- (2008) Volume 9, Issue 2
Susan Alsamarai1, Chris Zergebel2, Joshua Zhang2, Taro Furuie2, Peter D Urrea2, Muhammad Wasif Saif1
1Yale University School of Medicine. New Haven, CT, USA.
2Taiho Pharma USA, Inc.. Princeton, NJ, USA
Received December 28th, 2007 - Accepted January 21st, 2008
Context Pancreatic cancer is the third most common gastrointestinal malignancy in the United States. Due to difficulty in diagnosis, 40% of patients are stage IV by the time of diagnosis and median survival is only four to six months. Current therapy for advanced pancreatic cancer focuses largely on gemcitabine. However, a relatively new drug, S-1, is showing promising results. Phase II studies of S-1 monotherapy and recent combination with gemcitabine were conducted for the treatment of metastatic pancreatic cancer. The early phase II study demonstrated a response rate approaching 20% while the combination is reaching more than 35%. Case report We report a 68-year-old man who presented with stage IIB pancreatic cancer which advanced to stage IV after undergoing a Whipple procedure and adjuvant treatment with gemcitabine. The patient was refractory to treatment with gemcitabine as well as irinotecan, taxotere, and cetuximab. He subsequently participated in a trial involving the drug S-1. He achieved 10- month survival with preserved quality of life: he had 14 cycles of S-1 and maintained an ECOG performance status of 0-1 throughout. Conclusion For this patient, 14 cycles of S-1 were well-tolerated for 10 months after failing two prior chemotherapeutic regimens suggesting important insight that S-1 may be active and convenient for its oral use and it may have favorable safety profile in patients with metastatic pancreatic cancer. Randomized trials are warranted to determine the effectiveness of S-1 for the treatment of pancreatic cancer.
Dihydrouracil Dehydrogenase (NADP); Fluorouracil; gemcitabine; Pancreas; Pancreatic Neoplasms; S 1 (combination)
ECOG: Eastern Cooperative Oncology Group
Pancreatic cancer is the third most common gastrointestinal malignancy in the United States. Due to difficulty in diagnosis, 40% of patients are stage IV by the time of diagnosis and median survival remains only four to six months. Current therapy focuses largely on gemcitabine. However, a relatively new drug, S-1, is showing promising results. Investigators at the Institute for Pathogenic Biochemistry in Medicine in Japan wrote in 1998 about a novel 5-FU derivative, named S- 1, which is composed of one molar tegafur (FT), 0.4 molar gimeracil (CDHP) and one molar oteracil potassium (potassium oxonate). The aim is to have the efficacy of 5-FU but without the toxicities [1]. Use of this drug specifically in pancreatic cancer has been demonstrated in a few case reports (Table 1) as well as phase I and II trials. The patient in this report is a 68-year-old gentleman with advanced pancreatic cancer who was refractory to treatment with gemcitabine as well as irinotecan, taxotere, and cetuximab. He subsequently participated in a trial involving the drug S-1. He survived 14 cycles of S-1 and maintained an Eastern Cooperative Oncology Group (ECOG) performance status of 0-1 throughout.
At the age of 68, our patient presented to his primary care physician with complaints of diarrhea and weight loss. For the preceding year, he had started to notice an aversion to food. His past medical history includes type 2 diabetes, hypercholesterolemia, and hypertension. He did not smoke, drank alcohol only socially, and did not use any drugs. He worked as a tile installer and owned his own business. He was married with two children. In the work-up of his symptoms, an upper endoscopy was performed which revealed a Helicobacter pylori infection which was then treated. The patient started to regain some weight, but continued to have diarrhea and developed some light colored stools and pruritus. Liver function tests were obtained at that point, which were abnormally elevated. This prompted his primary physician to order a CT scan of the abdomen on February 9th, 2004, which revealed a 2 cm mass of the head of the pancreas with no liver or nodal involvement. This was followed by an endoscopic ultrasound on February 17th, 2004, which revealed a pancreatic mass abutting the superior mesenteric vein with no direct tumor invasion. A FNA during the EUS revealed adenocarcinoma. The patient was then evaluated by a surgeon and taken to diagnostic laparoscopy on March 2nd, 2004, and ultimately underwent a classic Whipple procedure. Pathology revealed a 3 cm adenocarcinoma invading the pancreas and common bile duct with negative proximal and distal margins. The adjacent pancreas had some chronic pancreatitis, and the gastric antrum had focal intestinal metaplasia. The ampulla and duodenum were negative for tumor. There were 4 of 12 involved peripancreatic lymph nodes and 6 additional perigastric lymph nodes that were negative. This was a T2 N1 MX, stage IIB, grade 2 pancreatic adenocarcinoma. The patient suffered a postoperative ileus, but was managed conservatively and soon discharged home.
