- (2015) Volume 16, Issue 3
Shabnam Shahrokh, Mohammad Bagher Miri, Mohammad Taghi Safari,Amir Houshang Mohammad Alizadeh
Research Center for Gastroenterology and Liver Diseases, Shahid Beheshti University of Medical Sciences, Taleghani Hospital, Tehran, Iran
Received February 24th, 2015- Accepted March 28th, 2015
Pancreatic tuberculosis is a rare occurrence in either immune-competent or immune-suppressed host. Pancreatic tuberculosis most commonly afflicts the region of the head and the uncinate process of the pancreas. It is often misdiagnosed due to low index of suspicion and masquerading of its symptoms as more common pancreatic conditions such as pancreatic malignancy. However, Endoscopic ultrasound fine needle aspiration sampling with an acid-fast smear is essential for establishing the diagnosis of pancreatic tuberculosis. If the diagnosis is delayed, pancreatic tuberculosis can be fatal. Pancreatic tuberculosis responds well to standard anti-tuberculous drugs.
Pancreas
Tuberculosis (TB) is a serious health problem worldwide [1]. It is a multi-systemic bacterial infection caused by different strains of mycobacteria, usually Mycobacterium tuberculosis. It occurs in nearly 9.7 million people [2, 3], and claims about 2 million lives each year worldwide [4] highest incidence being in Asia, South America, Eastern Europe, and most sub-Saharan African countries [2, 3].
Although the incidence of TB was low in developed countries, it has risen in recent years due to an increase in emigration from tuberculosis- endemic areas, the emergence of human immunodeficiency virus (HIV) infection [5], and widespread use of immunosuppressant drugs [6, 7]. For example Australia, Western Pacific and South East Asia have incidence rates of 5-6, 108, 181 per population, respectively [8, 9] and United States Center for Disease Control and Prevention (CDC) reported approximately 11200 new cases of TB in 2014 [10].
Although TB usually involves lungs, extra-pulmonary TB (EPTB) accounts for nearly 10-30% (approximately 15- 20%) of all cases of TB in immunocompetent hosts [3, 11, 12]. About 12.5% of all TB cases have EPTB [13] who nearly 50 percent of them are human immunodeficiency virus (HIV)-positive [14, 15].
By definition, EPTB is when TB occurs at sites other than the lung. It can occur in almost any organ system; most commonly in the lymph nodes, pleura, genitourinary system, and bone [14, 15]. Note that EPTB must not be confused with miliary TB, as miliary refers to EPTB with pulmonary involvement, and not EPTB in isolation [16].
Abdominal TB is a common site for EPTB [15]. It accounts for 5-12% of patients with tuberculosis (with the highest prevalence in developing countries) [17, 18] and almost 11-16% of patients with EPTB have abdominal involvement [13]. Abdominal TB includes infection of different combinations of gastrointestinal tract (especially ileocecal region), lymph nodes, peritoneum, and intraabdominal organs such as the spleen, liver, and pancreas [15, 19-29].
Pancreatic tuberculosis, either with or without peripancreatic lymphadenitis, is a rare occurrence in either immunocompetent or immunosuppressed host. It was first reported by Auerbach in 1944. In his series of 1656 autopsies of tuberculous patients, only 14 cases had pancreatic involvement that may have mimicked neoplasia but he did not find any cases of isolated pancreatic tuberculosis [28]. Pancreatic tuberculosis usually occurs in patients with miliary tuberculosis with pulmonary and extra-pulmonary involvement including pancreas, or in isolated EPTB cases without pulmonary involvement especially in acquired immunodeficiency syndrome (0.46%) [13, 14, 21, 23, 27, 28, 30, 31]. But even HIVinfected patients have an incidence of 0.46% [32, 33]. Autopsy studies have shown that the pancreas is involved in 2.1%–4.7% of patients with miliary tuberculosis [28, 34-38]. However, in a study from 1999-2004 from India detected pancreatic TB in 8.3% of the 384 patients who were diagnosed with abdominal TB [39].
As mentioned above, pancreatic tuberculosis is occasionally observed in people with other organ's involvement as a consequence of miliary TB or EPTB with involvement of other organs in addition to pancreas [28, 34]. But primary pancreatic tuberculosis (PPTB) is described as an isolated involvement of pancreas by mycobacterium tuberculosis in the absence of involvement of any other organ or previously identified TB [36]. PPTB is particularly rare [37, 38]; fewer than 100 cases have been reported worldwide [39] and its incidence is estimated to be less than 4.7% [28, 31]. Bhansali in a series of 300 miliary tuberculosis patients over 12 years in India, did not reveal even 1 with pancreatic involvement [2, 10, 15, 21].
