Keywords
Colorectal cancer; Flexible sigmoidoscopy; Screening; Quality standards
Introduction
Colorectal cancer (CRC) is the third most common type of cancer worldwide with 1.4 million cases reported every year, causing approximately 70,000 cancer related deaths in 2012 [1]. Its incidence continues to increase worldwide, predominantly in Eastern Europe, Asia and South America, despite the fact that these numbers vary from region to region as they are influenced by lifestyle factors and age [2,3]. Earlier data from the UK reported CRC to be the second most common cause of cancer related death with approximately 30 000 new cases per annum with an average five years survival of 40% in 1990 [4,5]. Where hereditary components play a strong etiological role, most cases of colorectal cancer are sporadic and develop slowly over many years through, what is stated as, the adenoma–carcinoma sequence [6,7]. Screening can identify the CRC at an early stage leading to the effective treatment and reduction in cancer related mortality. A number of studies have shown that early detection of CRC followed by successful treatment increases the 5 years survival rate from 8.1% for patients treated in stage IV, to 93.2% for patients diagnosed and treated in stages I-II [8]. Although a variety of screening methods have been shown to reduce the incidence of CRC and cancer related mortality, organised screening programmes have yet to be implemented in most countries. Currently the most widely implemented screening methods are faecal tests and endoscopy-based screening. There is substantial amount of data which has shown that CRC screening with fecal occult blood testing (FOBT) can reduce mortality from CRC both in men and women [9]. Both colonoscopy and flexible sigmoidoscopy (FS) have also been shown to offer substantial reduction in the mortality from distal colorectal cancer [10-12]. Faecal test based screening methods have been implemented as a screening tool for CRC in several countries including the UK. Current screening guidelines for colorectal cancer recommend the same screening tools and the same screening age for men and women. However, the data has shown that the incidence of proximal advanced neoplasia is higher in women than men of similar age [13]. This means that the screening by FS may not be fully effective in women, unless detection of distal colon cancer is coupled with a colonoscopy for a detailed examination of the proximal colon. A recent meta-analysis by Holm et al, however, showed that the ratio of distal to proximal colon cancers is only marginally different in women between the age ranges of 55-59 years and 60-64 years [14].
CRC screening based on faecal test is currently offered to both males and females followed by a colonoscopy examination in those with positive faecal tests. A number of significant trials have supported the role of one stop FS as a successful screening tool in reducing both the incidence and mortality of CRC [15,16]. One stop flexible sigmoidoscopy is now proposed to be the new screening method for distal colon cancer screening in men and women aged 50 -60 yrs. Although quality markers for colonoscopies have been widely adopted in the UK, similar practice for FS is variable. In order for this procedure to be used as an effective screening tool, there is need for the standardization in terms of quality assurance. In this study, we reviewed the practice of flexible sigmoidoscopy in three district general hospitals in the UK to reflect on the wide variability that exists in this examination, and highlighted the need of standardization of quality performance indicators for FS.
Methods
This is a retrospective study which was carried out using an endoscopy database to identify patients who had a FS performed during 2009-2011 in three district general hospitals serving a population of 600,000. The information was also gathered from hospital electronic resources where needed. Since there was no quality performance standard for flexible sigmoidoscopy, the parameters studied were those which are defined as quality assurance standards for colonoscopy examination. In brief the parameters studied were; patient’s age, sex, extent of examination, grade of endoscopist, use of medications, procedure tolerance, bowel visualization, missed left sided lesions, missed cancers, appropriate indication, waiting time, informed consent, review interval postpolypectomy, surveillance intervals post cancer resection, adenomas detection rate, endoscope withdrawal time, procedure tolerance, practice of biopsies for chronic diarrhea/ suspected cancer and IBD, complications in term of hemorrhage, perforation and cardiopulmonary effects requiring prolonged stay and incidence of post-polypectomy hemorrhage and practice of Endotherapy. A complete examination was accepted when the colon was intubated to the splenic flexure or beyond, as documented by the endoscopist in the report. Mucosal visualization and patient tolerance were graded as good, fair and poor.
