Original Article - (2021) Volume 22, Issue 1
Manami Ikemune1, Kazushige Uchida2, Masato Yanagawa1, Satoshi Tsukuda1, Takashi Tomiyama1, Toshihiro Tanaka1, Yugo Ando1, Tsukasa Ikeura1, Takashi Yamaguchi1, Toshiro Fukui1, Akiyoshi Nishio1, Kazuichi Okazaki3
1The third department of internal Medicine, Division of Gastroenterology and Hepatology, Kansai Medical University, 2-5-1 Shinmachi, Hirakata, Osaka 573-1010, Japan
2Department of Gastroenterology and Hepatology, Kochi Medical School, Kochi University, Kohasu, Oko-cho, Nankoku, Kochi, 783-8505, Japan
3Kouri Hospital, Kansai Medical Univerisity, Kansai Medical University, 8-45, Kourihondori, Neyagawa, Osaka, 572-8551 Japan
Received Date: November 24th, 2020; Accepted Date: December 21st , 2020
Introduction Type 1 autoimmune pancreatitis is a chronic fibro-inflammatory disorder. We previously reported the involvement of M2 macrophages and basophils in autoimmune pancreatitis. It is reported that Group 2 innate lymphoid cells (ILC2s) and basophils play an important role in asthma. Thus, this study investigated the roles of innate lymphoid cells in autoimmune pancreatitis. Subjects and methods We recruited 28 patients with autoimmune pancreatitis (25 men and 3 women; mean age, 68.4 years) who were not receiving steroid therapy, 10 patients with chronic pancreatitis (CP; 2 women and 3 men with idiopathic CP and 5 men with alcoholic CP; mean age, 65.9 years), and 30 healthy controls (HCs; 5 women and 25 men; mean age, 66.9 years). Peripheral ILCs were analyzed using flow cytometry. We also analyzed two types of ILC2s (lineage− CD127+ CD161+ c-Kit+/− CRTH2+ and lineage− CD25+ IL-33R+ cells). Results The proportions of ILC2s and ILC3s were significantly higher in the autoimmune pancreatitis (0.140% ± 0.083% and 0.055% ± 0.043%, respectively) and CP groups (0.119% ± 0.055% and 0.051% ± 0.040%, respectively) than in the HC group (0.054% ± 0.039% and 0.018% ± 0.017%, respectively). The proportion of ILC1s did not differ among three groups. There was no significant correlation between the counts of the two ILC2 types. Conclusion Elevated ILC2 and ILC3 counts may be involved in the development of chronic pancreatic inflammation.
Type 1 Autoimmune Pancreatitis; Chronic Pancreatitis; Innate lymphoid cells; fibrosis
AIP autoimmune pancreatitis; CP chronic pancreatitis; ILC innate lymphoid cells
The first case of autoimmune pancreatitis (AIP) was reported in a patient with chronic pancreatitis (CP) and hypergammaglobulinemia in 1961 [1]. Thirty years later, these pathological features were defined as lymphoplasmacytic sclerosing pancreatitis [2] and proposed to be referred to as “autoimmune pancreatitis” by Yoshida et al. [3] Hamano et al. described high serum immunoglobulin G4 (IgG4) levels in patients with AIP [4], and this condition is currently classified as type 1 AIP [5].
Although both type 1 AIP and established progressive CP are chronic fibro-inflammatory disorders, they can be distinguished by their clinicopathological features. Type 1 AIP is characterized by a lack of pain, narrowing of the main pancreatic duct, swollen parenchyma with abundant infiltration of IgG4-positive plasma cells, and storiform fibrosis, and it is often associated with other organ involvement [6, 7]. Therefore, type 1 AIP is currently defined as a pancreatic manifestation of IgG4-related disease (IgG4- RD) [8, 9, 10]. Conversely, patients with CP exhibit recurrent pain, parenchymal atrophy, fibrosis, duct distortion strictures, calcification, pancreatic exocrine/endocrine dysfunction, and dysplasia [11, 12]. However, recent studies suggested that some patients with AIP will display similar characteristics as those with CP, such as pancreatic calcification with pancreatic exocrine/endocrine dysfunction [13, 14], but the precise mechanism behind this finding remains unclear.