On April 1st, 2004, the patient presented to the medical oncology clinic for further treatment. He had lost 32 kg in the past year and complained of nausea, vomiting, and some dyspepsia. His medications at the time included atorvastatin, diltiazem, gemfibrozil, rofecoxib, insulin, lorazepam, metoclopramide, a multivitamin and docusate sodium as needed. He was taking time off of work and had an ECOG performance status of 2-3. Physical exam revealed a well-appearing male in a wheelchair in no acute distress. Head and neck exam revealed pupils anicteric sclera. His abdomen had a surgical incision site that was healing well with no evidence of infection. Bowel sounds were normoactive and there was no palpable hepatosplenomegaly.
It was decided at that point that this patient should ideally be offered adjuvant gemcitabine as single-agent chemotherapy for six months. Given his poor performance status, he was also set up with physical therapy. The patient completed four 7-week cycles of gemcitabine at 920 mg/m2 between April 2004 and July 2004. This was complicated by necrotizing vasculitis in the thighs in September 2004, felt to be related to the gemcitabine. Obstructive cholangitis then occurred in October 2004 and a biliary bypass procedure (hepaticojejunostomy) was performed in January 2005. The patient was recovering well from the prior events, when a staging CT then revealed low attenuation lesions in the liver consistent with metastatic disease. He was started on irinotecan, docetaxel, and cetuximab in early September 2005. He had significant diarrhea with the irinotecan and completed seven treatment cycles through February 2006, with only single agent taxotere by the last cycle due to drug toxicities (Table 2). Due to progressive hepatic metastases, discussion was held with the patient and his family about starting a trial with the drug S-1.

The patient agreed to join the trial in March of 2006 and was started on S-1 in early April of 2006. He tolerated the therapy very well and his ECOG performance status varied between 0-1. He suffered some grade 1 fatigue, but still enjoyed doing yard work and house renovations. His pre-study toxicity assessment noted grade 1 fatigue as well as grade 1 neuropathy, depression and taste changes.
These toxicities remained stable at grade 1 throughout the study period. Additionally, the patient had a brief period of grade 1 diarrhea and stomatitis, which resolved. Constipation became more of a concern, and was managed with laxatives and stool softeners, although this toxicity was not felt to be related to his treatment. The patient completed 14 cycles of S-1 (10.5 months) and continued to have stable disease based on the CT scan and CA 19-9 (Figure 1) performed every 6-week per the protocol. Finally, the patient was then taken off of the study when he was found to have a malignant pleural effusion in January 2007. Unfortunately, he had two long, complicated hospitalizations in January and February of 2007 where he was found not only to have the malignant pleural effusion, but also malignant ascites requiring placement of a tenckhoff catheter for symptomatic relief. He developed renal failure during this time and passed away on February 10th, 2007, likely from renal failure but the cause is not completely certain.
About 33,730 people will develop pancreatic adenocarcinoma each year in the United States, and 32,300 people will die from the disease representing the fourth most common cause of cancer mortality [2]. Surgical resection offers the only chance of a cure but only 15-20% of patients with pancreatic cancer are amenable for surgery. Approximately 40% of patients with pancreatic cancer will present with locally advanced, unresectable disease and the rest will present as metastatic disease. Approximately 80% of patients present with unresectable disease due to the presence of metastases or local extension and the latter group generally develop metastatic disease within the first year of therapy [3].
