- (2014) Volume 15, Issue 6
Ara B Sahakian, James L Buxbaum, Jacques Van Dam
Advanced Digestive Health Center, Keck/USC Medical Los Angeles, CA, USA
Received June 18th, 2014 – Accepted October 22nd, 2014
Pancreatitis remains as one of the most frequent and serious complications of ERCP. Research has identified several patient-related and procedural risk factors, which help guide the endoscopist in prophylaxis and management of pancreatitis. Recent studies have had a major impact on both procedural techniques and pharmacological methods for prophylaxis of post-ERCP pancreatitis. The purpose of this article is to review the relevant literature and describe the most recent and effective approaches in prevention and management of post-ERCP pancreatitis.
Abdominal Pain; Pancreatic Ducts; Pancreatitis
Endoscopic retrograde cholangiopancreatography (ERCP) is a fundamental tool in the management of in magnetic resonance cholangiopancreatography (MRCP) and endoscopic ultrasound (EUS), ERCP has biliary and pancreatic diseases. With technological advances evolved into a therapeutic rather than diagnostic procedure. Nevertheless, these procedures carry significant risk, with post-ERCP pancreatitis (PEP) being the most frequent and dreaded of these [1] (Figure 1). The incidence of pancreatitis after ERCP is approximately 5-10% [2-4], but exceeds 30% in certain high-risk groups [5]. It is essential for the modern advanced endoscopist to recognize PEP, understand the risk factors, measures for prevention, and the necessary steps in management. The primary aim of this article will be to review the recent advances in prevention and management of PEP.
PEP is defined as two of the following three criteria being present post-procedure: 1) epigastric pain, 2) amylase or lipase greater than three times the upper limit of normal, and 3) cross-sectional imaging findings consistent with pancreatic inflammation [6]. However, the gastrointestinal endoscopist must be cognizant that after ERCP, two of these criteria may be present, while acute pancreatitis may not. For example, epigastric abdominal pain after ERCP can occur from other causes, such as duodenal perforation, excessive contrast injection, or air insufflation. Amylase and lipase elevations often occur after ERCP even in asymptomatic patients [7]. Thus, imaging becomes an important modality to evaluate for PEP if there is any doubt about the diagnosis [8].
For purposes of research it is often requisite that the pain be persistent for 24-48 hours and have a significant severity using a validated pain scoring system [7, 9, 10]. In their classic paper Cotton and colleagues [6] classified post ERCP pancreatitis into mild, moderate, and severe categories. Mild pancreatitis requires hospital admission for 2-3 days, moderate pancreatitis for 4-10 days, and severe pancreatitis for >10 days, or the presence of hemorrhagic pancreatitis, phlegmon, pseudocyst, or the necessity for any intervention, such as percutaneous drainage or surgery.
More recently, investigators have used the Atlanta criteria and subsequent revisions to stage the severity of PEP. The Atlanta Classification defines severe acute pancreatitis by the presence of one of the following: pancreatic abscess, creatinine >2mg/dL after rehydration, PO2<60 mmHg, systolic blood pressure <90 mmHg, pseudocyst, pancreatic necrosis involving >30% of the gland, gastrointestinal bleeding >500 ml, and death. Mild pancreatitis is defined as the absence of criteria of severe pancreatitis. Recent revisions classify those with pancreatic necrosis >30% and/or peri-pancreatic collections without persistent or multi-organ failure as moderate pancreatitis. These patients with moderately severe pancreatitis have significant morbidity but low mortality [11-13].
While severe PEP is rare, occurring in <1% of ERCP, it carries significant morbidity and mortality and is a leading source of medical-legal action against endoscopists [5].
