- (2002) Volume 3, Issue 5
Raffaele Pezzilli1, Antonio Maria Morselli-Labate1, Elisabetta Romboli1, Francesco Dibenedetti2, Margherita Massa2, Marina Migliori1, Bahjat Barakat3, Giampaolo Merlini2, Roberto Corinaldesi1, Gian Vico Melzi d’Eril4
1Dipartimento di Medicina Interna e Gastroenterologia, Ospedale Sant’Orsola - Università di Bologna, Bologna. Italy
2Area di Biotecnologie e Tecnologie Biomediche, Clinica Pediatrica, Laboratori di Biotecnologie, IRCCS Policlinico San Matteo, Dipartimento di Biochimica, Università di Pavia. Italy
3Dipartimento di Emergenza, Ospedale Sant’Orsola, Bologna. Italy
4Chimica Clinica, Università dell’Insubria, Varese. Italy
Received: 22 July 2002 Accepted: 23 August 2002
Background There is a lack of data concerning pancreatic involvement during shock. Aim To evaluate possible pancreatic alterations in the early phase of shock. Setting Twelve consecutive patients with shock were studied within 2 hours from the onset of illness. Seven patients died during the hospital stay: 3 within 4 hours from admission, 3 within 4-8 hours and 1 within 12 hours. Main outcome measures Amylase, lipase, C- reactive protein, amyloid A, interleukin 6 procalcitonin and vascular cell adhesion molecule-1 serum concentrations were determined on admission and 4, 8, and 12 hours afterward. All patients underwent imaging studies of the pancreas. Results None of the patients developed clinical signs or morphological alterations compatible with acute pancreatitis. Serum amylase levels were above the upper reference limit in 7 patients (58.3%) and serum lipase levels in 2 patients (16.7%; P=0.062). There were no significant differences found between survivors and nonsurvivors in the serum concentrations of all the proteins studied. Conclusions In patients with shock, amylase seems to be more frequently elevated than lipase. None of the patients showed pancreatic alterations at imaging techniques.
Acute-Phase Proteins; Amylases; Clinical Medicine; Lipase; Pancreatitis; Shock
PCT: procalcitonin; SAA: serum amyloid A; VCAM-1: vascular cell adhesion molecule-1
It has been proposed that ischemic injury to the pancreas occurs in specific clinical settings, such as cardiopulmonary by-pass [1], surgery for thoraco-abdominal aneurysms [2], hemorrhagic shock [3], and transplantation of the pancreatic gland [4]. Experimental studies have shown that ischemia may induce acute pancreatitis [5] or precipitating acute pancreatitis [6]. However, there is a lack of data on the involvement of the pancreas in human shock. Furthermore, in shock immuneactivated T cells, IL-1 and TNF initiate the cascade of inflammatory mediators by targeting the endothelium [7].
The aim of this study was to investigate the possibility of pancreatic involvement in patients in shock by using serum pancreatic enzyme determinations and imaging techniques. The presence of immuno-response was detected by determining in serum specific inflammatory mediators such as interleukin-6 (IL-6), C- reactive protein (CRP), amyloid A (SAA) and procalcitonin (PCT). Vascular damage was also evaluated by determining circulating vascular cell adhesion molecule-1 (VCAM-1).
The study was carried out in the Emergency Department of Sant’Orsola Hospital, Bologna, Italy. The criteria for the presence of shock were a change in blood pressure consisting of at least one of the following criteria: mean arterial pressure less than 60 mmHg, systolic pressure less than 90 mmHg, an absolute decrease in systolic blood pressure of 40 mmHg or the use of adrenergic agents to maintain a mean arterial pressure greater than 60 mmHg, and at least one of the following other signs: oliguria (0.3 mL/kg/h for two consecutive hours), a change in the level of consciousness, serum lactate greater than 2.5 mmol/L, or an absolute mixed oxygen saturation of less than 60% [8]. From April 1998 to April 1999, 12 consecutive patients (3 M, 9 F; mean age 79.4 years, range 67-87 years) met these criteria and were enrolled in the study. In all of them, the elapsed time between the appearance of the shock and the study entry was less than or equal to 2 hours. All patients were treated according to current therapeutic modalities [9]. The final diagnoses are reported in Table 1 and associated co-morbidities are reported in Table 2.


Seven patients died during the hospital stay: 3 within 4 hours from admission, 3 within 4-8 hours from admission and 1 within 12 hours.
Serum samples were obtained upon admission to the hospital and 4, 8, and 12 hours afterward; they were kept frozen at –20 °C until analysis.
Serum concentrations of the following proteins were determined using commercially available kits: amylase (Alpha-Amylase, Sentinel Ch. Milan, Italy. Reference values <220 U/L), lipase (Lipase KC Sera-Pack, Bayer. Milan, Italy. Reference values <58 U/L), IL-6 (IL-6 Elisa Kit, R&D Systems. Minneapolis, MN, USA. Detection limit 3.13 ng/L), CRP (CRP, Dade Behring. Marburg, Germany. Reference values <5 mg/L), SAA (SAA, Dade Behring. Marburg, Germany. Reference values <6.4 mg/L), VCAM-1 (human sVCAM-1 ELISA Bender MedSystems, Bender MedSystems Diagnostica GmbH. Wien, Austria. Reference values <1693 ng/mL) and procalcitonin (LUMItest PCT, Brahms Diagnostica GmbH. Berlin, Germany. Reference values <0.5 ng/mL).