5-FU and gemcitabine have been mainstays of treatment, and several new drugs on the horizon. Some of these are under development and still have a long way to go, but one oral agent, S-1, has shown efficacy in the treatment of metastatic pancreatic cancer. The patient in this case failed treated with gemcitabine, then moved on to irinotecan, docetaxel, and cetuximab but failed that regimen as well. S-1 became third line treatment in this case, and the patient received 14 cycles over the course of 42 weeks (10.5 months) before his disease ultimately progressed.
S-1 is a fluoropyrimidine derivative that was designed with the intent to improve the antitumor activity of 5-FU but at the same time reduce its gastrointestinal toxicity [3]. S- 1 consists of tegafur (which is a prodrug of 5- FU) combined with two 5-FU biochemical modulators: 5-chloro-2,4-dihydroxypyridine (gimeracil or CDHP) and oteracil potassium. Tegafur is well absorbed orally and converted in the liver to 5-FU. Gimeracil is a competitive inhibitor of dihydropyrimidine dehydrogenase (DPD) which is the enzyme that catabolized 5-FU, and allows for an efficacious level of 5-FU to be maintained in plasma and tumor tissue. 5-FU is associated with serious gastrointestinal toxicities, including heartburn, nausea, vomiting, anorexia, stomatitis, esophagitis, and diarrhea. If the phosphorylation of 5-FU in the gastrointestinal tract is inhibited, its gastrointestinal toxicity can be reduced. Oteracil potassium is able to do this by competitively inhibiting the enzyme orotate phosphoribosyltransferase [4].
S-1 also carries the advantage of being administered orally. Though targeted and molecular therapies may prove to be successful for the future treatment of pancreatic cancer, at the current time, palliation is the goal of therapy for most pancreatic cancer patients [5]. The convenience this conveys can certainly improve quality of life. Additionally, complications of central venous access such as infection, thrombosis, and bleeding can be avoided.
S-1 has shown efficacy in other gastrointestinal malignancies. Several phase II studies in Japan revealed response rates of 35% for advanced colorectal cancer, 22% for non-small cell lung cancer, and 29% for head and neck cancer. Most recently, a multiinstitutional phase II study of S-1 monotherapy in advanced gastric cancer [6] showed an overall response rate of 19.3% (95% confidence interval, 9.2-29.5%). Over a median follow-up duration of 265 days, the median time to progression and overall survival time were 126 and 264 days, respectively. The 1-year survival rate was 34%.
As far as pancreatic cancer, a phase II study published in 2005 performed by Ueno et al. studied 19 patients with metastatic pancreatic adenocarcinoma. These patients were treated with S-1 orally at 40 mg/m2 twice per day for 28 days followed by a 14 day rest period. Of the 19 patients, four patients achieved a partial response (21.1%), no change was noted in 10 patients (52.6%), and 5 patients progressed (26.3%). The median time from entry into the study and demonstration of response was 34.5 days, with median response duration of 7.0 months. Overall median survival was 5.6 months with a 15.8% one year survival. Toxicities included nausea (68.4%, grade ≥1) and anorexia (57.9%, grade ≥1) most commonly. Three patients had grade 3 ileus requiring hospitalization and one patient had grade 3 neutropenia [3].
This was followed up by a late phase II trial which studied the effect of S-1 at 80, 100, and 120 mg per day (depending on body surface area) in 40 patients with chemo-naïve metastatic pancreatic cancer. One patient was observed to have complete response, and 14 patients had partial response. Median survival time was 9.2 months. Anorexia, fatigue, hemoglobin decrease, nausea, and pigmentation change were noted most commonly. Two patients did have disseminated intravascular coagulation, which resolved with anticoagulation [7]. A phase III study is yet to be completed.