Results
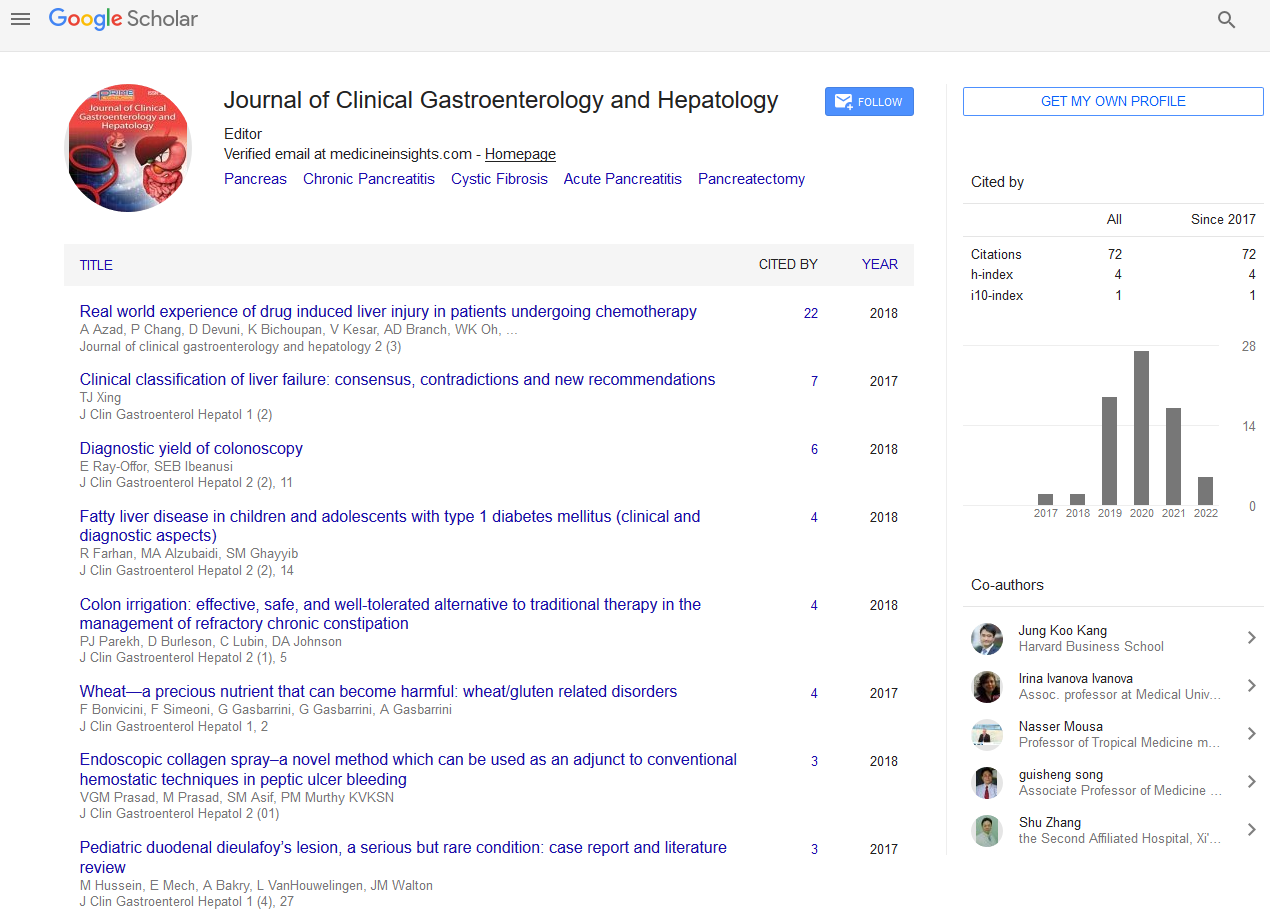
A total of 3823 procedure were recorded from the three different hospitals during the years 2009-2011. Indications of FS are shown in Figure 1.
