- (2011) Volume 12, Issue 2
Bryan W Chang1, Muhammad Wasif Saif2
1Department of Therapeutic Radiology, Yale University School of Medicine. New Haven, CT, USA.
2Division of Hematology/Oncology at Columbia University College of Physicians and Surgeons. New York, NY, USA
Eight abstracts highlighting the continued evolution of the management of locally advanced pancreatic cancer were presented at the 2011 American Society of Clinical Oncology (ASCO) Gastrointestinal Cancers Symposium (Abstracts #195, #239, #259, #281, #287, #305, #307, #324). These studies address key issues including the role of novel targeted therapies, what chemotherapy regimen should be used in conjunction with radiotherapy, and whether locally advanced pancreatic cancer can be managed with chemotherapy alone.
bevacizumab; erlotinib; Fluorouracil; gemcitabine; oxaliplatin; Pancreatic Neoplasms; Radiotherapy
FOLFIRINOX: 5-fluorouracil, leucovorin, irinotecan, and oxaliplatin
Locally advanced pancreatic cancer, in which the tumor has not metastasized but encases the celiac axis or superior mesenteric artery, is by definition unresectable and represents about 25% of pancreatic cancer cases at presentation. Patients with limited vascular involvement by tumor are considered to have borderline resectable disease and are often treated as locally advanced pancreatic cancer. Treatments range from chemoradiation to chemotherapy alone to induction chemotherapy followed by chemoradiation for patients who do not metastasize [1, 2, 3]. The median survival is limited to 10-12 months, leaving significant room for improvement.
Novel Agents for Locally Advanced Pancreatic Cancer Phase I trial of gene-mediated cytotoxic immunotherapyin combination with chemoradiation for locally advanced pancreatic cancer
Bloomston et al. injected escalating doses of an adenoviral vector containing HSV-thymidine kinase gene into tumors of a dozen patients with locally advanced pancreatic cancer, followed by treatment with an anti-viral prodrug and standard 5-fluorouracil (5-FU) and radiation. (Abstract #195). No doselimiting toxicities were seen and there were two partial responses [4].
A phase I study of erlotinib, bevacizumab, and external beam radiation therapy (RT) for patients with localized pancreatic carcinoma (PC)
Dr. Czito and the Duke University group completed a phase I dose-escalation trial of erlotinib, a smallmolecule inhibitor of the epidermal growth factor receptor (EGFR), with bevacizumab, a monoclonal antibody inhibitor of vascular endothelial growth factor (VEGF) (Abstract #281). Borderline resectable and locally advanced pancreatic cancer patients were enrolled. Three of the 9 patients enrolled went to surgery, but only one underwent successful resection [5].
Discussion
The combination of gemcitabine with erlotinib has been shown to modestly extend survival in patients with metastatic pancreatic cancer in comparison to gemcitabine monotherapy [6]. Numerous other biological and targeted therapies have been studied in metastatic and locally advanced disease, without benefit (Table 1). Gene-mediated cytotoxic immunotherapy is a fascinating and elegant concept and appears safe in phase I, but further studies are required to evaluate its true benefit in terms of efficacy. A recent phase III trial of an injectable adenoviral vector bearing tumor necrosis factor-alpha failed to show a survival benefit when added to conventional chemoradiation [7].