We previously investigated the roles of acquired immune responses in patients with type 1 AIP. Our previous report indicated that decreased counts of naïve regulatory T cells (naïve Tregs; CD4+ CD25+ CD45RA+) may play a critical role in the development of type 1 AIP [15]. Increased counts of inducible costimulatory (ICOS)+ Tregs influence IgG4 production in type 1 AIP via IL-10. In addition, ICOS− Tregs are responsible for fibrosis via TGF-β [16]. We also reported that CD19+ CD24+ CD38high regulatory B cell (Bregs) counts were increased in type 1 AIP, whereas CD19+ CD24high CD27+ Bregs counts were decreased. These data indicate that CD19+ CD24high CD38high Bregs counts are increased to suppress disease activity, whereas CD19+ CD24high CD27+ Bregs might be involved in the development of type 1 AIP [17].
Innate immune responses have attracted substantial attention for their possible involvement in several diseases. In the context of the innate immune response, we reported that M2 macrophages and basophils may be involved in the development of type 1 AIP [18, 19]. M2 macrophages play an important role in pancreatic tissues in patients with type 1 AIP through Toll-like receptor 7 (TLR7) [18]. Moriyama et al. reported that IL-33 produced by M2 macrophages promotes Th2 cytokine production, conferring a Th2-dominant pathophysiology in IgG4- related sialadenitis [20]. M2 macrophages might play an important role in IgG4-RD in addition to type 1 AIP. Moreover, in our recent report, basophils were activated through TLRs in type 1 AIP [19]. Watanabe et al. also reported that basophils promote IgG4 production through B cell-activating factor and IL-13 in type 1 AIP [21]. In allergic diseases, basophils were required for the transformation of inflammatory monocytes into M2 macrophages [22]. It has also been reported that the interaction of basophils and group 2 innate lymphoid cells (ILC2s) controls IL-33-mediated allergic responses [23].
ILCs have important roles in the innate immune system. They do not express a lineage marker, and thus, they directly respond to sensitization through recognition by a pattern recognition receptor. Additionally, they contribute to intestinal homeostasis and tissue remodeling, and they are associated with allergic and inflammatory diseases [24, 25]. They are categorized into three groups (ILC1s, ILC2s, and ILC3s), according to the interleukins, cytokine production, and transcription factors essential for their development [26]. Three subsets of ILCs are well documented: lineage− CD127+ CD161+ CD117− (c-Kit−) CD336− (NKp44−) ILC1s, lineage− CD127+ CD161+ c-Kit− or c-Kit+ CRTH2+ ILC2s, and lineage− CD127+ CD161+ c-Kit+ NKp44− ILC3s [26]. Based on the presence of different surface markers, lineage− IL33R+ CD25+ ILC2s have also been described [27]. To clarify the role of ILCs, we investigated ILCs in peripheral blood from patients with type 1 AIP and CP in this study.
Patients and Controls
We recruited 28 patients with type 1 AIP (25 men and 3 women; mean age, 68.4 years; range, 52–80 years) with no history of steroid therapy, 10 patients with CP (2 women and 3 men with idiopathic pancreatitis and 5 men with alcoholic pancreatitis; mean age, 65.9 years; range, 43–82 years), and 30 healthy controls (HCs; 25 men and 5 women; mean age, 66.9 years; range, 50–83 years) for this study (Tables 1, 2, 3). All patients and HCs visited Kansai Medical University Hospital. All patients with type 1 AIP were diagnosed according to the international consensus diagnostic criteria for AIP [5], whereas those with CP were diagnosed according to the clinical diagnosis criteria for CP proposed by the Japan Pancreatic Society [28]. HCs consisted of people visiting Kansai Medical University Hospital because of positivity on fecal occult blood testing; however, there were no findings on colonoscopy. Patients and HCs who did not provide written informed consent were excluded. This study was approved by the ethics committee of Kansai Medical University.