Isolated pancreatic TB is predominantly observed in the following patient types:
• Patients who reside in endemic tuberculose zones,
• Patients in areas of widespread TB dissemination such as a military setting and developing countries [19, 20, 25, 27],
• Patients who are immuno-compromised [40]
However pancreatic TB has also been reported with increased frequency from western world [41] and the reported number of such cases in immunocompetent patients has increased over the past decade [31]. This increasing incidence is probably due to globalization, increased use of immunosuppressants, the worldwide resurrection of M. tuberculosis and the HIV pandemic [31, 34, 42, 43].
A recent review reported that 23% of the 62 cases of pancreatic TB occurred in patients who were HIV infected [44]. In AIDS cases, tuberculous pancreatic abscesses are most common, accounting for 70.0% of cases. In addition, 71.1% of cases have no previous serological evidence of HIV infection, and 76.2% of patients are severely immunocompromised hosts with a CD4 cell count of ≤190/ mm3 [5, 29, 45].
More than half of patients with pancreas tuberculosis in the world literature are young adults (<30 years old) [39, 46]. Convincing epidemiological data with regards to sex are conflicting, suggesting that pancreatic TB is more common in men [27, 39].
Pancreatic TB most commonly afflicts the region of the head and the uncinate process of the pancreas [47, 48]. It is often misdiagnosed due to low index of suspicion and masquerading of its symptoms as more common pancreatic conditions such as pancreatic malignancy [36, 47, 49]. If the diagnosis is delayed, pancreatic TB can be fatal; it has a 10.8% mortality rate (comparing to the mortality rate of 9.1% in immunocompetent patients) [50]. However, pancreatic TB responds well to standard antituberculous drugs (ATDs).
Pathogenesis
Because of its rarity, the pathogenesis of pancreatic/ peri-pancreatic involvement of TB is not known with certainty and it is not yet clear how the infection can only affect the pancreas [51, 52]. It is believed that this low frequency is due to retroperitoneal location of pancreas as well as pancreatic enzymes including lipases and deoxyribonucleases that interfere with the colonization, seeding and proliferation of the bacteria [4, 38, 53, 54].
Pancreatic secretions also showed an antitubercular effect in vitro [51, 52], thus a large intrapancreatic inoculum of Mycobacterium tuberculosis is required to cause pancreatic lesions [53, 55].
Although the primary site of TB is usually not evident in most cases of hepatobiliary and pancreatic TB [56], several possible mechanisms for pancreatic involvement of tuberculosis have been discussed. Ingestion of infected material from an active pulmonary lesion is one. Following ingestion the bacilli gain access to the gastrointestinal tract where necrotizing granulomas may develop and then spread to the lymphatics affecting any organ in the GI tract, including hepatobiliary and pancreatic tissue [57]. Other mechanisms include lymphohematogenous dissemination from pulmonary disease, reactivation of latent tuberculosis in the pancreatic focus and toxic-allergic reaction of the pancreas involving an inflammatory response to generalized tuberculosis [19, 28, 37, 56, 58, 59].
Clinical Features
Over the last two years, 39 patients with pancreatic tuberculosis were reported in the literature. Of these patients, 26 were males and age ranged from 13 to 58 years. The outstanding signs and symptoms of pancreatic tuberculosis are summarized as Table 1. Most common location of pancreatic tuberculosis as a mass has been reported in the head (29-74.4%) followed by body (7- 17.9%), uncinate (3-7.7%), and tail (2-5.1%). Abdominal pain is the most common (31-79.5%) symptom followed by fever (20-51.3%), weight loss (19-48.7%), appetite loss (11-28.2%), and jaundice (8-20.5%) that were obstructive in nature.