All patients also underwent, within 12 hours from admission, ultrasonography and/or computed tomography to detect pancreatic abnormalities.
The study was approved by the local institutional review board with a priori patient or appropriate proxy consent obtained prior to the participants’ entry into the study. The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as revised in 1983.
Data are reported as mean values ± standard deviations. Statistical analyses were carried out by means of the Mann-Whitney U-test, the matched-pairs Wilcoxon test, the McNemar test, and the Spearman rank correlation. The statistical analyses were performed by running the SPSS/PC+ statistical package (SPSS Inc., Chicago, IL, USA) on a personal computer. Two-tailed P values less than 0.05 were considered statistically significant.
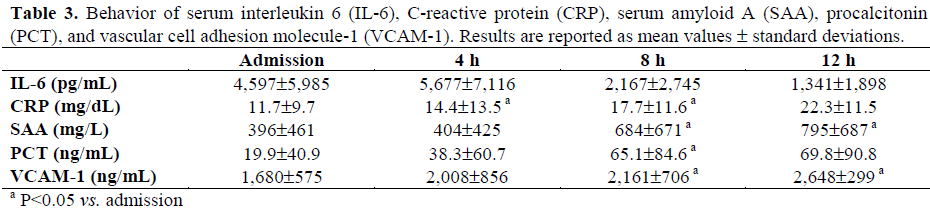
The behavior of the serum concentrations of amylase and lipase is reported in Figure 1. Serum amylase concentrations were distributed around the upper reference limit during the entire study period whereas mean serum lipase concentrations were consistently normal. Serum amylase and lipase concentrations at 4, 8, and 12 hours were not significantly different when compared to the admission values. The values of the other proteins studied during the study period are reported in Table 3. IL-6 peaked at 4 hours from hospital admission, whereas CRP, SAA, PCT, and VCAM-1 progressively increased in the 12 hours following admission. A statistically significant increase as compared to the admission values was found for CRP at 4 and 8 hours, for PCT at 8 hours, and for both SAA and VCAM-1 at 8 and 12 hours. There were no significant differences found between survivors and non-survivors in the serum concentrations of all the proteins studied (data not shown).
During the study period, 7 patients (58.3%) had at least one serum amylase determination above the upper reference limit whereas only 2 (16.7%) had an increase of serum lipase concentrations (P=0.062). None of the patients developed clinical signs or morphological alterations compatible with acute pancreatitis.
Pooling all data obtained at the different time intervals, no significant relationship was found between serum amylase and lipase concentrations (P=0.425).
Ischemia followed by reperfusion results in a breakdown of the microcirculation in the pancreas as in other organs and it is considered to be a critical factor in the pathogenesis of acute pancreatitis [10, 11]. Microvascular perfusion failure is a characteristic hallmark of pancreatitis [11, 12, 13]. Since the first report of Panum [14], other researchers have shown that the pancreas is highly sensitive to ischemia [6, 15, 16, 17, 18]. In humans, development of acute pancreatitis has been reported after embolic closure of the vessels supplying the pancreas [19], after hypoperfusion of the pancreas during cardiac surgery [1, 3] and surgery for thoraco-abdominal aneurysms [2], after splanchnic hypoperfusion [20] and after pancreatic transplantation [21]. However, there is a lack of data on the involvement of the pancreas in human shock. In this study involving 12 patients, we found that during the 12 hours after admission serum amylase concentrations remained elevated, whereas serum lipase concentrations remained belove the upper normal limit, thus suggesting that hyperamylasemia is of non-pancreatic origin. Considering the values above the normal range, 58% of the patients had an increase in serum amylase concentrations whereas only 17% of them had an increase in serum lipase levels. These data suggest that even if 17% of the patients had an alteration of serum lipase, none of them developed clinical signs or morphological alterations compatible with acute pancreatitis. Our data are different from those reported by Hegewald et al. [22], Gmaz-Nikulin et al. [20] and Warshaw and O'Hara [3]. However, in the studies of Hegewald et al. [22] and Gmaz-Nikulin et al. [20], pancreatic alterations were detected by electron microscopy and by histology whereas, in our study, imaging techniques, such as ultrasonography and computed tomography, were utilized to evaluate alterations of the pancreatic gland. The difference between our study and that of Warshaw and O’Hara [3] may be due to different selection criteria. In their study, these authors found that 69% of patients had hyperamylasemia of pancreatic origin but developed this condition over several days following the shock.
Regarding the acute phase proteins, the results of our study agree with those previously published [23, 24]. The early stage of shock is characterized by acute phase protein upregulation, leading to the activation of both immune effector cells and vascular endothelium. The IL-6 increase precedes the other acute phase proteins, thus confirming previous observations also in conditions of shock [25, 26, 27]. No significant difference in all the immunological parameters studied was observed when comparing survivors and non-survivors; these data agree with the previous observations of Stoiser et al. [23].
In conclusion, in patients in shock, serum amylase is more frequently elevated than serum lipase and it is of non-pancreatic origin. None of the patients showed pancreatic injury at imaging techniques.