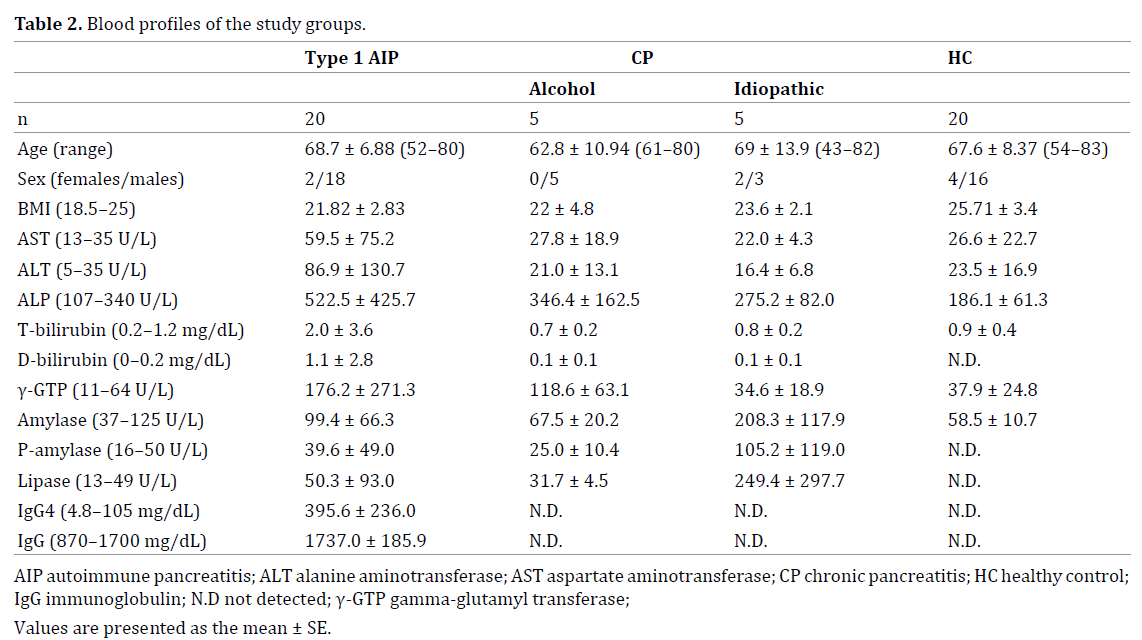
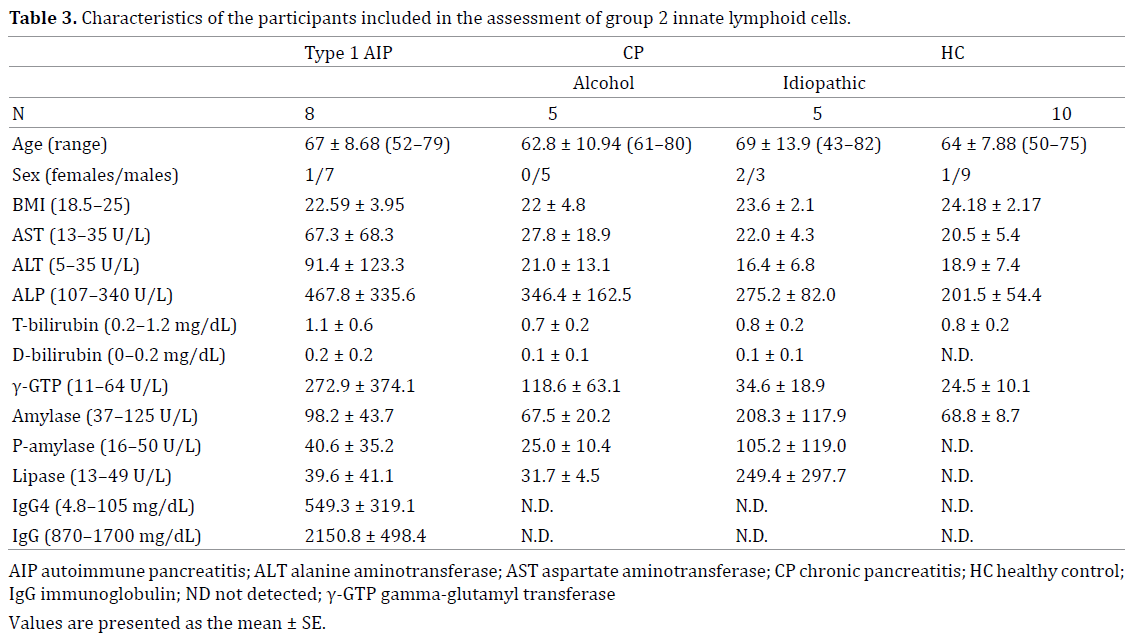
In this study, one cohort was examined for four subsets of ILCs: lineage− CD127+ CD161+ CD117− (c-Kit−) CD336− (NKp44−) ILC1s, lineage− CD127+ CD161+ c-Kit−/+ CRTH2+ ILC2s, and lineage− CD127+ CD161+ c-Kit+ NKp44− or lineage− CD127+ CD161+ c-Kit+ NKp44+ ILC3s (Figure 1a–e, Table 2) [26]. A second cohort was examined for lineage− CD127+ CD161+ c-Kit−/+ CRTH2+ ILC2s and lineage− IL33R+ CD25+ ILC2s (Figure 1f–g, Table 3) [27]. All patients with CP were included in both analyses.