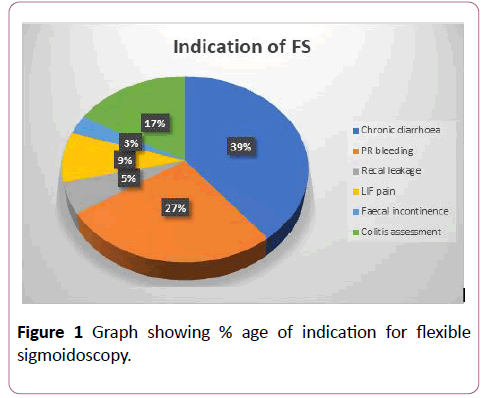
Figure 1: Graph showing % age of indication for flexible sigmoidoscopy.
Out of these procedures, 87.5% were carried out as outpatients and the remaining as in-patients. In about 92% of cases, a phosphate enema was used as a standard bowel preparation. Only 6.56% of cases were performed under full bowel preparation, using Movi-prep or Kleen-Prep, whereas 3.44% of cases were performed without any bowel preparation (Figure 2).
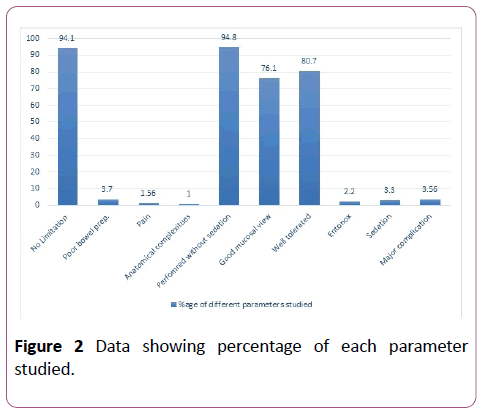
Figure 2: Data showing percentage of each parameter studied.
Poor bowel preparation accounted for procedure failure in 3.7% of the examinations, where the endoscopist had to abandon the procedure. Similarly, pain accounted for 1.56% of procedure failures, anatomical complexities and pathology encounter in 1% of failures, while in 94.1% of cases, there were no limitations. 94.8% of procedures were performed without sedation while 3.3% of patients required controlled sedation with midazolam +/- opioids (Midazolam range 1-5 mg median dose 3 mg, Morphine range 2.5-5 mg, mean=2.75 mg). A total of 2.2% of procedures were performed under Entonox only. Good mucosal visualization was achieved in 76.1% of cases while the procedure was abandoned due to number of limitations in about 33.9% of cases. In 80.7% of cases, the procedure was reported to be well tolerated by patients with a pain score of mild severity while in 17.7% of cases, impatiens experienced moderate pain and in about 1.56% of the cases, the procedure had to be abandoned due to severity of pain. Major complications in term of bleeding, cardiopulmonary effects requiring prolonged hospital stay and perforation, were encounterd in 3.56% of the cases. No death was recorded as a direct result of flexible sigmoidoscopy.
In 56.7% of cases, the scope was inserted to the splenic flexure. Among those where the procedure was documented as complete, the success rate was better in males compared to females (M=36.5%, F=22.2%, p<0.05). Overall, in 22.2% of cases, the examination was limited to the left colon and the scope did not reach the splenic flexure (M=7.4%, F=12.8). A total of 18.7% examinations were limited to the sigmoid colon (M=8.4%, F=10.3%) and in 4.6% of cases, the scope could not be passed beyond the rectum due to reasons explained below (M=2.6%, F=2%). The completion rates to different anatomical landmarks are shown in Figure 3.