Patient-Related Risk Factors
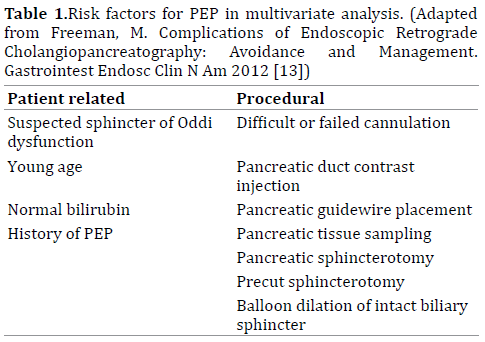
Several prospective studies have identified patient related risk factors for PEP [2-4, 14-17]. Research has consistently demonstrated that suspected sphincter of Oddi dysfunction (SOD) and history of PEP are the strongest predictors of PEP [2, 14], with a meta-analysis demonstrating an increase in risk for PEP of 4-fold and 2-fold respectively [18]. Risk factors may also be additive. Patients with several risk factors, such as SOD, difficult cannulation, and normal bilirubin, were shown to have a risk of PEP of approximately 40% [2].
However, it is important to emphasize that in SOD patients, it is not manometry that confers an increased risk of PEP, but rather the underlying condition itself. Singh et al. [19] demonstrated in patients with suspected SOD who underwent ERCP with manometry the risk of post ERCP pancreatitis was no higher than those with suspected SOD who underwent ERCP without manometry (OR=0.72; 95% CI 0.08-9.2).
Female sex and normal bilirubin level have also been reported as risk factors for PEP in multiple prospective studies [2, 4]. Older data implicated a non-dilated bile duct as a risk factor for PEP [17], however, this has not been corroborated in other analyses [2, 16]. Certain patient characteristics, such as the presence of chronic pancreatitis, older age, and pancreatic malignancy, are considered protective against PEP [2, 20].
Ampullary trauma has often been implicated as a causative factor in PEP. Difficult cannulation, although a vague term, is generally described as several or prolonged attempts to obtain access to the desired duct. Several studies have shown difficult cannulation to be a risk factor for PEP [2, 4, 14]. Access sphincterotomy using a needle knife (i.e. “pre-cut” sphincterotomy) has also been shown to be an independent risk factor for PEP [4, 16, 21], including a meta-analysis which showed relative risk of PEP of 2.7 [18]. However, precut sphincterotomy as a predictor of PEP remains controversial, as it is often preceded by difficult cannulation, which also predisposes to PEP. Interestingly, meta-analyses by Cennamo et al. [22] and Gong et al. [23] now suggest that early pre-cut sphincterotomy may actually reduce the risk of PEP. However, of the six trials included in these meta-analyses, only one demonstrated statistical significance for PEP risk reduction with early pre-cut sphincterotomy [24].
Balloon dilation of an intact papilla has also been implicated as a risk factor for PEP (OR 4.5) [2]. A randomized controlled multi-center study of 237 patients comparing stone extraction after sphincterotomy compared to dilation of a native papilla showed that pancreatitis occurred significantly more often in the dilation group (15.4% versus 0.8%). Moreover, 2 deaths from pancreatitis occurred in the dilation group [25]. Based on this data, dilation of a native papilla has become an ERCP technique that is seldom used, aside from certain situations such as uncorrectable coagulopathy. Interestingly, balloon dilation after biliary sphincterotomy has not been shown to confer an increased risk of PEP and is emerging as a favored technique for large stone extraction [26-28]. It is theorized that the initial biliary sphincterotomy separates the biliary orifice from the pancreatic orifice, thereby directing the force of balloon toward the bile duct and away from the pancreatic duct [29].
Several other techniques have been implicated as independent risk factors for PEP. Both major and minor papilla pancreatic sphincterotomy have been shown to increase the risk of PEP [2, 14]. Trainee involvement may also be associated with increased PEP risk [14]. Multiple contrast injections have been demonstrated to be a risk factor in multiple prospective trials [2, 4], although one large prospective multi-center study did not confirm this data [14] (Tables 1-3).