Figure 1. Gating strategy for group 1 innate lymphoid cells (ILC1s), ILC2s, NKp44− and NKp44+ ILC3s, and lineage− CD25+ IL-33R+ ILC2s from peripheral blood.
First, the cells were gated using forward scattered light and side scattered light (a). Lineage− CD127+ cells were isolated. The lineage markers included CD3, CD5, TCRαβ, and TCRγδ as T-cell markers; CD19 as a B cell marker; CD11b as a neutrophil and monocyte marker; CD11c as a monocyte and macrophage marker; CD56 as an NK cell marker; and FcεR1α as a basophil and mast cell marker (b). CD161+ cells were isolated from lineage− CD127+ cells (c). Lineage− CD127+ CD161+ c-Kit− or c-Kit+ CRTH2+ cells were defined as ILC2s (d). Lineage− CD127+ CD161+ c-Kit− CRTH2− cells were defined as ILC1s. Lineage− CD127+ CD161+ c-Kit+ CRTH2− were defined as ILC3s. ILC3s were divided into NKp44+ and NKp44− subsets (e).
Lineage− CD25+ IL-33R+ ILC2s were gated using forward scattered light and side scattered light (f). Lineage− cells (g) were analyzed for CD25 and IL33-R expression. Lineage− CD25+ IL33-R+ cells were defined as another subset of ILC2s (h).
Flow Cytometric Analysis of ILCs in Peripheral Blood
First, we analyzed ILC subsets as previously described (Figure 1a–e, Table 2) [26]. We also analyzed another subset of ILC2s (lineage− IL33R+ CD25+ ILC2s; Figure 1f–g, Table 3) [27].
Fluorescein isothiocyanate (FITC)-labeled anti-human CD3, CD5, CD11b, CD11c, CD19, CD56, TCRαβ, TCRγδ, and FcεR1 antibodies (BD Biosciences, Franklin Lakes, NJ, USA); PE-labeled anti-human IL33-receptor antibody (Bioss Inc. Woburn, MA, USA); allophycocyanin (APC)- labeled anti-human CD25 antibody (Thermo Fisher Scientific, Waltham, MA, USA) and NKP44 antibody (BD Biosciences); phycoerythrin-cyanine 7 (PE-Cy7)-labeled anti-human CD127 antibody (BD Biosciences); peridinin chlorophyll protein complex-cyanine 5.5 (PerCP-Cy5.5)-labeled antihuman CD294 (CRTH2) antibody (BD Biosciences); Brilliant Violet 421-labeled anti-human CD117 (c-Kit) antibody (BD Biosciences); and APC/Cy7-labeled anti-human CD161 antibody (BioLegend, San Diego, CA, USA) were used.
Flow cytometry was performed according to a previously described method [16, 17, 19]. Heparinized peripheral blood was layered over an equal volume of Ficoll-Paque PLUS (GE Healthcare Biosciences AB, Björkgatan, Sweden) and centrifuged at 440 × g for 30 min at room temperature. The upper layer of plasma was removed, and the mononuclear layer was left at the interface. The mononuclear layer was transferred to a clean centrifuge tube and centrifuged at 860 × g for 10 min. The supernatant was gently removed, and the cells were resuspended in 10 mL of PBS. Then, the resuspended cells were again centrifuged at 860 × g for 10 min, and the supernatant was gently removed. The mononuclear cells were then suspended in the medium. The antibodies were added, followed by incubation for 20 min at room temperature away from light. Then, 1000 μL of PBS were added, the cells were centrifuged at 440 × g for 5 min, and the supernatant was again removed. Finally, the suspended mononuclear cells were added to 300 μL of PBS before conducting flow cytometry using FACS Canto II (BD Biosciences).