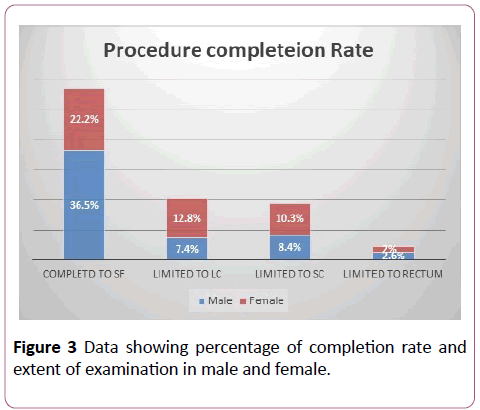
Figure 3: Data showing percentage of completion rate and extent of examination in male and female.
Overall, 37% of the examinations were reported normal while pathologies were detected in 50% of cases. In about 13% of cases, the procedure was reported as inconclusive due to patient intolerance or poor bowel preparation. The percentage of different pathologies detected is shown in Figure 4. A large proportion of patients were found to have diverticular disease (24% of cases). Whereas, benign polyps were found in 14% of examinations, non-specific colitis in 5% of cases, ischemic colitis in 3% of cases and IBD in 2% of cases. Further review of records revealed that 3.2% of those initially reported as nonspecific colitis were subsequently diagnosed as UC accounting for overall diagnosis of IBD as 5.2%. In our study, the overall distal cancer detected rate was 2%, while 13% of cases were reported to be non-conclusive, where no definite diagnosis was recorded. In 3.3% of cases, small benign polyps (<5 mm) were missed which were documented in subsequent examinations performed within the next 3 months. 11 cases (0.29%) of malignant lesions (polyps with high grade dysplasia/ malignant lesions) were missed on the first FS examination (detailed below in discussion) and later identified on subsequent examinations within 3 months (including colonoscopy and radiological studies). Mean number of biopsies taken for suspected malignant lesions were 5, for IBD were 3 and for chronic diarrhea a mean of 2 biopsies was taken. No information was available about scope withdrawal time or rectal retroflexion. The average waiting time for an urgent procedure was 15.5 days, for an emergency procedure was 56 hours, and for a routine procedure, the average waiting time was 8.2 weeks.
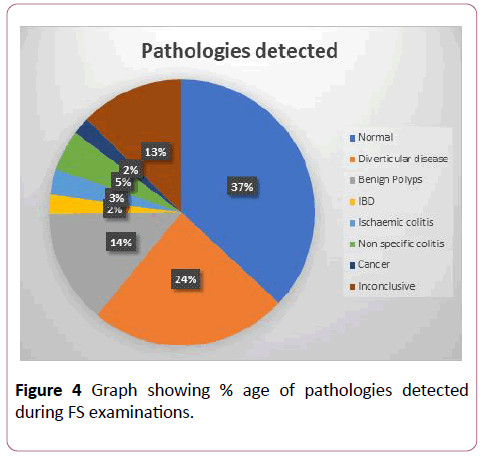
Figure 4: Graph showing % age of pathologies detected during FS examinations.
Discussion
CRC is a leading cause of cancer-related deaths worldwide, with early detection and removal reducing the mortality significantly. Two thirds of CRC, in their symptomless and adenomatous stage, are located in the sigmoid colon and rectum and can be detected early by the FS. Removal of such adenomas in the initial stages can offer long term protection against the development of CRC [17]. FS has been shown to be well accepted and tolerated by the general population, and hence has been proposed as a one-stop screening tool for the detection of CRC located in the distal colon [18-20].
