- (2014) Volume 15, Issue 2
Daniel Gorovets, Muhammad Wasif Saif, Kathryn Huber
Tufts Medical Center. Boston, MA, USA
Despite decades of research, pancreatic cancer remains essentially incurable for patients with unresectable tumors. In theUnited States, most patients with locally advanced pancreatic cancer are treated with chemotherapy alone or combined withconventionally fractionated radiotherapy. Regardless of the treatment strategy, average survival for these patients is lessthan 1 year, indicating that the current approaches are indisputably inadequate. For locally advanced pancreatic cancerpatients, effective local-regional control is not only crucial for any hope at long-term survival, but also for symptommanagement. The aim of this paper is to highlight abstracts from the 2014 ASCO Gastrointestinal Cancers Symposium thatdemonstrate the use of novel local-regional therapies in locally advanced pancreatic cancer. Abstracts #317, #328, and #361describe their results with an advanced method of delivering radiation called stereotactic body radiation therapy (SBRT). Inthese studies, patients treated with combined chemotherapy and SBRT had exceptional local control rates and acceptabletoxicity. An innovative alternative to radiation for local-regional treatment is presented in Abstract #270. This study showsencouraging results from a phase I investigation of a regionally delivered siRNA that targets the K-rasG12D mutation.Investigation of novel approaches such as those presented here holds the greatest promise for improving treatment of thisdeadly disease.
Genes, ras; Pancreatic Neoplasms; Radiosurgery
FOLFIRINOX: oxaliplatin, irinotecan, fluorouracil, and leucovorin; GERCOR: Groupe Coopérateur Multidisciplinaire en Oncologie; SBRT: stereotactic body radiation therapy; SCALOP: Selective Chemoradiation in Advanced Localised Pancreatic Cancer; siRNA: small interfering RNAs
Pancreatic cancer is the fourth most common cause of cancer death in the United States [1]. Currently the only chance for cure in pancreatic cancer is with complete surgical removal of the disease; however, only 20% of tumors are resectable at diagnosis. In 30-40% of patients, the tumor is confined to the pancreatic region, but resection is not feasible. Treatment of this group of patients, said to have locally advanced pancreatic cancer, is highly controversial. In the United States, the recommended treatment options include chemotherapy (gemcitabine, capecitabine or combination of oxaliplatin, irinotecan, fluorouracil, and leucovorin (FOLFIRINOX)) alone, concurrent with, or followed by, conventionally fractionated radiotherapy. Regardless of the treatment strategy, the average overall survival for patients with locally advanced pancreatic cancer remains less than 1 year. The universally dismal outcomes seen in this disease underscores a serious need for advancements in both local and systemic treatment approaches.
Over the last 30 years, multiple prospective randomized controlled trials have been conducted in patients with locally advanced pancreatic cancer. The results of these trials have conveyed conflicting messages regarding the best method of treatment, but have consistently shown poor outcomes. One potentially promising strategy is to use chemoradiation or radiation alone only after a response to initial chemotherapy has been demonstrated. This approach allows for patients to declare themselves as metastatic early and possibly avoid 5 to 6 weeks of chemoradiation. The results of the Groupe Coopérateur Multidisciplinaire en Oncologie (GERCOR) phase II and III studies demonstrated improved progression free and overall survival in patients with a response to initial chemotherapy who were subsequently treated with chemoradiation compared to continuing chemotherapy alone [2]. Similarly, the Selective Chemoradiation in Advanced Localised Pancreatic Cancer (SCALOP) phase II study showed improved results with initial chemotherapy followed by capecitabine-based chemoradiation [3]. In contrast to the aforementioned studies, the phase III LAP-07 trial showed no difference in survival between capecitabine-based chemotherapy alone compared to concurrent chemoradiation in patients without progression after initial chemotherapy [4]. The results of the LAP-07 trial imply that standard radiotherapy might have limited efficacy in this subset of patients. Perhaps if higher radiation doses with tumor ablative intent could be safely delivered, then more consistently favorable outcomes could be achieved.
Over the last decade, there have been significant advancements in the field of radiation oncology. Stereotactic body radiation therapy (SBRT) is an image-guided, high-dose per fraction, non-invasive technique of delivering radiation. It is highly conformal and thus potentially limits toxicity to surrounding normal tissues. SBRT is typically delivered in 1-5 total treatment sessions. To date, there have been no prospective randomized trials evaluating SBRT against standard radiotherapy in pancreatic cancer; however multiple studies have shown approximately equivalent survival outcomes. What appears to be a possible advantage of SBRT in locally advanced pancreatic cancer patients is the achievement of higher rates of local control with several studies boasting 1-year local control rates of 84-94% when SBRT is combined with chemotherapy [5, 6]. Likely as a result of better local control, important improvements in symptoms and reduction in pain have also been observed in locally advanced pancreatic cancer patients treated with SBRT [7, 8]. Similar to standard radiotherapy, gastrointestinal toxicity is the most common doselimiting side effect of pancreas SBRT. Late grade 3 or more severe adverse effects are experienced by approximately 5-10% of patients [9]. A severe late adverse effect of pancreas SBRT is gastrointestinal bleeding and a small number of bleeds have actually been fatal. The optimal method of safely delivering effective SBRT still needs to be established. Abstracts #317, #328, and #361 discussed below, describe their institutions’ experiences with SBRT in the treatment of locally advanced pancreatic cancer [10, 11, 12].
