Case Report - (2018) Volume 0, Issue 0
Awad Magbri*, Saad Rashid, Zach Crowe and Shaukat Rashid
Toledo Vascular Access Center, Toledo, Ohio 43617, USA
*Corresponding Author:
Awad Magbri MD FACP, FASDIN
Toledo Vascular Access Center
Toledo, Ohio 43617, USA
Tel: +1 4122715106
E-mail: elmagbri@hotmail.com
Received Date: April 24, 2018; Accepted Date: May 07, 2018; Published Date: May 18, 2018
Citation: Magbri A, Rashid S, Crowe Z, Rashid S (2018) Nocuous Skin Manifestations of Spironolactone: Drug Induced Leukocytoclastic Vasculitis. Acta Psychopathol 4:13. doi: 10.4172/2469-6676.100169
The author discusses a case of drug induced vasculitis caused by spironolactone which is very rare complication of the drug. The authors give account on the type of vasculitis caused by drug induced allergy and also the differential diagnoses if the presentation is unusual with only hematuria and kidney injury. The benefits of Spironolactone and the side effects are also illustrated.
Keywords
Vasculitis; Drug rash; Spironolactone; Hyperkalemia; ANCA associated vasculitis; Cryoglobulinemia; Leukocytoclastic vasculitis; Hypocomplementemia
Case History
83 year-old Caucasian female with past medical history of hypertension, chronic kidney disease stage 3-4 secondary to hypertensive nephrosclerosis with minimal proteinuria, hypokalemia, and Sjugrene syndrome without extra-glandular involvement. The patient was admitted to hospital because of urinary tract infection and edema of the lower extremities. She was treated with antibiotics, and diuretics for the edema. Her blood pressure was not optimum at this visit and spironolactone 25 mg was added to her medication to control her BP, hypokalemia, and proteinuria. She was on amlodipine, frusemide, clonidine, Irbesartan, and potassium supplement. The treating nephrologist decided to try spironolactone for better control of high BP and hypokalemia and phased out the potassium supplementation. At this encounter her micro-albumin-creatinine ratio was 379.3 mg/ gr, (normal value<29.9 mg/gr creatinine). Physical examination was otherwise normal except bilateral leg edema, and high blood pressure (149/65 mmHg). Her estimated glomerular filtration rate (eGFR) was 28 ml/min).
She returned to the nephrology clinic after 2 weeks for bilateral lower extremities itch, burning and painful rash involving the buttocks, upper thighs and shins bilaterally as shown in the Figures 1 and 2. The rash was palpable, purpuric nodules symmetrically distributed over the lower extremities. She was send for dermatological opinion and skin biopsy of the rash. The biopsy was consistent with leukocytoclastic vasculitis. On further questioning the patient, she recalled that she had had a similar rash long time ago when she was placed on “aldactone” and the doctor had to stop the medication because of the rash, subsequently, the rash went away. This triggered discontinuation of the spironolactone and she was treated with prednisone and topical steroids by the dermatologist. The current rash faded away in 3-4 weeks, however, she was still on 5 mg of prednisone when she was last seen in the clinic. Her laboratory values are illustrated in Table 1.
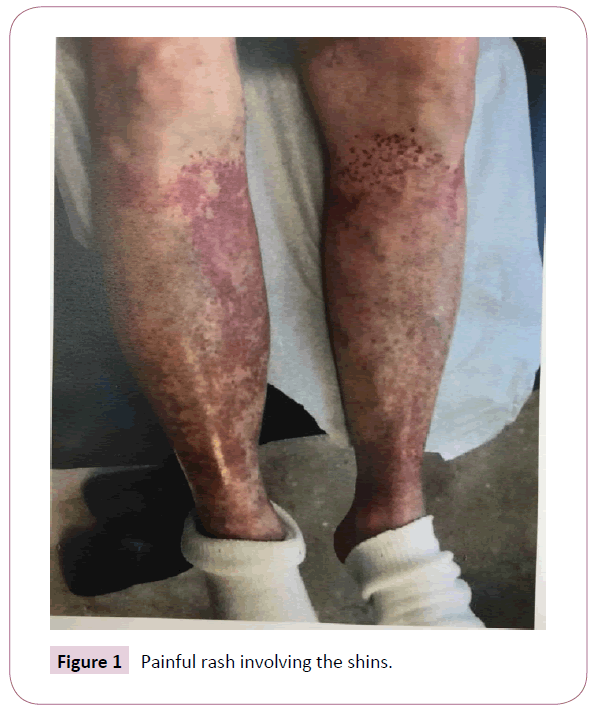
Figure 1: Painful rash involving the shins.
