Mini Review - (2018) Volume 4, Issue 1
Santiago R Unda*
Instituto de Biotecnología, Centro de Investigación e Innovación Tecnológica, Universidad Nacional de La Rioja, Argentina
*Corresponding Author:
Santiago R Unda
Instituto de Biotecnología, Centro de Investigación e Innovación Tecnológica
Universidad Nacional de La Rioja, Av. Maipú 1367, La Rioja, Argentina.
Tel: 3804277348
E-mail: santiagounda94@gmail.com
Received date: January 08, 2018; Accepted date: January 19, 2018; Published date: January 24, 2018
Citation: Unda SR (2018) Micrornas Potential Biomarkers and Therapeutic Targets for Intracranial Aneurysm. J Clin Epigenet. 4:2. doi: 10.21767/2472-1158.100087
Intracranial aneurysms (IAs) are sac-like dilatations of arteries inside the brain. This pathology can cause severe neuronal and non-neuronal injuries with high impact in the quality of life. Recently, MicroRNAs have been proposed to be a major switch in a wide variety of vascular diseases including the ones in which vascular smooth muscle cells (VSMCs) changes it`s phenotype. The critical role that miR`s may have in the development, progression, and rupture of aneurysms propose them to be a potential tool as a predictor biomarker for clinical diagnostic and progression and also as a future target for new therapeutic interventions. However, miRNAs and their connection with target genes require much efforts before miRNA expression analysis is applied in the clinical practice.
Keywords
MicroRNA; Intracranial aneurysm; Biomarkers
Introduction
Intracranial aneurysms (IAs), also referred to as cerebral aneurysms, are sac-like dilatations of arteries inside the brain [1]. About a 2-3% of the general population is affected by IAs [2]. Most of the patients with an unruptured IAs are asymptomatic, however, IAs rupture can cause a sever type of stroke known as Subarachnoid hemorrhage (SAH) [3]. The SAH is a devastating condition that affects mostly young people (mean age 50 years), of whom a third dies and 50% of survivors are left disabled [4]. This neurological injuries seriously affect the quality of life not only in the economic point of view [5] but also for their secondary complications like rebleeding, early brain injury, cerebral vasospasm, delayed cerebral ischemia and chronic hydrocephalus [6]. As well as extracerebral organ dysfunctions [7].
Despite the efforts of many research groups in understanding the IAs physiopathology [8] and the brain mechanisms that triggers the aneurysm rupture combined with the advances of new surgical and endovascular techniques, the SAH derived from ruptured IAs still remains a challenge for neurosurgeons.
Some of the recent studies identified pro-inflammatory molecules as potential biomarkers [9] based on the role of extracellular matrix turnover factors and inflammatory factors, such as interleukin (IL)-1β, IL-6, IL-8, IL-18, interferon-γ, tumor necrosis factor-α and major histocompatibility complex class II gene, in the development, progression, and rupture of aneurysms [10]. However, new evidence is pointing MicroRNAs (miRNAs/MiR`s) as major switches to mediated post-transcriptional regulation for the proper functioning of cardiovascular homeostasis [11] and fundamental keys in IAs development pathways [12]. This work briefly review the potential role of MiR`s as new biomarkers and therapeutic targets.
Micrornas in Ias Pathogenesis
MiRNAs are single-stranded small noncoding RNAs that consist of approximately 22 nucleotides [13]. This molecules have been involved in a wide range of biological process including vascular diseases. In IAs pathogenesis vascular smooth muscle cells (VSMCs) plays a critical role [14]. VSMCs possess the ability to modulate their phenotype in response to changing local environmental cues [15]. The phenotypic changes in VSMCs improves the migration, proliferation, and production of extracellular matrix components [16]. In this phenotypic expression the MiR`s have seen to modulate functional pathways, the MiR-145 is the most abundant miRNA in vascular walls [17] and recently it`s been proposed for a novel VSMCs phenotypic marker. Preclinical models showed that MiR-145 mediate phenotypic effects through KLF5/myocardin pathway while in clinical studies it was found that rs4705342 TC and TC/CC genotypes in the promoter of the miR-143/145 cluster are related to a lower risk of IA [18]. However, there is still unknown the details of this mechanisms.