For patients with locally advanced pancreatic cancer, radiation is not the only hopeful approach for effective local-regional treatment. As we learn more about the fundamental molecular drivers of pancreatic cancer, new potential targets for therapy are revealed. Activating K-RAS mutations are found in 70-90% of pancreatic cancers and are thought to represent a relatively early, yet significant genetic event in tumorigenesis. Over the recent years, we have welcomed an expansion in our armamentarium against the molecular and cellular pathways exploited by tumors. In pancreatic cancer, targeted therapy has been primarily tested in the laboratory and metastatic setting. K-RAS inhibition with tipifarnib, a farnesyltransferase inhibitor, unfortunately did not show any benefit when added to gemcitabine in metastatic patients. However, in a subgroup of locally advanced pancreatic cancer patients, there was a trend toward improved survival with the addition of K-RAS inhibition [13]. Other methods of targeting the K-RAS pathway are also being developed. Small interfering RNAs (siRNA) inhibit protein production through posttranscriptional gene silencing. In vitro, preclinical data demonstrate reliable anti-neoplastic effects of mutant specific K-ras siRNA in pancreatic cancer cell lines [14]. Presented below are the promising early clinical results of a study using an innovative loco-regional delivery method of a siRNA targeting the mutated K-RAS oncogene [15].
At the 2014 ASCO Gastrointestinal Cancers Symposium multiple presentations showcased the potential utility of novel local-regional therapies in locally advanced pancreatic cancer (Table 1). Abstracts #317, #328, and #361 describe single institution, retrospective studies of patients with locally advanced pancreatic cancer treated with various combinations of chemotherapy and SBRT. Pollom et al. in Abstract #317 presented their results of 199 locally advanced pancreatic cancer patients primarily treated with gemcitabine-based chemotherapy followed by or concurrent with SBRT (25-33 Gy in 1-5 fractions). With a median followup of 7.1 months, the 6- and 12-month local control rates were approximately 92% and 87.5%, respectively. Median overall survival was 14.3 months with 71.2% alive after 6 months and 36.2% alive beyond 1 year. No predictors for local control were identified; however, patients who received chemotherapy had relatively prolonged overall survival. Gastrointestinal toxicity was experienced in 13%; most of which were late events including ulcers, strictures, and bleeding. Similar findings were made by Moningi et al. in Abstract #328 where 66 patients with locally advanced pancreatic cancer or borderline resectable disease were treated with SBRT (20-33 Gy in 5 fractions) combined with a variety of chemotherapies (gemcitabine, cisplatin, FOLFIRINOX, 5-FU or paclitaxel). Local control rates at 6 months and 1 year were 84.6% and 81.8%, respectively. Notably, five R0 resections were achieved and there was a relatively high median survival of 17.8 months. There was 5% incidence of late grade 3/4 gastrointestinal toxicity. One patient had a grade 5 gastrointestinal bleed, which was felt to be the result of tumor invasion into the duodenum. Gurka et al. in Abstract #361 analyzed the results from 27 patients treated with SBRT (25-30 Gy in 5 fractions) concurrent with chemotherapy (gemcitabine, modified combination of 5-flouorouracil, leucovorin and oxaliplatin (mFOLFOX) or capecitabine). Median progression free survival was 8.7 months and median overall survival was 13 months. Similar to the others, they reported a very high local control rate with distant failure predominating. Again this treatment was associated with a few cases of severe late gastrointestinal toxicity including one late grade 4 biliary stricture and one late grade 5 gastric hemorrhage.
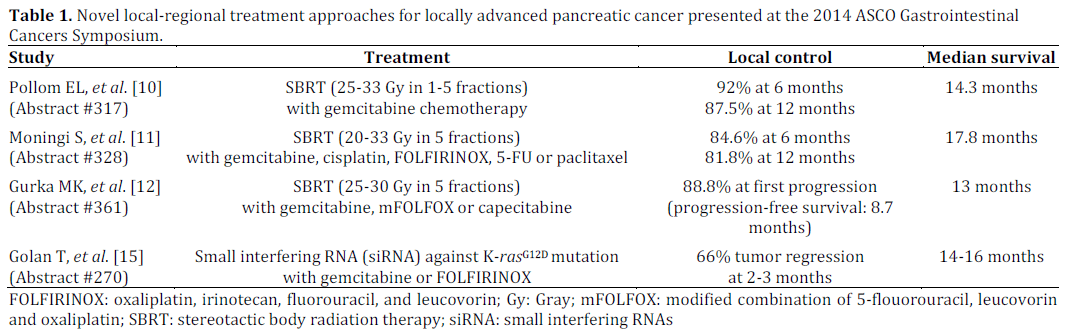
An innovative local-regional treatment presented at the symposium was by Golan et al. in Abstract #270. They presented their results from a phase I study of 13 patients with locally advanced pancreatic cancer treated with an endoscopically delivered miniature biodegradable polymeric matrix that contains siRNA against the K-rasG12D mutant (siG12D LODER). The siG12D LODER was given at increasing doses concurrently with weekly gemcitabine in the first line setting. The derived recommended phase II dose was also combined with FOLFIRINOX. At 8-12 weeks, tumor regression was seen in 66% of patients and CA 19-9 decreased in 64% of patients. Median survival was approximately 14-16 months in all treatment groups. Most side effects were mild; however, 4 patients experienced serious adverse events.