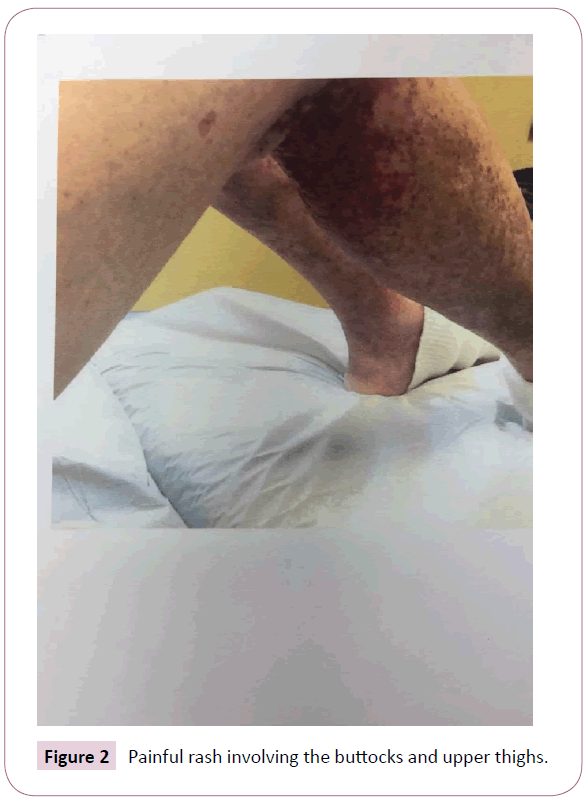
Figure 2: Painful rash involving the buttocks and upper thighs.
| Test | 1st visit | 2nd visit | 3rd visit | 4th visit |
|---|---|---|---|---|
| WBC (4.2-9.5) | 7.6 | 9 | 9.1 | 8.9 |
| Hemoglobin (12-15 gr/dl) | 11.7 | 11.5 | 11.9 | 11.9 |
| Hematocrit (35.3-43.6%) | 34.8 | 34.7 | 35.8 | 35.8 |
| Glucose (74-106 gr/dl) | 96 | 75 | 74 | 108 |
| BUN (7-18 mg/dl) | 27 | 18 | 32 | 35 |
| Creatinine (0.55-1.02 mg/dl) | 1.35 | 1.52 | 1.82 | 1.5 |
| Calcium (9-10 mg/dl) | 9.2 | 9.6 | 9.4 | 9.5 |
| Sodium (136-145 mEq/dl) | 136 | 136 | 134 | 136 |
| Potassium (3.5-5.1 mEq/dl) | 3.8 | 4.1 | 3.6 | 4.2 |
| Chloride (98-107 mEq/dl) | 96 | 97 | 96 | 98 |
| Carbone dioxide (21-32 mEq/dl) | 34 | 32 | 31 | 32 |
| Anion gap (3-11 mEq/dl) | 6 | 7 | 7 | |
| PTHI (11-67 pg/ml) | 79.7 | 32.1 | 55.8 | |
| ANCA | Negative | |||
| ANA | Negative | |||
| Complement | normal | |||
| Cryoglobulins | negative |
Table 1: Laboratory values of patient during visits.
Case Discussion
Spironolactone is a potassium sparing mineralocorticoid receptor antagonist (MRA) which acts on the distal tubules and collecting ducts of the kidneys and antagonizing the effect of aldosterone, thereby causing inhibition of sodium and chloride reabsorption, and potassium secretion in the distal tubules. The bioavailability of spironolactone is 73%, and it is >90% protein-bound. The drug is extensively metabolized in the liver and excreted by renal (47- 57%), bile and eventually fecal route (35-41%). The elimination half-life of the drug ranges from 1.4 to 15-hours depending on the type of metabolites. It is indicated for heart failure with reduced ejection fraction, hypertension especially when associated with hyperaldosteronism, hypokalemia, precocious puberty, hirsutism and female virilization syndrome. The American geriatric association (AGS) recommends that the drug should be avoided in patients >65 years old when creatinine clearance <30 ml/minute due to increased potassium levels associated with the medication [1]. Spironolactone has also been used for liver cirrhosis with ascites and diabetic nephropathy with proteinuria. It can be added to patients with metabolic alkalosis with hypokalemia due to diuretic use.
