrow biopsy should be carried out for every patient who is suspect in ordered to complete the staging. CT imaging in pancreatic lymphoma also lacks specificity.
Two types of morphological presentation have been reported on CT; one is a tumor-like, localized, well-circumscribed mass presenting as a hypoechogenic mass, and the second is a diffuse enlargement infiltrating the pancreas. Pancreatic lymphoma with a lower incidence may show the symptoms of acute pancreatitis on CT scan which appears as diffuse pancreatic enlargement while the results of typical pancreatitis in CT including inflammation around the pancreas or fat stranding do not exist; otherwise, they can be seen usually minimal. Fluid accumulation around the pancreas, pancreatic fat necrosis and rupture of the pancreatic duct are not seen in lymphoma [7].
Vascular involvement by tumor can rarely be seen in lymphoma. Breast Lymphoma (BL): Breast lymphoma (BL) is a rare Non Hodgkin Lymphomas (NHL), accounting for only 0.04%-0.7%2 of breast tumors and about 2% of extrinsic lymphoma. The origin of breast DLBCL is unclear, but the prevalence rate is lower than that of the lack of lymphatic tissue in the breast, and the main clinical manifestation is a palpable breast mass with the right breast.
BL can be divided into two categories: Primary Breast Lymphoma (PBL) and Secondary Breast Lymphoma (SBL). The most common type Is Diffuse Large B-Cell Lymphoma (DLBCL). SBL refers to the simultaneous or subsequent invasion of systematic lymphoma, accounting for 17% of metastatic tumors of the breast. Not only are multiple lumps and bilateral breast involvement more common, but also accompanied by lymphoma symptoms (fever, weight loss and night sweats). However, in our case, we found lymphoma infiltrating the pancreas through EUSFNA and DLBCL was also found in the subsequent brea st mass, which is very rare among the reported cases. With bone destruction as the incipient performance of Acute lym phoblastic leukemia (Acute lymphoblastic leukemia, ALL) [8].
Acute leukemia with fever, infection, hemorrhage, liver, spleen and lymph node enlargement mainly, but the patients with subacute progressive bone pain as the main performance, laboratory examination early without routine blood apparently unusual, imaging revealed multiple bone destruction, multiple myeloma, and osteomyelitis, bone metastases with osteoporosis or bone destruction onset of acute lymphoblastic leukemia is relatively rare. Because in this kind of patient, do not have the clinical manifestation of common leukemia caused by blood image abnormality when onset, easy to lead to misdiagnosis and mistreatment. Therefore, if the patient has symptoms such as bone pain, bone destruction or fracture, it is necessary to exclude the possibility of hematologic malignancy, and it is very necessary to improve bone marrow biopsy [9].
The mechanism by which ALL causes bone damage is unclear. Mori et al. believed that it was related to the production of interleukin-6 and the increase of tumor necrosis factor in bone marrow mesenchymal cells, while Shimonodan et al. believed that it was related to the increased secretion of parathyroid hormone. Bone destruction or periosteum response is clear evidence of bone invasion by leukemic cells. Acute lymphoblastic leukemia with pancreatic involvement: Pancreatic infiltration of ALL is a very rare manifestation and only a few cases have been reported previously in adults. Clinical pancreatic involvement in ALL has also been reported in only 6 pediatric case reports [10].
Despite of the rarity, the hepatobiliary and pancreatic surgeon should be aware of this condition as accurate diagnosis is important for the following effective treatment. Classically, patients with acute leukemia presented with pancytopenia and clinical manifestations of pallor and fatigue caused by anemia; additionally, patients may present with unspecific symptoms, such as persistent or intermittent fever, similar to those of a viral infection.
Reported that pancreatic leukemia show 3 different morph ologic types: A well or ill-circumscribed nodular form; a diffuse, infiltrative form and a combination of the nodular and diffuse infiltrative forms on CT findings. Compared to CT imaging, MRI scan revealed more pathologic lesions in pancreas and extra findings of liver and retroperitoneal organs (bilateral kidney, lymph nodes, and lumbar and thoracic vertebrae) involvement, which were not well visualized on enhanced CT.
This indicated that MRI has much higher sensitivity in detecting tumor cell infiltrating lesions than CT. On EUS, pa ncreatic involvement by ALL showed multiple irregular hyp er-to-hypodensity lesions without clear margins. EUS-FNA of the pancreas is helpful to diagnose.
Pancreatic infiltration with lymphoma/leukemic cells is a rare manifestation of ALL or NHL, especially for adults. Leukemia/lymphoma of the pancreas should be considered if there are multiple or diffusing pancreatic nodules on imaging findings, and MRI possesses higher sensitivity than CT scan in identifying tumor lesions. Patients with ALL/NHL pancreas infiltration usually displayed unspecific symptoms. EUS-FNA is helpful to make the pathological diagnosis. FISH can better help in diagnosing and categorizing lymphomas.
[Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Google Scholar] [PubMed]
Citation: Liu L. Secondary Breast, Pancreatic Involvement with Diffuse Large B-Cell Lymphoma/Acute Lymphoblastic Leukemia: Case Report. JOP. J Pancreas. (2023) 24:804.
Copyright: This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.