In locally advanced pancreatic cancer, SBRT offers patients the advantage of a noninvasive local therapy with a relatively short duration that does not interfere with the delivery of systemic treatments. Four abstracts presented at the 2014 Gastrointestinal Cancers Symposium exhibited their respective institutions’ experiences treating locally advanced pancreatic cancer with SBRT and chemotherapy. Generally, their results add to the growing evidence that excellent local control rates can be achieved with this approach. Collectively, 1- year local failure was 12.5-20%, compared to 25- 50% seen in patients treated with chemotherapy and standard radiotherapy. In the Moningi et al. study (Abstract #328 [11]), a median survival of 17.8 months was demonstrated, which is better than historic outcomes. However, the percentage of the patients with borderline resectable disease in this study was not reported. Unfortunately, no significant improvements in survival were seen in the other studies that purely analyzed unresectable locally advanced pancreatic cancer patient outcomes. Some other possible drawbacks of these studies are that they were retrospective, potentially confounded by selection bias, and reflect only single institution experiences treating a somewhat heterogeneous patient population with different combinations of chemotherapy and SBRT regimens.
The major hindrance for dose escalation in conventionally delivered radiotherapy is normal tissue toxicity. SBRT is a radiation treatment technique that is highly targeted and can theoretically deliver higher, potentially ablative, doses to a tumor while sparing surrounding normal tissue. With the SBRT regimens used in the studies presented at the symposium, the authors concluded that SBRT was well tolerated. Looking at these studies collectively, there was an approximately 5- 15% incidence of serious late gastrointestinal toxicity. Many patients and their providers might not consider this an acceptable risk of treatment. Additionally, there was one fatal gastrointestinal bleed in both the Moningi et al. study (Abstract #328 [11]) and the Gurka et al. study (Abstract #361 [12]). Despite the excellent local control rates reported in these studies, it clearly carries with it a risk of serious toxicity. It is imperative that future studies seek to determine the optimal fractionation scheme that provides the highest therapeutic ratio. Future research should focus on more accurately defining normal tissue constraints for SBRT, so that patients can reliably and safely benefit from this method of treatment.
Significant improvements in long-term survival will likely be achieved through ongoing attempts to exploit pancreatic cancer’s molecular pathogenesis. There are multiple ongoing studies evaluating the role of targeted therapies in locally advanced pancreatic cancer. Good preclinical and now early clinical data suggest anti-tumor activity of the siRNA that inhibits K-rasG12D. The siG12D LODER used by Golan et al. (Abstract #270 [15]) is injected into the tumor and the K-ras specific siRNA is released regionally. Possible advantages of this approach include an intensification of locoregional therapy while avoiding added systemic toxicities. Although this requires endoscopic delivery, this is less invasive than other procedures used in the treatment of pancreatic cancer. A potential disadvantage of the siG12D LODER is that the effective physical range of drug penetration is unclear and probably patient/tumor dependent. Nonetheless, in this small group of patients, the majority of their tumors regressed. The efficacy of the K-ras-targeted siRNA in combination with chemotherapy should be further tested. When combined with chemotherapy, the K-ras-targeted siRNA was reportedly well tolerated; however, 4 of the 13 treated patients experienced “serious adverse events.” It is difficult to judge the acceptability of these adverse events without more information. It is also worth mentioning that the patients in the Golan et al. study (Abstract #270 [15]) did not receive radiation. K-ras knockdown by siRNA or inhibition of prenyltransferase activity has been shown to result in radiation sensitization in vitro and in vivo in tumors with oncogenic K-ras mutations [16]. Future clinical investigations should incorporate combinations of local radiation, locoregional K-ras inhibition and systemic therapies.
The treatment of locally advanced pancreatic cancer remains a major challenge for oncologists. The research presented at the 2014 ASCO Gastrointestinal Cancers Symposium indicates a positive trend towards the development of novel therapeutics for locally advanced pancreatic cancer. Ongoing efforts like these will hopefully make significant impacts on this currently dismal disease.
The authors have no potential conflict of interest