General Approaches
The best approach to preventing PEP is to avoid performing ERCP in patients with marginal indications. ERCP is often higher risk and lower yield in patients with weaker indications. Patients with obstructive physiology, such as bile duct stones or pancreatic cancer, tend to tolerate ERCP well. Whereas patients who require ERCP for higher risk indications, such as sphincter of Oddi dysfunction or pancreatic endotherapy, should be referred to an expert center [30]. Diagnostic ERCP should generally be avoided. Powerful alternative imaging modalities such as MRCP and EUS will provide the necessary diagnostic information in most cases, while avoiding the risks of ERCP.
Pancreatic Duct Stents
Pancreatic duct stents have been increasingly used in recent years for prevention of PEP. They are thought to prevent pancreatitis by allowing adequate ductal drainage in the setting of ampullary trauma or over-injection of contrast. There are now several meta-analyses that confirm the effectiveness of pancreatic duct stents in preventing PEP [31-33]. The most recent by Choudhary et al. [31] included 8 randomized controlled trials (656 subjects) and showed that prophylactic placement of pancreatic duct stents in high-risk patients lowered the odds of PEP (OR 0.22; 95% CI, 0.12-0.18) with an absolute risk difference of 13.3%. Stent placement reduced the likelihood of mild and moderate pancreatitis, but only a trend toward reduction of severe pancreatitis was seen. However, two other metaanalyses have shown a decreased likelihood of severe pancreatitis in subgroup analysis [32, 34].
Prophylactic stenting of the pancreatic duct (Figure 2A) has been shown to decrease the risk of PEP in several situations, including SOD, pre-cut sphincterotomy, balloon dilation of the biliary sphincter, ampullectomy, and pancreatic guidewire assisted cannulation of the bile duct [30, 35, 36]. Prophylactic stenting should be considered in all patients prior to performing pre-cut sphincterotomy. This is supported by data showing that the rate of PEP is only 4.3% when pre-cut sphincterotomy for biliary access is performed over a pancreatic stent [37]. It is also thought that the stent may help delineate the ductal anatomy and assist in selective cannulation of the biliary orifice after pre-cut sphincterotomy. There is also data to suggest that pancreatic duct stenting may be protective in all patients undergoing ERCP [38]. However, the standard of care for prophylactic stenting remains in patients at high risk of PEP. This is also corroborated by a cost-effectiveness analysis, which demonstrates the superiority of prophylactic stenting in patients at high risk of PEP compared to stenting all patients or no patients undergoing ERCP [39].
The endoscopist must also consider the optimal type of pancreatic duct stent to place for PEP prevention. Small caliber short 5F or 3F stents are most commonly used due to their ease of placement compared to longer stents. 5F stents are often preferred because they can be placed over a 0.035” guidewire, obviating the need to switch to a smaller diameter wire, making them faster and easier to use. No difference in PEP rates has been found between the two sizes [40]. Despite the obvious advantages of prophylactic pancreatic stenting, certain drawbacks do exist. Some reports have shown that stents can cause damage to the pancreatic duct, occurring in up to 80% of patients when 5F or larger sized stents are placed [41, 42]. This may be an argument for smaller caliber stents made of softer material. However, high quality comparative data is lacking. Other unexpected pitfalls include inadvertent advancement of the stent into the pancreatic duct and spontaneous internal migration, This complication can be prevented by use of a stent that has a single pigtail on the duodenal end. Attempt and subsequent failure to stent the pancreatic duct is also considered a significant risk factor for PEP [43]. Pancreatic duct disruption caused by a pancreatic stent is a rare but has been reported [35]. Thus, only those with considerable expertise and specifically trained in this technique should only perform stenting of the pancreatic duct. Pancreatic duct stenting is now considered a requisite skill for all trainees completing a 4th year advanced endoscopy fellowship.