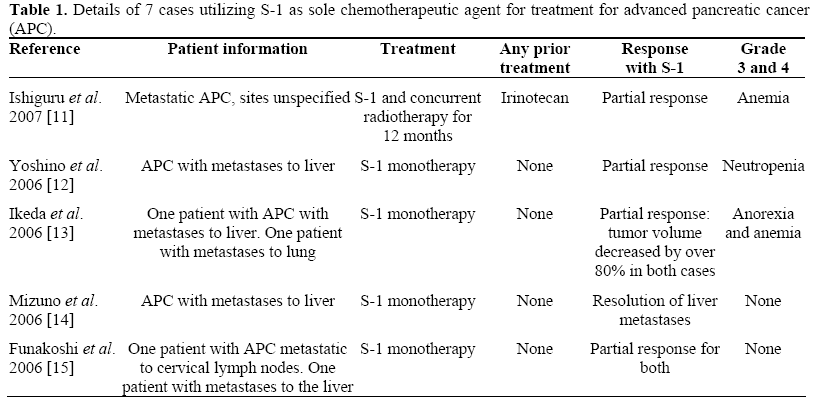
A few case studies have been published in Japan showing efficacy of S-1 for advanced pancreatic cancer, in terms of decreased tumor volume and prolonged survival with maintenance of good quality of life scores. Unfortunately, only the abstracts of these cases are available in English. The details are outlined in Table 1. One such case report noted improvement when S-1 was combined with radiation therapy. This is interesting in light of another study which suggests that the gimeracil component of S-1 has a potent radiosensitizing property [4]. Another phase I study with 21 participants investigated the maximum-tolerated dose of S-1 based on the frequency of dose-limiting toxicities (DLT) of S-1 with concurrent radiotherapy in patients with locally advanced pancreatic cancer [8]. The recommended dose of S-1 therapy with concurrent radiotherapy was 80 mg/m2 based on the study. A follow-up study (a multiinstitutional phase II trial of this regimen in patients with locally advanced pancreatic cancer) is currently underway.
Additionally, there is evidence that S-1 can be combined with the current standard of care, gemcitabine, to produce even better results. A phase II trial published in 2006 studied 33 patients with advanced pancreatic cancer receiving a combination of gemcitabine and S-1 [9]. This study demonstrated 48% response rate (one complete response and fifteen partial responses) after eight cycles. The investigators in this study compare this to the response rate in prior studies for gemcitabine with 5-FU, which has been around 19%. Additionally, in this study, median survival was 12.5 months and 1-year survival was 54%. Myelosuppression, particularly neutropenia, was the primary toxicity in combining the two agents (33%).
More importantly, is to find the efficacy of S- 1 in recurrent and refractory pancreatic cancer patients. A recent study by Togawa et al. treated 17 patients with gemcitabine-resistant pancreatic cancer. Among these 17 patients, 13 had undergone pancreatectomy, and 2 underwent choledochojejunostomy and gastrojejunostomy without pancreatectomy [10]. The regimen included S-1 (80 mg/m2 per day) orally administered for 21 consecutive days, followed by a 14-day rest period and cisplatin (40 mg/m2) i.v. on day 8 repeated every 5 weeks until the occurrence of disease progression, unacceptable toxicities, or the patient's refusal to continue. Overall, five patients (29.4%) achieved a partial response and two (11.8%) had stable disease. In 5 of 15 patients (33.3%) who had elevated serum CA 19-9 levels at the start of treatment, the CA 19-9 was reduced by more than 50%. The median survival time was 10 months. This study further strengthen the finding in our patient that S-1 (with and without cisplatin) has a promising effect against gemcitabine-resistant pancreatic cancer, with easily manageable toxicities.
Despite advances in diagnostic capabilities and understanding of the mechanism of disease, pancreatic cancer remains the fourth leading cause of cancer death in the United States. Advanced, refractory pancreatic adenocarcinoma presents an even greater treatment dilemma. Fortunately, drug development continues and S-1 is showing promising results in several gastrointestinal malignancies, including pancreatic cancer. For the patient discussed above, 14 cycles of S-1 were well-tolerated after failing two prior chemotherapeutic regimens. Additionally, S-1 may not only be beneficial in and of itself, but potentially has value in combination with gemcitabine or radiotherapy.
Dr. S Alsamari has no conflict of interest. Drs. C Zergebel, J Zhang, T Furuie, and PD Urrea are employed by Taiho Pharma USA, Inc. (Princeton, NJ, USA). Dr. MW Saif received research grant from Taiho Pharma USA, Inc. (Princeton, NJ, USA)