The MiR-34a has been reported to regulate vascular smooth muscle cell (VSMC) differentiation from stem cells in vitro and in vivo [19] by reducing Notch1 expression levels, regulator in VSMC functions and arterial remodeling. MiR`s expression are also altered after vascular injury, miR-133 plays a modulatory role in VSMC phenotypic switching [20] and others like the miR-21 and miR-221/222 have a modest effect on neointimal growth. Recently, miR-125 was also involve in modulating the cell proliferation of vascular smooth muscle cells taking as a target the nitric oxide synthase 1 (NOS1) gene [21]. Most of the MiR`s are yet to reveal they role in IAs formation through VSMC proliferation and phenotypical expression. However, the MiR`s could represent a novel tool as biomarkers to predict the patients progression and the best therapeutic targets. In Figure 1 is represented the MiR`s intervention IAs biogenesis.
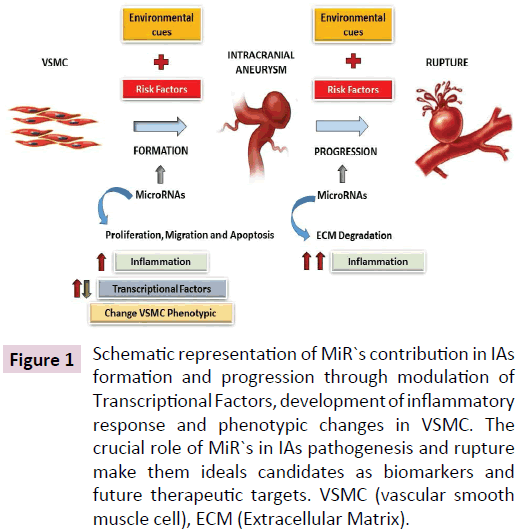
Figure 1: Schematic representation of MiR`s contribution in IAs formation and progression through modulation of Transcriptional Factors, development of inflammatory response and phenotypic changes in VSMC. The crucial role of MiR`s in IAs pathogenesis and rupture make them ideals candidates as biomarkers and future therapeutic targets. VSMC (vascular smooth muscle cell), ECM (Extracellular Matrix).
MiR`s Expression in IAs patients
As was previously mentioned, the MiR-145 has been propose for a novel biomarker, for its central role in vascular proliferative diseases. In peripheral blood of SAH patients, a microarray study indicated that 86 miRNAs were significantly dysregulated [22]. Recently, it was reported that patients with low miR-29a expression had longer disease-free survival (DFS) and overall survival (OS) than those with high miR-29a expression [23]. In other case-control study, lower levels of let-7a in plasma were associated with highest risk for the development of IA [24]. The MiR`s 132 and 324 were also found to upregulate in aneurysmal subarachnoid hemorrhage suggesting a role of this MiR`s in the rupture of IAs [25]. In relation with cytokines, in patients with abdominal aortic aneurysm (AAA), MCP-1 is negative correlated with miRs 146a,-124,-223, the TNFα with miRs-123 and -226 and the TFGβ with miR-146a, suggesting that miRs expression changes are correlated closely with inflammatory process in vascular diseases [26].
Some studies have associated the odds ratio values of MiR`s in IAs patients. The miRâ€ÂÂÂÂ16 and miRâ€ÂÂÂÂ25 levels in plasma were significantly changed in patients with either ruptured or unruptured IAs [27]. The MiR`s -21, miRNA-22 miRNA-720 and miRNA-3665 also increase their expression in IA patients compared to healthy patients [28]. Finally most recent studies identified MiR-29b downregulation induced VSMCs phenotypic modulation by directly activating ATG14-mediated autophagy, which is associated with the formation, growth and rupture of IAs [29].
Final Remarks and Conclusions
Based on the recent studies about the MiR`s modulations in the IAs pathological pathways through target specific genes involved in development, progression, and rupture of aneurysms. MiR`s appear to be a potential tool as molecular biomarkers with high specificity and sensibility. As in other fields, like cancer research, nanomedicine has shown immense potential in advancing treatment regimens [30] so for neurosurgery practice, the use of antogomirs or miRs in nanoparticles could enhance patient’s outcome. However, there´s still some challenges, like the chemotherapeutic agents needs a particular target in oncology therapeutics, the drug delivery is fraught with many limitations [31], in the case of IAs, the nanocarriers system needs to target especific receptors or proteins or such other regions of interest otherwise the stealth capacity of these carriers is compromised and eventually they will be rapidly cleared by the liver, spleen and other RES organs thus showing very little accumulation in aneurysm regions [32]. Summing up, miRNAs could change the prognosis, outcome and quality of life of patients but their connection with target genes requires much efforts before their expression analysis is applied in the clinical diagnosis and treatment of intracranial aneurysm [22].
Conflict of Interest
None to declare.