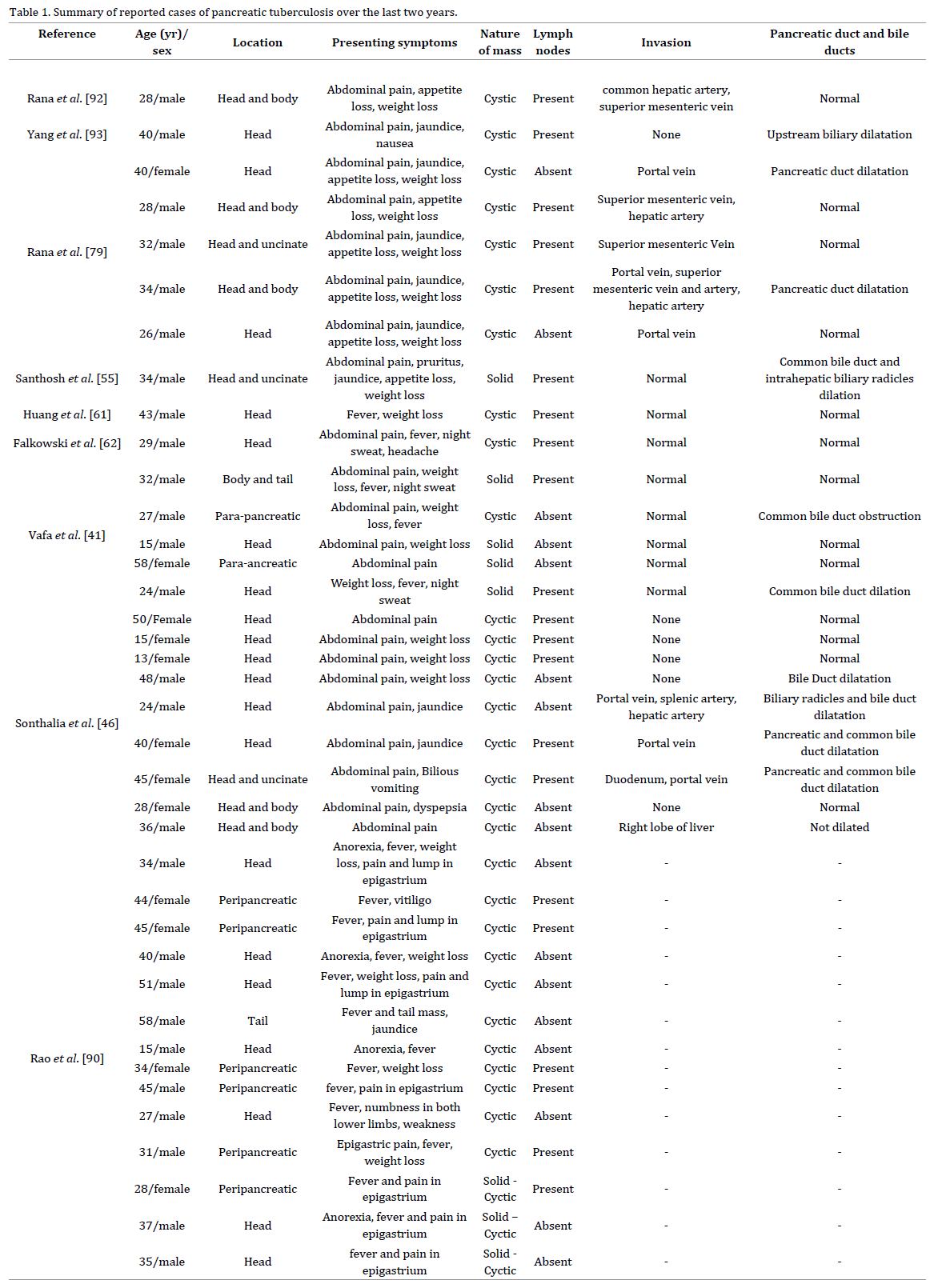
Three patients had the past history of pulmonary tuberculosis and lymphadenopathy was present in 22 patients. Pancreatic tuberculosis leaded to local vascular invasion in 10 patients also causing diagnostic confusion with locally advanced or metastatic pancreatic malignancy. Portal vein, superior mesenteric vein and hepatic artery involvement were reported in 6 (15.4%), 4 (10.3%), and 4 (10.3%) patients, respectively.
Diagnosis
Diagnosing pancreatic TB is challenging due to its nonspecific clinical, laboratory or radiological features. Since pancreatic TB may present as cystic or solid pancreatic masses, abscesses, lymphomas, pseudocysts or acute or chronic pancreatitis, cytological or histopathological as well as bacteriological confirmation is necessary for the diagnosis of pancreatic tuberculosis.
Pancreatic tuberculosis is a potential mimic of pancreatic malignancy. Although local vascular invasion is often considered to be an imaging feature of malignant lesions, the presence of vascular invasion cannot exclude possibility of pancreatic tuberculosis [60]. Also, the dilation of the pancreatic main or branch duct does not distinguish one condition from the other. However, there was other evidence against pancreatic malignancy such as fever (unless pyogenic cholangitis develops due to biliary duct compression) and the normal values of tumor markers [61]. In patients without typically tuberculosis manifestations (such as fever, night sweats, weight loss or cough), a range of cystic pancreatic lesions, including common lesions (pseudocysts, serous or mucinous cystadenomas, intraductal papillary mucinous neoplasm) and rare lesions such as solid pseudopapillary tumors pancreatic epithelial cysts, may need to be considered [62].