Statistical Analysis
The Kruskal–Wallis H test was used to analyze differences between multiple groups, and Spearman’s rank correlation coefficients were adopted to analyze the correlation between the two ILC2 types. P < 0.05 denoted statistical significance.
Flow Cytometric Analysis of Peripheral Blood
The Proportions of Total Lymphocytes in Peripheral Blood
There were no significant differences in the proportions of total lymphocytes among the HC (31.44% ± 7.90%), type 1 AIP (29.20% ± 5.68%), and CP groups (29.54% ± 7.066%; Figure 2).
Figure 2. The proportions of lymphocytes in peripheral blood.
The proportions of lymphocytes in peripheral blood from subjects in the healthy control (HC; 31.44% ± 7.90%), type 1 autoimmune pancreatitis (AIP; 29.20% ± 5.68%), and chronic pancreatitis (CP) groups (29.54 ± 7.066). There were no significant differences among the groups.
The Proportions of Total ILCs in Peripheral Blood
The proportions of total ILCs were higher in the type 1 AIP (0.287% ± 0.187%, P < 0.01) and CP groups (0.248% ± 0.111%, P < 0.05) than in the HC group (0.137% ± 0.087%). There was no significant difference between the type 1 AIP and CP groups (Figure 3).
Figure 3. The proportions of total innate lymphoid cells (ILCs) in peripheral blood.
The proportions of total ILCs in peripheral blood were higher in the type 1 autoimmune pancreatitis (AIP; 0.287% ± 0.187% P < 0.01) and chronic pancreatitis (CP) groups (0.248% ± 0.111% P < 0.05) than in the healthy control (HC) group (0.137% ± 0.087%). There was no significant difference in the proportions between the type 1 AIP and CP groups (P = 0.99). *P < 0.05, **P < 0.01, ***P < 0.001.
The Proportions of ILC1s in Peripheral Blood
The proportions of ILC1s did not differ among the HC (0.055% ± 0.066%), CP (0.078% ± 0.050%), and type 1 AIP groups (0.116% ± 0.125%; Figure 4a).
The proportions of ILC2s in Peripheral Blood
The proportions of ILC2s (lineage− CD127+ CD161+ c-Kit+ or c-Kit− CRTH2+ ILC2) was significantly higher in the type 1 AIP (0.140% ± 0.083%; P < 0.01) and CP groups (0.119% ± 0.055%, P < 0.05) than in the HC group (0.054% ± 0.039%). There was no significant difference in the values between the type 1 AIP and CP groups (P = 0.877; Figure 4b).
The Proportions of ILC3 in Peripheral Blood
The proportions of ILC3s in peripheral blood were significantly higher in the type 1 AIP (0.055% ± 0.043%, P < 0.01) and CP groups (0.051‰ ± 0.040‰, P < 0.05) than in the HC group (0.018% ± 0.017%; Figure 4c).
Figure 4. The proportions of various innate lymphoid cell (ILC) subsets in peripheral blood.
The proportions of lineage− CD127+ CD161+ c-Kit− NKp44− ILC1s in peripheral blood were not significantly different among the healthy control (HC; 0.055% ± 0.066%), chronic pancreatitis (CP; 0.078% ± 0.050%), and type 1 autoimmune pancreatitis (AIP) groups (0.116% ± 0.125%) (a). The proportions of lineage− CD127+ CD161+ c-Kit+ or c-Kit− CRTH2+ ILC2s in peripheral blood were higher in the type 1 AIP (0.140% ± 0.083%, P < 0.01) and CP groups (0.119% ± 0.055%, P < 0.05) than in the HC group (0.054% ± 0.039%). There was no significant difference in the proportions between the type 1 AIP and CP groups (P = 0.877) (b). The proportions of lineage− CD127+ CD161+ c-Kit+ NKp44− ILC3s in peripheral blood were higher in the type 1 AIP (0.055% ± 0.043%, P < 0.05) and CP groups (0.051% ± 0.040%, P < 0.05) than in the HC group (0.018% ± 0.017%). There was no significant difference between the type 1 AIP and CP groups (P = 0.962) (c). *P < 0.05, **P < 0.01, ***P < 0.001.
