- (2014) Volume 15, Issue 2
Andre Luiz De Souza, Muhammad Wasif Saif
Department of Hematology and Oncology, Tufts Medical Center. Boston, MA, USA
Adenosquamous carcinoma of the pancreas is a rare type of pancreatic cancer. Although its molecular biology profile hasbeen shown to be similar to pancreatic ductal adenocarcinoma tumors, it has different prognostic features. There is noconsensus or guidelines to treat this tumor differently from pancreatic adenocarcinoma, but therapies based on gemcitabineand platinum chemotherapeutics such as cisplatin and oxaliplatin have been used based on results of a few case reports. Wediscuss the Abstract #269 from the 2014 ASCO Gastrointestinal Cancers Symposium showing better outcomes fromplatinum-based therapy in this type of tumors. Triggered by this study, we also present our experience. Prospective studiesto investigate the clinical outcomes from platinum-based therapy and the role of target therapies such as erlotinib arewarranted.
Carcinoma, Adenosquamous; Pancreas; Platinum Compounds
5-FU: 5-fluorouracil; AE1: anion exchanger 1; CAM 5.2: anti-keratin antibody detected in carcinomas; CEA: carcinoembryonic antigen; CK: cytokeratin; DFS: disease free survival; DPC4/MAD4: deleted in pancreatic cancer 4 tumor suppressor gene/mothers against decapentaplegic 4 protein, transcription repressor complex component; FOLFIRINOX: 5- fluorouracil, leucovorin, irinotecan and oxaliplatin; K-ras: kirsten rat sarcoma oncogene; NCCN: National Comprehensive Cancer Network; p16/CDKN2A: protein 16/cyclin D-kinase N2A, involved in cell cycle arrest; PR: partial response; S1: tegafur/uracil; SD: stable disease; TP53: gene of the tumor suppressor 53, involved in cell cycle arrest
Adenosquamous carcinoma represents 1-4% of all pancreatic carcinomas [1]. The disease distribution shows an approximately 1:1 male/female ratio [2]. They have a worse prognosis and higher potential for metastasis. After resection, median overall survival is 6.8 months, with a 3-year overall survival of 14.0% [3]. Following resection, patients with adenosquamous carcinoma have worse 2-year survival (29% versus 36%; P<0.0001) compared with adenocarcinoma of the pancreas [4]. Patients with adenosquamous carcinoma of the pancreas are slightly older, smoke less, and have more involvement of the body and tail than the head of the pancreas (44.6% versus 53.5%; P<0.0001) [4]. Adenosquamous carcinomas are more likely to be poorly differentiated (71% versus 45%; P<0.0001), node positive (53% versus 47%; P<0.0001), and larger in size (5.7 versus 4.3 cm; P<0.0001) [4]. They harbor the same molecular signatures as pancreatic adenocarcinoma, such as gene of the tumor suppressor 53, involved in cell cycle arrest (TP53), protein 16/cyclin D-kinase N2A, involved in cell cycle arrest (p16/CDKN2A), deleted in pancreatic cancer 4 tumor suppressor gene/mothers against decapentaplegic 4 protein, transcription repressor complex component (DPC4/MAD4) and kirsten rat sarcoma oncogene (K-ras) mutations. Also, p16 mutations suggest the squamous component of the tumor, even when cytology is not clear [5].
In order to be diagnostic, the squamous component of adenosquamous carcinoma of the pancreas should represent at least 30% of the tumor. In a clinicopathological series of 25 patients with adenosquamous carcinoma of the pancreas, all cases tested were immunoreactive with keratin (anion exchanger 1:3; AE1:AE3 and cytokeratin 1; CK1), whereas other keratin markers were variably expressed: CK5/6 (88%), CK7 (68%), anti-keratin antibody detected in carcinomas (CAM 5.2; 41%), and CK20 (26%). CA 19-9 (84%) and carcinoembryonic antigen (CEA; 74%) were positive in the majority of the cases. K-ras oncogene mutations were identified in seven of thirteen cases. In that particular series, all patients died from their disease an average of 5.8 months after diagnosis (range: 1 to 33 months) [6].
Multimodality therapy involving aggressive surgery, intraoperative radiation therapy, and locoregional chemotherapy have been improving the survival of patients with adenosquamous carcinoma of the pancreas resulting in published survivals of up to 40 months [7].