Cannulation using a guidewire (Figure 2B) rather than contrast-directed cannulation is considered a valuable technique in preventing PEP. This enables access to the desired duct using a soft-tipped guidewire, while avoiding contrast injection into the pancreatic duct. It is also thought that inadvertent advancement of a guidewire into the pancreatic duct may be less traumatic to the pancreas than contrast opacification. Wire-guided cannulation first received considerable attention when a prospective study randomized 400 patients to wireguided cannulation versus conventional contrast directed cannulation. Cannulation success rates were similar, but no PEP developed in the guidewire group, whereas PEP occurred in 4% in the contrast group (P<0.01) [44]. Since then, several studies have indicated that wire-guided cannulation reduces the risk of PEP. Subsequent large randomized trials have consistently demonstrated PEP rates of 2-9% in wire-guided versus 10-17% in contrast guided ERCP [44-46], which has also been supported by several meta-analyses [47-50]. However, there is some heterogeneity in the data [47] and the wireguided technique has come under scrutiny as two recent prospective studies have not shown a reduction in rates of PEP [51, 52]. There may be several reasons for the mixed data on wire-guided cannulation. First, the endoscopists performing the procedures in these studies may have variable experience with one particular technique, which can potentially bias the data. The second reason is that “wire-guided cannulation” is not a single technique, but rather a method of cannulation that can be performed in many different ways. Pure wire-guided cannulation involves introducing the wire into the biliary orifice, while keeping the tip of the sphincterotome in the duodenal lumen. Another variation of wire guided-cannulation is when the tip of the cannula is inserted into the common channel and the wire is gently probed back and forth at varying angles until the biliary orifice is entered. The wire can be controlled by the assistant or by the physician. A hybrid method also exists, where the desired duct is first opacified with contrast, then the guidewire is used to access the duct [30]. Most advanced endoscopists use a combination of these methods, making generalization of studies on wire-guided cannulation problematic. However, based on the abundance of favourable data, it is reasonable to conclude that the wire-guided technique should be considered a first-line modality for cannulation.
Prophylactic medications
Until recently, research on pharmacologic prophylaxis of PEP yielded generally disappointing results. Vasoactive drugs, such as nifedipine, lidocaine, epinephrine, botulinum and nitrates have been administered with the aim of improving ductal drainage by relaxing the sphincter of Oddi [53]. Controlled trials of these agents yielded disappointing results for prevention of PEP [53-55]. Glyceryl trinitrate was shown to lower rates of PEP in two meta-analyses [56, 57]; however, in a third meta-analysis, the drug was not found to perform any better than placebo in the prevention of PEP [53]. Due to the adverse side effect profile glyceryl trinitrate is not recommended for use in PEP prophylaxis [58]. An additional approach has been to “rest” the glandular activity of the pancreas. Somatostatin and its synthetic variant octreotide have been studied extensively with disappointing results [59].
Multiple anti-inflammatory agents have been administered prophylactically with the aim of curbing the chain reaction that underlies PEP. In a multicenter trial of >1000 patients Sherman et al. [5] randomized patients to prophylactic corticosteroids versus placebo. No difference was found between the treatment and control group. The protease inhibitor gabexate mesylate, which inhibits the activity of trypsin, has been administered prophylactically to prevent PEP and promising initial data prompted widespread use in Japan. However, a meta-analysis of 9 trials suggested that it was not effective [59]. These results, along with the inconvenient route of administration (continuous infusion), have limited its use. Several other agents including heparin, interleukin-10, allopurinol, phosphodiesterase-5 inhibitor and N-acetylcysteine, have not shown any benefit in reducing the risk of PEP [60-64]. Much more favorable results have been recently reported with the use of non-steroidal anti-inflammatory drugs (NSAIDs). Single center randomized trials of prophylactic rectal administration of diclofenac and indomethacin have shown promise in preventing PEP, with a meta-analysis showing a 64% decrease in risk of PEP and a 90% decrease in risk of moderate/severe PEP [65-67]. This prompted the European Society of Gastrointestinal Endoscopy (ESGE) to recommend rectal NSAIDS for prophylaxis of PEP [58]. A major multicenter randomized controlled trial by Elmunzer et al. [10] subsequently demonstrated that rectal administration of indomethacin significantly decreases the risk of PEP. The investigators randomized 602 patients at high risk of PEP to 100mg of rectal indomethacin or placebo given immediately after ERCP. Suspected sphincter of Oddi dysfunction was the risk factor for PEP in 82% of the patients. The authors found that PEP occurred significantly less in the indomethacin group (9.2%) compared to the placebo group (16.9%). Rectal indomethacin also significantly reduced the likelihood of moderate/severe pancreatitis. Since the emergence of this data, administration of rectal indomethacin has become the standard practice for patients at high risk of PEP. Because of the favourable side effect profile of indomethacin, many practicing endoscopists currently use it in all patients with intact papillae, although data to support this practice is lacking.