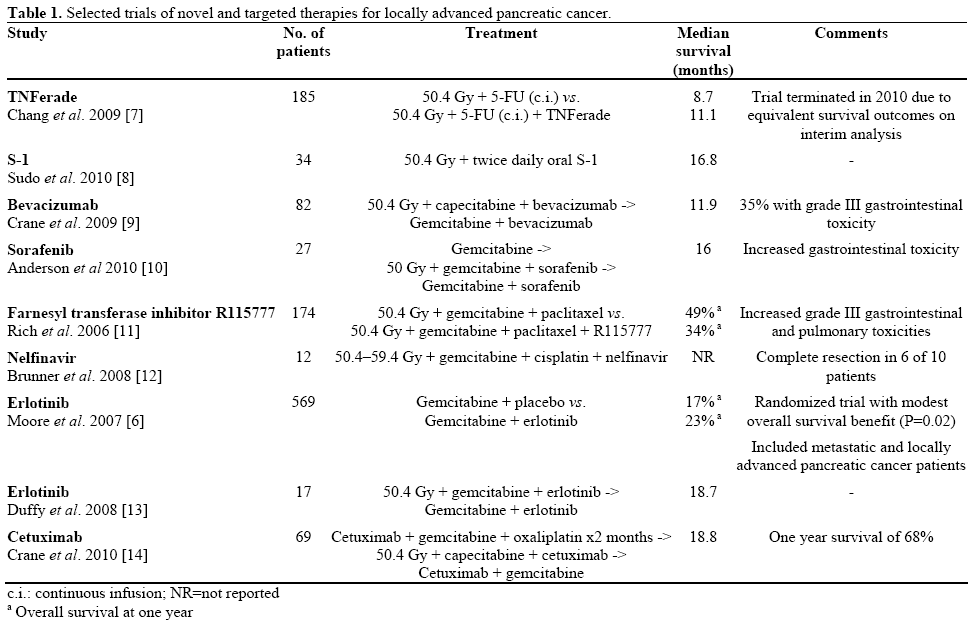
Erlotinib and bevacizumab are both felt to have enhance the antitumor effect of radiotherapy [15, 16], but efficacy in the Duke series does not seem overwhelming. A note of caution is in order as bevacizumab is known to cause wound healing complications after surgery [17], and two patients in this series had serious post-operative complications.
Which Chemotherapy to Combine with Radiotherapy Long-term results of full-dose gemcitabine with radiation therapy compared to 5-fluorouracil with radiation therapy for locally advanced pancreatic cancer.
Huang et al. retrospectively reviewed records of 93 patients with locally advanced pancreatic cancer (Abstract #287). Patients treated with concurrent fulldose gemcitabine and radiation had improved overall survival compared with those who received chemoradiation with 5-FU, and 11% survived for 5 years. There was no difference in acute gastrointestinal toxicity [18].
A phase II study of neoadjuvant gemcitabine/5- fluorouracil followed by 5-fluorouracil/oxaliplatin concurrent with radiation in patients with locally advanced pancreatic cancer.
Dr. Lin and other researchers from University of Nebraska treated 29 borderline resectable patients with 6 weeks of induction chemotherapy followed by chemoradiation to 50-50.4 Gy with 5-FU and weekly oxaliplatin (Abstract #259). Almost half of the patients experienced grade 3-4 acute toxicity, but 7 underwent margin-negative resections. The median survival was 26 months for patients undergoing resection but 10 months for the entire population [19].
Concurrent chemoradiotherapy with gemcitabine and S-1 for unresectable locally advanced pancreatic adenocarcinoma: A phase I study
Ioka et al. treated 15 patients on a phase I protocol with escalating doses of S-1 and gemcitabine combined with 50.4 Gy (Abstract #305). Dose-limiting toxicities were found to be anorexia and neutropenia. The 2-year survival rate was 44.4% [20].
Phase II trial of neoadjuvant full-dose gemcitabine, oxaliplatin, and radiation (RT) in patients with resectable (R) or borderline resectable (BR) pancreatic cancer.
Dr. Kim et al. reported the results of a multiinstitutional trial of neoadjuvant chemoradiation in resectable and borderline resectable patients (Abstract #239). The radiation dose was 30 Gy in 15 fractions. In the borderline resectable population, 19 of 30 patients had a margin-negative resection, and the median survival was 18 months [21].