Shibagaki et al. presented a case of unresectable pancreatic adenosquamous carcinoma treated with systemic chemoradiotherapy with low-dose cisplatin and 5-fluorouracil (5-FU) achieving partial response and followed by combined chemotherapy with S-1 and cisplatin resulting in complete response at 20 months [8]. Tanaka et al. published a case report of adenosquamous carcinoma of the pancreas with survival of 12 months after interferon-alpha plus tumor necrosis factor-alpha neoadjuvant combined with 5-FU chemotherapy. An initially unresectable tumor would be operated resulting in a 5-month progression free survival [9]. Wilkowski et al. studied the efficacy and toxicity of concurrent chemoradiation with gemcitabine and cisplatin in 47 patients with locally advanced, unresectable pancreatic carcinomas. This treatment improved overall survival to 10.7 months in patients whose tumors were rendered completely resectable, with median progression free survival of 7.8 months [10].
Efficacy of Platinum Chemotherapy Agents in the Adjuvant Setting for Adenosquamous Carcinoma of the Pancreas. (Abstract #269 [11])
Wild et al. presented a retrospective study with 48 confirmed cases of adenosquamous carcinoma of the pancreas who underwent platinum-based adjuvant chemotherapy [11]. There was a better overall survival for patients with negative lymph nodes and resection margins after surgery and for those who received platinum-based chemotherapy. The group concludes that platinum-based chemotherapy should be used as first-line chemotherapy for adenosquamous carcinoma of the pancreas. However, the last update from the National Comprehensive Cancer Network (NCCN) guidelines considers gemcitabine-based combination chemotherapy as first line category 1 of evidence for treatment of pancreatic ductal adenocarcinoma (when combined with either erlotinib or nab-paclitaxel) as well as 5-fluorouracil, leucovorin, irinotecan and oxaliplatin (FOLFIRINOX). How many of the patients in the study received one of these category 1 evidence regimens would impact in the selection of first line combination chemotherapy as the cases were treated from 1986 to 2012 (i.e., some of them years before the new regimens were consolidated); thus, causing bias in this retrospective study. Gemcitabine-based regimens are already underrepresented as they were not given all along as part of the gemcitabine erlotinib or gemcitabine nabpaclitaxel regimens. This is particularly important in view that the molecular biology of adenosquamous and its K-ras status is similar to pancreatic ductal adenocarcinoma and the impact that EGFR inhibitors, such as erlotinib, would cause in wild K-ras tumors. Also, the absence of reference of radiation therapy as part of the adjuvant therapy or the impact of staging in the multivariate regression model limit the conclusion of the study. Still, case reports with impressive results substantiate the use of combinations such as gemcitabine and cisplatin and gemcitabine and oxaliplatin in these tumors. Nevertheless, the above mentioned presentation suggests that a platinumcontaining regimen such as FOLFIRINOX could have a role as first line treatment of adenosquamous carcinoma of the pancreas, although the role of fluoropyrimidines, such as 5-FU and capecitabine, cannot be underestimated in this group of patients.
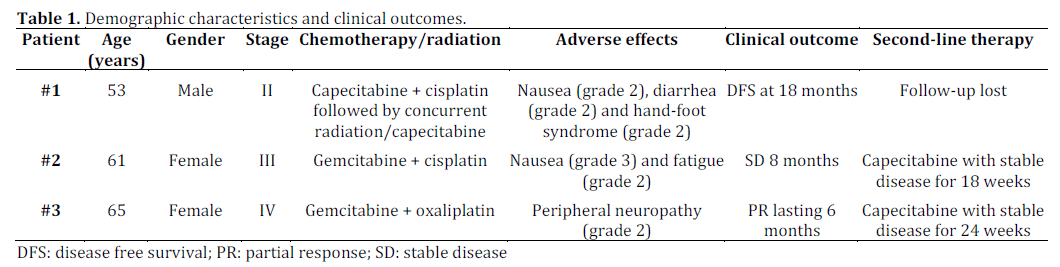
We reviewed the medical records of patients with adenosquamous cell carcinoma of the pancreas who were treated by our multidisciplinary group. Table 1 summarizes the patients’ characteristics. Three patients (1 male and 2 females) with adenosquamous cell carcinoma of the pancreas were identified. The median age of the patient population was 61 years-old (range: 53-65 years- old). All patients had histology confirmed adenosquamous cell carcinoma of the pancreas. These patients received a variety of regimens consisting of platinum as well fluoropyrimidines as shown in Table 1. Our data showed that these agents are beneficial to treat patients with adenocarcinoma of the pancreas. The toxicities were consistent with their known adverse effect profile.
Adenosquamous carcinoma of the pancreas is a rare type of pancreatic tumor that has a molecular biology similar to pancreatic ductal adenocarcinoma. Data from the 2014 ASCO Gastrointestinal Cancers Symposium, as well as our experience, showed better outcomes from platinum-based therapy in this type of tumor. As a retrospective study, there are limitations to conclude that cisplatin-based therapy is the most optimal therapy. Further studies after identifying this subtype are needed.
The authors have no potential conflict of interest