Analysis of a Second ILC2 Subset in Peripheral Blood
There was no significant difference in the proportions of lineage− IL33R+ CD25+ ILC2s among the HC (0.274% ± 0.223%), type 1 AIP (0.290% ± 0.29%), and CP groups (0.269% ± 0.143%; Figure 5).
Figure 5. Flow cytometric analysis of lineage− IL-33R+ CD25+ group 2 innate lymphoid cells (ILC2s).
There was no significant difference in the proportions among the healthy control (HC; 0.274% ± 0.223%), type 1 autoimmune pancreatitis (AIP; 0.290% ± 0.29%), and chronic pancreatitis (CP) groups (0.269% ± 0.143%).
There was also no correlation between the proportions of the two ILC2 subsets in the HC (r = 0.128, P = 0.724), type 1 AIP (r = −0.586, P = 0.127), or CP group (r = −0.121, P = 0.556) or in the entire study population (r = −0.147, P = 0.454; Figure 6).
Figure 6. Comparison of two subsets of group 2 innate lymphoid cells (ILC2; lineage− CD127+ CD161+ c-Kit+ or c-Kit− CRTH2+ ILC2s and lineage− IL33R+ CD25+ ILC2s).
There was no significant correlation between the proportions of the two subsets in the healthy control (HC; (r = 0.128, P = 0.724), type 1 acute pancreatitis (AIP; (r = −0.586, P = 0.127), and chronic pancreatitis (CP) groups (r = −0.121, P = 0.556) or in the entire study population (r = −0.147, P = 0.454).
In the present study, our data illustrated that the proportions of ILC2s and NKp44− ILC3s were increased in peripheral blood from patients with type 1 AIP and CP compared with the findings in HCs.
ILCs were identified by Moro et al. in 2010 [28], and they are similar to lymphocytes producing cytokines but lack lineage markers. ILCs can be classified into three groups (ILC1s, ILC2s, and ILC3s) according to the cytokines they produce and the transcription factors required for their development, and these types correspond to Th1, Th2, and Th17 cells, respectively [29, 30]. ILC1s produce INF-γ (Th1 cytokine) in response to IL-12 or IL-18 stimulation. ILC1s highly express T-bet for their development. ILC2s produce some Th2 cytokines such as IL-4, IL-5, and IL-13 through IL-25 or IL-33 stimulation. GATA3 is essential for ILC2 development and function. ILC3s depend on retinoidrelated orphan receptor γt (RORγt) for their development and function. The signature cytokine of ILC3s is IL-22 and IL-17 [31]. From previous studies using PBMCs from HCs, CD127+ ILCs comprise 0.01%–0.1% of circulating lymphocytes, and almost of them are ILC2s [26]. ILCs instantly produce cytokines through direct cytokine stimulation, and they have key roles in the innate immune system.
ILC2s play important roles in the pathophysiology of allergic diseases such as bronchial asthma and atopic dermatitis. In patients with asthma, it has been reported that the number of ILC2s in peripheral blood is elevated.32 In inflammatory conditions, such as atopic dermatitis, macrophages have been considered to develop into M1 macrophages to promote inflammation; however, in recent studies, IL-4–derived basophils were stated to participate in the recruitment of monocytes and their differentiation into M2 macrophages to suppress inflammation in allergic skin [22]. In addition, IL-4 derived from basophils enhances the function of ILC2s, including the production of cytokines and chemokines [23]. From our previous reports, M2 macrophages activated via TLR7 signaling might play an important role in pancreatic fibrosis in patients with type 1 AIP [18]. We detected basophils in the pancreatic tissue of patients with type 1 AIP. Moreover, the proportion of activated basophils, which are activated via TLR4 stimulation, among circulating basophils was significantly higher in patients with type 1 AIP in patients with CP and HCs [19]. In type 1 AIP, basophils may influence ILC2s and the differentiation from inflammatory monocytes into M2 macrophages in a similar manner as observed in bronchial asthma. In the present study, we analyzed two subsets of ILC2s (lineage− CD127+ CD161+ c-Kit+/− CRTH2+ and lineage− IL33R+ CD25+ cells) according to previous findings. Unexpectedly, the proportions of lineage− IL33R+ CD25+ ILC3s did not significantly differ among the HC, type 1 AIP, and CP groups. Moreover, there was no correlation between the abundance of these two ILC2 subsets. From these data, lineage− IL33R+ CD25+ cells may represent a distinct subset from lineage− CD127+ CD161+ c-Kit− or c-Kit+ CRTH2+ ILC2s.