Discussion
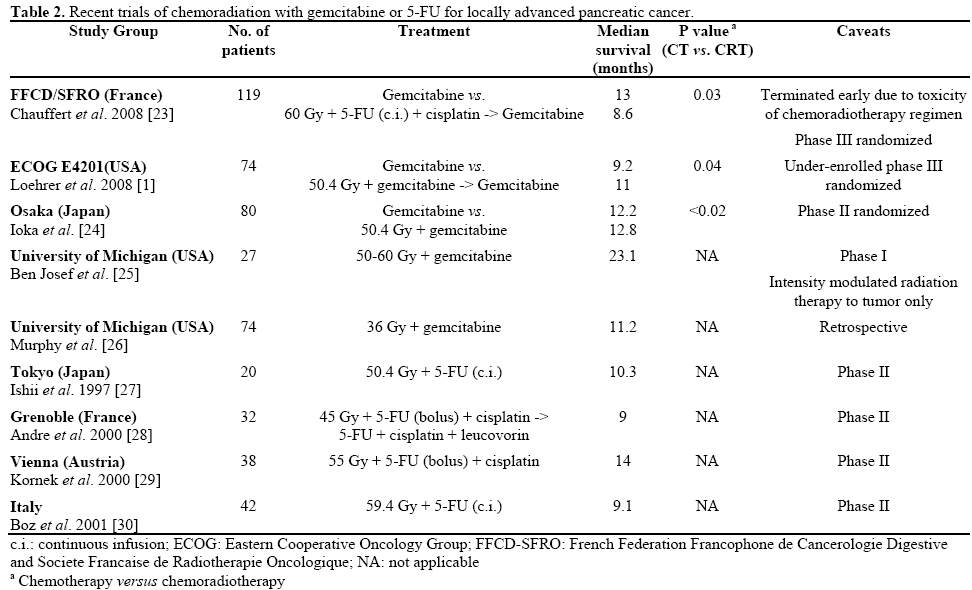
One flawed randomized trial compared gemcitabinebased chemoradiation to 5-FU-based chemoradiation, showing a survival benefit for gemcitabine [22]. Selected series of patients treated with 5-FU and gemcitabine-based regimens are presented in Table 2. Recent studies such as the results presented by Huang et al. [18] have shown promising results for gemcitabine-based chemoradiation. Of note, severe acute toxicity with concurrent full-dose gemcitabine [31] has generally necessitated dose reductions of radiotherapy, as in the Kim et al. study [21], or use of limited-field radiotherapy [32], an approach used successfully by Ioka et al. [20]. Further study in homogeneous patient populations will be needed to better define the efficacy of these promising regimens.
Chemotherapy Alone for Locally Advanced Pancreatic Cancer
Pilot study of neoadjuvant FOLFIRINOX in unresectable locally advanced (LA) pancreatic carcinoma (PC)
Hosein et al. treated 12 patients with locally advanced pancreatic cancer using 5-FU, leucovorin, irinotecan and oxaliplatin (FOLFIRINOX) off protocol (Abstract #324). Four patients were able to undergo marginnegative resections. The overall median survival was estimated at 20.7 months [33].
Phase I/II study of arterial infusion with 5-fluorouracil combined with systemic gemcitabine for unresectable pancreatic cancer
Dr. Tanaka together with a Japanese group studied the toxicity and efficacy of a chemotherapy-alone regimen including hepatic and pancreatic arterial-infusion 5-FU (Abstract #307). A number of metastatic patients were included. The partial-response rate was 68% [34].
Discussion
Trials of chemotherapy for metastatic disease have sometimes included locally advanced pancreatic cancer as well [6], which has led some investigators to treated locally advanced pancreatic cancer without radiation. While the regimen of the Tanaka et al. study appears safe, it requires further study for efficacy in a locally advanced pancreatic cancer population. FOLFIRINOX has been shown to dramatically improve survival in metastatic disease compared with gemcitabine alone [35]. The researchers from Miami and Sao Paulo [33] have reported some of the first results from use of this promising but rigorous regimen in patients with locally advanced pancreatic cancer. Both the median survival and the fact that a third of patients were rendered resectable are impressive, and further study is certainly warranted.
The 2011 ASCO GI Cancer Symposium showcased a number of promising, early-phase results with a variety of therapeutic approaches. The search continues for a targeted therapy with clinically significant activity in locally advanced pancreatic cancer. Meanwhile, researchers are growing increasingly adept at safely and effectively combining gemcitabine-based chemotherapy regimens with radiation. It is clear that some very good results can be obtained with gemcitabine-based chemoradiation, although the optimal approach is not yet clear. Finally, FOLFIRINOX has demonstrated intriguing activity in locally advanced pancreatic cancer, which will need to be confirmed. This regimen may also be useful as induction prior to chemoradiation for patients who do not metastasize early, as large retrospective series have shown that this is a beneficial way to deliver chemoradiation [3, 36]. There is also a need to investigate tailored or adaptive strategies to guide therapy for locally advanced pancreatic cancer. Such strategies could be guided by molecular tumor characteristics or by imaging, such as positronemission tomography. Future trials should include provisions to aid in the development of flexible strategies that maximize therapeutic index and patient benefit.
The authors have no potential conflict of interest