The management of PEP primarily consists of supportive measures and is similar to management of other causes of acute pancreatitis. Nutrition and multidisciplinary management of complications such as necrosis and abscesses is critical (Figure 3). Brown et al. [68] have shown that hemoconcentration, as measured by elevated blood urea nitrogen and creatinine, is associated with inadequate resuscitation and necrosis and also may correlate with mortality in acute pancreatitis [69, 70]. Early and aggressive fluid resuscitation appears to decrease the risk of systemic inflammatory response syndrome and organ failure [71]. Randomized work suggests that resuscitation with lactated Ringer’s solution decreases the risk of SIRS compared to use of saline. Wu et al. propose that lactated Ringer’s, which is a more pH balanced fluid, may be less likely to stimulate inflammatory mediators which function better in an acidic environment [72]. Patients at the highest risk of PEP (e.g., SOD, history of PEP, pancreatic endotherapy) may be admitted post-procedure for bowel rest, IVF, and to initiate early therapy should pancreatitis occur. Retrospective work suggests that these patients are less likely to develop moderate or severe pancreatitis [73].
Early and aggressive intravenous fluid (IVF) resuscitation is recommended as a mainstay in the management of patients with acute pancreatitis. ERCP is unique in that it reliably induces pancreatitis in a significant proportion of patients, which makes it an appealing setting for prophylaxis. In a small pilot trial we recently randomized 62 patients undergoing first-time ERCP to aggressive or standard hydration using lactated Ringer's solution [9]. None of the patients in the aggressive hydration group developed PEP, compared to 17% of patients in the standard hydration group (p=0.016). Two multicenter randomized trials testing this theory are underway in the Midwestern United States and the Netherlands.
Another group of investigators reported a case series of six patients at presumed high-risk of severe pancreatitis who underwent emergency rescue ERCP early in the course of PEP for placement of a small caliber pancreatic stent [74]. Pancreatic necrosis did not occur in any of the patients. Although the results are promising, this was a small case series, making the data difficult to extrapolate. Furthermore, one must consider the risk of failing to place a pancreatic stent in the setting of an edematous papilla, potentially worsening the course of pancreatitis. Therefore, this approach cannot be recommended until larger scale randomized controlled data is available.
ERCP is a powerful tool for the treatment of biliary and pancreatic diseases. However, it should be used judiciously, and alternate imaging modalities such as EUS and MRCP should be considered in diagnostic cases. Research has identified several factors, both procedural and patientrelated, that increase the risk of PEP. The endoscopist should consider placing a prophylactic small caliber pancreatic stent if the any of the strongest risk factors (SOD, history of PEP, pancreatic endotherapy, pre-cut sphincterotomy) or if several minor risk factors are present. Rectally administered indomethacin should be considered if any risk factor for PEP is present. Wire-guided technique should be employed as a first-line method of cannulation. Although data is limited, prophylactic aggressive fluid hydration with lactated Ringer’s solution can be considered in all patients with intact papillae who are not at risk of fluid overload. Future research is necessary to determine the optimal combination of current prophylactic agents and for discovery of new methods to prevent PEP.
Authors have no conflict of interest.