In the present study, the proportions of ILC2s were significantly higher in the type 1 AIP and CP groups than in the HC group. According to recent reports, ILC2s are considered to be involved in and wound healing and the development of fibrotic diseases, including idiopathic pulmonary fibrosis (IPF), atopic dermatitis, asthma, and hepatic fibrosis [33, 34, 35, 36]. Thymic stromal lymphopoietin may also influence the development of several fibrotic diseases by activating ILC2s [37]. In terms of hepatic fibrosis, IL-33 produced by hepatocytes in response to chronic hepatocellular stress stimulates the accumulation and activation of ILC2s in the liver [36]. IL-13 produced by activated ILC2s activates STAT6 and SMAD family transcription factors in rat hepatic stellate cells through TGF-β signaling [36]. In lung fibrosis, M2 macrophages are associated with fibrosis via the induction of ILC2s [34]. M2 macrophages and ILC2s play important roles in fibrotic changes in various organs [33].
Recent studies suggested that patients with type 1 AIP usually develop CP [13, 14]. In type 1 AIP, it has been reported that profibrotic cytokines (such as TGF-β and platelet-derived growth factor (PDGF)) and their receptors are expressed in macrophages, myofibroblasts, and epithelial cells [38]. The basic processes of fibrotic change are common among types of CP including alcoholic and idiopathic pancreatitis despite their differing etiologies [39, 40, 41]. Pancreatic injury leads to cell necrosis and/or apoptosis and the subsequent release of cytokines such as TGF-β and PDGF, in addition to chemokines, from immigrating inflammatory cells, macrophages, and epithelial or mesenchymal cells. Moreover, macrophages phagocytose damaged cells and release several cytokines that activate pancreatic acinar cells. This cascade causes fibrogenesis in the pancreas through the deposition of extracellular matrix [42, 43]. ILC2s may contribute to chronic inflammatory conditions such as pancreatic fibrosis in type 1 AIP and CP.
ILC3s represent the most heterogeneous subset of ILCs, and they are defined by the expression of RORγt as well as the production of IL-17, IL-22, granulocyte-macrophage colony-stimulating factor, and/or tumor necrosis factor-α in response to IL-23, IL-1β, and aryl hydrocarbon receptor ligands [44].44 It is well known that ILC3s are involved in the development of inflammatory bowel diseases [45, 46, 47] and psoriasis [48, 49].
IL-17 has been implicated in the generation of fibrosis, as its levels are elevated in patients with IPF and cystic fibrosis [50, 51]. Furthermore, IL-17A has a critical role in the generation of bleomycin-induced pulmonary fibrosis, which is dependent on TGF-β, suggesting codependent roles for IL-17A and TGF-β in the pathogenesis of fibrosis [52]. IL-17 is consider a key player in the development of lung fibrosis. ILC3 counts are elevated in animal models of asthma and in the bronchoalveolar lavage fluid of patients with asthma [53]. ILC3s have been implicated in the development of IL-17–mediated fibrosis [54] because they represent an important source of IL-17. From these reports and our findings, both ILC2s and ILC3s may be involved in the fibrotic changes observed in patients with type 1 AIP and CP.
ILC2s and ILC3s may be involved in the pathophysiology of chronic inflammatory conditions of the pancreas. However, further studies are necessary to clarify their precise roles in the pathogenesis of type 1 AIP and CP.
This study was partially supported by (1) Grant-in- Aid for Scientific Research (C) of the Ministry of Culture and Science of Japan (18K07921, 17K09468, 17877850), (2) the Research Program on Intractable Diseases, from the Ministry of Labor and Welfare of Japan, (3) MHLW Research Program on Rare and Intractable Diseases Grant Number JPMH20FC104, (4) the Research Program from the Japan Medical Research and Development (AMED) (17824893), and (5) Grants-in-Aid from the Ministry of Education, Culture, Sports, Science and Technology of Japan from the CREST Japan Science and Technology Agency. We warmly thank Dr. Hiroyuki Gonda for technical assistance for flow cytometry analysis.
The authors had no conflicts of interest to declare in relation to this article.