Pancreatic TB can be classified radiologically into 3 groups: mass-forming (with or without diffuse pancreatic enlargement), a diffuse form and a small, nodular form. The mass-forming is the most common form that accounts for 94.4% of cases [63].
Several imaging methods like CT scan, MRI, transcutaneous ultra-sound and endoscopic ultrasound are used to assess pancreatic pathology. Ultra sound (US) scan is noninvasive, simple, readily available and cost-effective; thus, it is usually used as an initial diagnostic tool. CT scan is used to rule out associated pathologies and to plan for disease management. It is the investigation of choice for pathologies of pancreas because of its high sensitivity. Although imaging findings may suggest the possibility of tuberculosis, none of them are neither specific nor pathognomonic for pancreatic TB [64]. They can reveal diffuse enlargement of the pancreas [64-66], focal hypoechoic mass on ultrasound or cystic lesions [67-73] (mostly in the head and uncinate process), heterogeneously hypo-isoechoic or isodense lesions on ultrasound and CT Scan, respectively, (primarily in the head) [39, 64, 65, 67, 69, 74] and enlarged peripancreatic [67, 70, 75] and other abdominal LNs [45, 64, 69, 70, 76, 77].
In pancreatic TB common bile duct and the pancreatic duct appear normal in images, even if the mass is localized centrally in the head of the pancreas. Contrary to pancreatic TB, in pancreatic adenocarcinoma the pancreatic duct is dilated in the tumors that are positioned centrally in the head region. Pancreatic enlargement with narrowing of the main pancreatic duct and heterogeneous enhancement is the characteristic of the diffuse form of pancreatic tuberculosis [78]. Bile cytology on endoscopic retrograde cholangiopancreatography (ERCP) may help in establishing the diagnosis [10, 56].
However, EUS-FNA sampling is essential for establishing the diagnosis of pancreatic tuberculosis. The samples are evaluated by staining, cytology, bacteriology, culture and polymerase chain reaction assay [79-86]. A definitive decision is usually made based on a histopathological or microbiological examination of a specimen that is obtained from the pancreas or based on peripancreatic LNs. In a study, Song et al. [87] were able to diagnose pancreatic/ peri-pancreatic tuberculosis in 76.2% of patients using EUS-FNA. The microscopic features of tuberculosis are granuloma, caseation necrosis (seen in 75%-100% of cases) and presence of acid fast bacilli (identified in 20%-40% of cases) [84]. EUS is a reliable technique for differentiating pancreatic lesion from peripheral structures [87]. It is also preferred for tissue biopsy because of less chances of needle tract dissemination especially when the mass seems malignant. Rana et al. found that the majority of patients with pancreatic tuberculosis (83.3%) had granulomas with culture for Mycobacterium tuberculosis being positive in 1 of 2 patients (50.0%) tested and acid fast bacilli being seen in only 1 of 6 (16.7%) [79]. In addition, acid fast bacilli smear using auramine or Ziehl-Neelsen staining can also be employed for this purpose.
As a result, and given the simplicity of the test and the rapid results that it produces, EUS-FNA with an acid-fast smear should be required for pancreatic TB diagnosis. A PCR assay yields highly specific same-day results, when used to detect mycobacterial DNA. Although its sensitivity to TB in FNA specimens has not yet been established, the PCR assay is increasingly used adjunctive to special staining techniques and mycobacterial cultures. It may show positive results even when specimen cultures are negative [77].
Therefore, diagnosis is a challenge, calling for a team approach with the goal of making the diagnosis noninvasively.
Treatment
Most cases of pancreatic TB respond well to ATD [32, 71, 88-90]. Therefore invasive treatments such as surgery and the drainage of fluid are not necessary most of the times [32]. Directly observed therapy with a standard multiple ATD regimen including isoniazid, rifampicin, pyrazinamide, and ethambutol or streptomycin for 6-12 months, is usually effective [74]. However for those patients whose tuberculous pancreatic mass is enlarged and causes symptoms even after ATDs therapy for a reasonable period of time, we should consider minimally invasive procedures [32] such as endoscopic internal drainage, percutaneous catheter drainage or biliary stenting. Percutaneous catheter drainage is preferably used in patients with no pancreatic duct strictures and pancreatic duct-pseudocyst communications and in those with immature or infected pseudocysts, or when the patient is at high surgical risk, or exhibits malnourishment [91, 92]. Also ATDs have hepatotoxic effects, and malnourished patients with jaundice are at higher risk. Therefore, patients with underlying liver disease might require modification of ATDs [79].
The authors had no conflicts of interest