CRC screening using a FS has been included in the UK bowel cancer screening program for both men and women at age 55 years. In 2013, six screening centres had started the one-stop FS screening and full invitation is expected in 2018. For colonoscopies, there has been widely studied and agreed upon parameters for the standardization of a complete examination. For example, Ileal intubation is a generally accepted landmark for the completion of c colonoscopy procedure, while photographic evidence of appendicular orifice, Ileo-caecal valve (ICV) and caecal tri-radial folds are also accepted as completion landmarks in other countries including the UK. In terms of FS, since there are no fixed identifiable anatomical landmarks in the left colon, completion rates can often be falsely reported by the endoscopists. Our study has shown the wide variability in the FS practice and the lack of standard criteria in reference to the extent of examination, procedure completion parameters and bowel preparation. While many endoscopists consider complete examination when the scope reached the splenic flexure, others believe that the sole examination of the sigmoid colon is adequate based on certain indications. The adenoma detection rate in our study was 14% with no significant variability between the three centres. Benign and small polyps (<5mm) were missed in about 3.3% of the cases which were detected and reported on subsequent examinations. Several factors are reported to affect the adenoma detection rate (ADR), for example studies that evaluated the quality assurance of colonoscopy examination suggested that scope withdrawal time, bowel cleansing agent, maneuvering of position during the examination, second observer, newer endoscopic technologies, and above of all, endoscopic skills were significantly related to ADR [21-23].
This data can be stipulated to conclude that similar parameters would also affect the ADR during FS examination. Another important factor related to missed lesions was considered to be rectal retroflexion. For example, a study by Pickhardt et al period found a 12% adenoma miss rate due to lack of rectal retroflexion during colonoscopy examination as these were located at the fold behind the anal verge [24]. Other studies also suggested failure of doing rectal retroflexion as a significant contributing factor in missed lesion rate [25,26].
In our studies, there was no documentation of rectal retroflexion although it was assumed that the majority of endoscopists perform rectal retroflexion albeit poor documentation. Another reason for this pathology miss rate was discrepancies between the extents of examination documented by the endoscopists. For example, some examinations were documented to be completed to the proximal descending colon and were reported as normal by the first endoscopist, but a lesion was found in the sigmoid colon on subsequent examination by the second endoscopist a few months later. Lack of good visualization, poor orientation of the colonic segment examined, and poor experience were among the most important causes of missed lesions. We also found that 11 cases of CRC (0.29%) were missed at the first FS examination and later found on subsequent examinations (including colonoscopy and radiological studies). For example, in two cases, a diagnosis of mild proctitis and proctosigmoiditis was made for PR bleeding with the examination documented to be complete to the descending colon, but on subsequent examination with a CT scan/repeat FS performed a few months later for worsening clinical conditions despite treatment, CRC in the proximal sigmoid colon was found in both cases. One of these patients later passed away due to wide spread metastasis and the other underwent partial colectomy. It is evident from these cases that in addition to poor endoscopist experience, the lack of adequate quality parameters for FS has had a significant impact on outcomes.
Performance characteristics of FS are highly dependent on the examiners, and many studies have highlighted its influence on FS practice. For example, a study by Atkin et al. reported wide variability among the examiners performing FS during Flexible Sigmoidoscopy Screening Trial in UK. This study found a large variation in polyp detection and adenoma detection rates across the examiners with coefficient variation (CVs) of 26% for polyp detection and 21% for adenoma detection among 13 FSG examiners each with 2400–3900 examinations performed [27]. Another study by Pinsky et al. examined variability in FSG performance among 64 examiners at 10 different centers and reported CV across examiners of 36% for screen-positivity rate, 29% for polyp detection rate, and 21% for adenoma detection rate [28]. Setting out quality standard criteria for FS, in particular, the extent of examination, procedure completion parameters and bowel preparation would reduce this variability.
In contrast to colonoscopy examinations where there are fixed identifiable landmarks (such as appendicular orifice, Ileocaecal valve and caecal tri-radial fold), there are no fixed and reliable anatomical landmark in the distal colon. Often the splenic flexure is identified with splenic reflection across colonic wall or triangular luminal appearance of transverse colon, but in real practice, it is not uncommon for a lesion documented in the transverse colon on initial endoscopic examination to be found in the descending colon on subsequent endoscopic examination or surgery. These discrepancies have significant clinical implications in terms of the accurate anatomical location of the pathology and the subsequent plan of the type and extent of surgery required for a particular lesion at a specific anatomical site. To deal with this issue, injection of tattoo ink adjacent to a lesion has now become a widely adopted practice to confirm the location and identification of the lesion on subsequent examinations or surgery.