In renal impairment the drug can be used only if the potassium is low and the (eGFR is >30 mL/minute). The drug is very effective for hypertension associated with hyperaldosteronism. There is black box warning that spironolactone has been shown to be tumorigenic in chronic toxicity studies in rats. It is contraindicated in acute renal insufficiency with low GFR (<30 mL/minute), Addison disease, hyperkalemia, or other conditions associated with high potassium.
Painful gynecomastia, hyperkalemia, hypomagnesemia, hyponatremia, or hypocalcemia are rare side effects of the drug. It is also contra-indicated in pregnancy but compatible with breastfeeding and lactation. Spironolactone has been used in heart failure with reduced ejection fraction (<35%), and after myocardial infarction with either symptomatic heart failure or diabetes mellitus and left ventricular ejection fraction (LVEF<40%).
The adverse effects of spironolactone are estimated to be 1-10% and includes the followings;
a) Gynecomastia (9%)
b) Frequency not defined
c) Vasculitis
d) Ataxia, confusion, dizziness
e) Alopecia, erythematous maculopapular rash, pruritus, Steven- Johnson syndrome, toxic epidermal necrolysis, urticarial
f) Electrolyte disturbances
g) Erectile dysfunction, impotence, mastalgia
h) Agranulocytosis, leukopenia, thrombocytopenia
i) Hepatic toxicity
j) DRESS syndrome
k) Increased blood urea nitrogen, renal failure and fever
Leukocytoclastic vasculitis is an uncommon side effect of spironolactone. It is a histological term that describe small vessel involvement of the subcutaneous tissue with inflammatory infiltrate which is composed of neutrophils [2-6]. It is mainly caused by drugs like phenytoin, allopurinol, non-steroidal anti-inflammatory medications and many antibiotics [7-10], infections with hepatitis C and cryoglobulinemia and malignancy can also be offenders.
Many times, the vasculitis appear after 9-12 days after exposure to the medication. Arthralgia, arthritis, and fever occurring at the same time as the typical rash has been described. Cutaneous small vessel vasculitis (CSVV) with petechial, purpuric eruptions which is non-blanching involving mainly the lower extremities and buttocks are classic presentation of the drug [11,12]. Evidence for circulating immune complex formation including hypocomplementemia, and deposition of the immune complex in the vessel wall of the subcutaneous tissues can be detected by immunofluorescence (IF) [13-15]. This type of vasculitis is attributed to type-III immunological reaction. A lapsing period of 10-14 days is needed for the antigen-antibody deposition in the skin with appearance of the vasculitic rash. Involvement of the visceral and vital organs can sometimes accompany the cutaneous rash.
Low serum complement levels, especially low C4, may be present in mixed cryoglobulinemia and systemic lupus erythematosus vasculitis. The presence of anti-neutrophil cytoplasmic antibodies (ANCA) are indicative of ANCA associated vasculitis. These diseases have to be ruled out when evaluating a vasculitic rash, and pertinent laboratory tests have to be ordered.
Diagnosis and Conclusion
The diagnosis of CSSV is usually suggested by the clinical findings and by the history of drug exposure or infection [12]. Immunofluorescence of the skin biopsy can sometimes demonstrate the deposition of immunoglobulin G (IgG), IgM and or complement. The absence of immune deposition in the histological specimen, however, does not exclude immune complex vasculitis and can be attributed to late timing of biopsy relative to the skin rash, or may be associated with cases of pauci-immune type of vasculitis such as microscopic polyangiitis. If IgA is deposited in the skin on IF microscopy together with gastro-intestinal and joint involvement, IgA vasculitis and Henoch-Schonlein purpura (HSP) should be considered. Non-specific findings like high erythrocyte sedimentation rate (ESR), C - reactive protein, and anemia are common findings with the drug rash.
The differential diagnosis should include systemic vasculitis (ANCA-associated), malignancy which is frequently due to lymphoproliferative disease, and infection.
This case showed a rare side-effect of spironolactone in the form of vasculitis without visceral involvement. The treatment is focused on excluding systemic involvement and symptomatic management of the rash. Prednisone can sometimes be used to haste the healing process. Removing the offending drug is of paramount importance.