Despite this, issues of inaccurate of lesion sites exist mainly due to performance characteristics of endoscopists. This observation has been supported by studies which have reported the tattoo inaccuracy and inadequacy during colonoscopy examinations. For example, a study by Conaghan et al. reported a case series of 52 patients who had a total of 54 lesions identified and were tattooed during colonoscopy examination. During subsequent surgical procedures, 7% of the tattoos were found to be inaccurate in location, when compared to the documentation by the endoscopist. In about 15.3% of cases, the tattoos were not even visible during laparoscopic resection [29].
In another study that evaluated the intraoperative visibility of colonic India ink tattoos, the tattoos were not visible in 31.5% of the patients. Moreover, during postoperative histologic examination of the specimens, the ink was detected in only 73.6% of the patients [30]. Regular and adequate training is required to improve the performance characteristics of endoscopists.
Since there is no agreed quality standard for flexible sigmoidoscopy examination, we believe that this study would be one of the initial studies to set a base for the need of defining quality assurance standards for flexible sigmoidoscopy examination. However, one of the limitations of the study was being a retrospective studies and majority of the information was gathered from endoscopy data base, endoscopy reports, patient clinical notes and hospital electronic systems. Also, limited information was available about the review interval after polypectomy, use of Endotherapy and withdrawal time.
Conclusion
In summary, with the increasing role of FS in CRC screening, the need for standardization of quality markers for FS examination has been suggested across the board. Healthcare providers performing flexible sigmoidoscopy in a screening program should have proficiency in performing this examination. Our study has also re-affirmed that FS is a widely practiced and useful diagnostic tool but to make it a more effective screening tool for colorectal cancer, a standardization process for quality indicators is required. Regular audits of the local practice and continue training and development of the skill is now widely proposed to enhance the screening and diagnostic role of FS.
References
- Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, et al. (2015) Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer 136.
- Center MM, Jemal A, Smith RA, Ward E (2009) Worldwide variations in colorectal cancer. CA Cancer J Clin 59: 366-78.
- https://www.wcrf.org/int/cancer-facts-figures/data-specific-cancers/colorectal-cancer-statistics
- Office of Population Censuses and Surveys (1994) Cancer statistics. Registration 1989. England and Wales. (HMSO, London); Series MBI; No 22.
- National Screening Committee (1998) A summary of the colorectal cancer screening workshops and background papers. National Screening Committee.
- Peipins LA, Sandler RS (1994) Epidemiology of colorectal adenomas. Epidemiol Rev 16: 273-297.
- Labianca R, Nordlinger B, Beretta GD, Brouquet A, Cervantes A, et al. (2010) Primary colon cancer: ESMO Clinical Practice Guidelines for diagnosis, adjuvant treatment and follow-up. Ann Oncol 21: v70-v77.
- O’Connell JB, Maggard MA, Ko CY (2004) Colon cancer survival rates with the new American Joint Committee on Cancer. (6th edn) staging. J Natl Cancer Inst 96: 1420-1425.
- Hewitson P, Glasziou P, Watson E, Towler B, Irwig L (2008) Cochrane systematic review of colorectal cancer screening using the fecal occult blood test (hemoccult): an update. Am J Gastroenterol 103: 1541.
- Hoff G, Grotmol T, Skovlund E, Bretthauer M (2009) Risk of colorectal cancer seven years after flexible sigmoidoscopy screening: randomised controlled trial. BMJ 338: b1846.
- Schoen RE, Pinsky PF, Weissfeld JL, Yokochi LA, Church T, et al. (2012) Colorectal-cancer incidence and mortality with screening flexible sigmoidoscopy. N Engl J Med 366: 2345-2357.
- Schoenfeld P, Cash B, Flood A, Dobhan R, Eastone J, et al. (2005) Colonoscopic screening of average-risk women for colorectal neoplasia. N Engl J Med 352: 2061-2068.
- Ulrik H (2017) Flexible sigmoidoscopy screening for colorectal cancer- Editorial BMJ 356: j75.
- Holme Ø, Schoen RE, Senore C, Segnan N, Hoff G, et al. (2017) Effectiveness of flexible sigmoidoscopy screening in men and women and different age groups: pooled analysis of randomised trials. BMJ 356: i6673.
- Segnan N, Armaroli P, Bonelli L, Risio M, Sciallero S, et al. (2011) Once-only sigmoidoscopy in colorectal cancer screening: follow-up findings of the Italian Randomized Controlled Trial—SCORE. J Natl Cancer Inst 103: 1310-1322.
- Atkin WS, Edwards R, Kralj-Hans I, Wooldrage K, Hart AR, et al. (2010) Once-only flexible sigmoidoscopy screening in prevention of colorectal cancer: a multicentre randomised controlled trial. Lancet 375: 1624-1633.
- Atkin WS, Morson BC, Cuzick J (1992) Long-term risk of colorectal cancer after excision of rectosigmoid adenomas. N Engl J Med 326: 658-662.
- UK Flexible Sigmoidoscopy Screening Trial Investigators (2002) Single flexible sigmoidoscopy screening to prevent colorectal cancer: Baseline findings of a UK multicentre randomised trial. Lancet 359: 1291-1300.
- Taylor T, Williamson S, Wardle J, Borrill J, Sutton S, et al. (2000) Acceptability of flexible sigmoidoscopy screening in older adults in the United Kingdom. J Med Screen 7: 38-45.
- Newcomb PA, Storer BE, Morimoto LM, Templeton A, Potter JD (2003) Long-term efficacy of sigmoidoscopy in the reduction of colorectal cancer incidence. J Natl Cancer Inst 95: 622-625.
- Aranda-Hernández J, Hwang J, Kandel G (2016) Seeing better-Evidence based recommendations on optimizing colonoscopy adenoma detection rate. World J Gastroenterol 22: 1767.
- Bressler B, Paszat LF, Vinden C, Li C, He J, et al. (2004) Colonoscopic miss rates for right-sided colon cancer: A population-based analysis. Gastroenterol 127: 452-456.
- Kaminski MF, Regula J, Kraszewska E, Polkowski M, Wojciechowska U, et al. (2010) Quality indicators for colonoscopy and the risk of interval cancer. N Engl J Med 362: 1795-1803.
- Pickhardt PJ, Nugent PA, Mysliwiec PA, Choi JR, Schindler WR (2004) Location of adenomas missed by optical colonoscopy. Ann Intern Med 141: 352-359.
- Chandran S, Parker F, Vaughan R, Mitchell B, Fanning S, et al. (2015) Right-sided adenoma detection with retroflexion versus forward-view colonoscopy. Gastrointest Endosc 81: 608-613.
- Hewett DG, Rex DK (2011) Miss rate of right-sided colon examination during colonoscopy defined by retroflexion: An observational study. Gastrointest Endosc 74: 246-252.
- Atkin W, Rogers P, Cardwell C, Cook C, Cuzick J, et al. (2004) Wide variation in adenoma detection rates at screening flexible sigmoidoscopy. Gastroenterol 126: 1247-1256.
- Pinsky PF, Schoen RE, Weissfeld JL, Kramer B, Hayes RB, et al. (2005) Variability in flexible sigmoidoscopy performance among examiners in a screening trial. Clin Gastroenterol Hepatol 3:792-e1.
- Conaghan PJ, Maxwellâ€ÂÂÂÂArmstrong CA, Garrioch MV, Hong L, Acheson AG (2011) Leaving a mark: The frequency and accuracy of tattooing prior to laparoscopic colorectal surgery. Colorectal Dis 13: 1184-1187.
- Aboosy N, Mulder CJ, Berends FJ, Meijer JW, Sorge AA (2005) Endoscopic tattoo of the colon might be standardized to locate tumors intraoperatively. Rom J Gastroenterol 14: 245-248.