Keywords
Helicobacter pylori; Infection; Autoantibodies; Cytokines; H+-ATPase; Ionic pump; H+, K+-ATPase; Na+, K+- ATPases; Sudden deafness.
Introduction
In 1984, Helicobacter pylori (H. pylori) were first discovered in the stomachs of patients with gastritis and stomach ulcers by Marshall and Warren [1]. It is a microaerophilic, helixshaped curved rod, spirochaete-like Gram negative bacterium, around 3 micrometres (m) long and with a diameter of around 0.5 μm. It is able to obtain energy by oxidizing the H2 molecular by hydrogenase and it produces oxidase, catalase, and urease; the urease enzyme produced allows H. pylori to improve its colonization by altering the organism microenvironment [2].
Since 1984, it was the subject both of investigation of basic biochemical and clinical research and of debate. Its relevance to peptic ulcer disease, gastritis, and gastric malignancy is indisputable [3].
The incidence and prevalence of H. pylori infection, its mode of transmission, and any risk factors have been studied and the results hold that the prevalence
• Varies in relation to the location of the study group and the characteristics of the population studied [4].
• Increases with age [5] and correlates positively with a low socioeconomic status during childhood [6].
It has been highlighted that the acquisition of H. pylori occurs during childhood, via fecal-oral or oral-oral route, and probably the gastro-oral route plays a fundamental role in transmission [7]. Some studies highlight the relevant prevalence of the infection, e.g. a study of 619 blood donors in Turin [8] found an overall prevalence of infection of 51% in males and 27% in females, whilst anti-CagA was found in 17.4% and 18.7% respectively.
Moreover, cecropins produced by H. pylori inhibit the growth of competing organisms [9], as well as a P-type ATPase, which helps prevent excessive alkalinization of the microenvironment by urease [10]. This last result allowed to hypothesize a role played by H. pylori in other diseases and some evidences has also been pointed out; indeed, this bacterium was shown to survive within monocytes and polymorphonuclear cells, thereby potentially travelling throughout the body [11-13].
The aim of this paper is to highlight the possible relation between the H. pylori and the Sudden Sensorineural Hearing Loss (SSHL). SSHL, also known as sudden deafness, consists in a rapid hearing impairment encompassing not less than 30 dB, concerning at least three continuous frequencies and developing in three days maximum [14]. This disease, whose estimated annual incidence is 5 to 20 cases per 100.000 individuals [15], has a dramatic impact on a mainly young and otherwise healthy population. Nevertheless, only 10% to 15% of patients attain an etiological diagnosis [16], e.g. ototoxic drugs, trauma, cancer, immunological [17,18] or metabolic disorders are the most frequent etiological causes reported.
The anatomic site responsible for the functional loss is cochlea. Cochlear damage has been correlated with viral infections [19], mainly by Herpes viridae, such as Cytomegalovirus [20]. Experimental and clinical evidences indicate that a reduction in cochlear blood supply may lead to cochlear ischemia [21,22] and eventually SSHL [23]. In cases characterized by ischemic lesions, cardiovascular risk factors, such as hypertension, hyperlipidemia, hyperhomocysteinemia, as well as local inflammatory status, may often be involved.
The pathophysiologic mechanisms leading to SSHL are not fully understood. Cochlear endolymph is characterized by an extraordinary high extracellular concentration of potassium (140-180 mEq/L) and low concentrations of sodium (13-16 mEq/L) resulting in the generation of a high resting potential, called endocochlear potential. Potassium ions circulate from the endolymph into the sensory hair cells, causing the depolarization of plasma membrane and transduction of sound waves into electrical impulses. Endocochlear potential drives K+ to flow towards the hair cells along a voltage gradient that must be continuously maintained by recycling K+ back into the endolymph [24]. Re-absorptive pathways contributing to K + cycling involve K+ channels [25], a Na+, K+-ATPase [26] and an H+, K+-ATPase [27]. K+ recycling requires an anatomic structure called the spiral ligament, which is located within the cochlea; its integrity is mandatory for the correct functions of the vascular structure (stria vascular) and as a consequence of the entire organ of Corti.
Among all the factors that could regulate the ion flux in the channels, cytokines deserve particular attention. They may alter ion homeostasis within the spiral ligament [28,29] leading to a modified functional status that may alter the activity of gap junctions. The importance of spiral ligament is supported by the observation that mutations of genes encoding for the cochlear connexins are associated with hereditary deafness [30].
All the conditions characterized by increased levels of inflammatory cytokines in the blood stream could contribute to determine a cochlear damage. Helicobacter pylori (H. pylori) infection is the most diffused chronic bacterial infection worldwide, after dental caries. H. pylori is a gram-negative, micro-aerophilic and acidophilic bacterium present in the stomachs of approximately half the world’s population [31,32]. It is the only microorganism capable of living in the hostile gastric environment of man for the entire host’s life [3,32] and causes acute and chronic gastritis that greatly increases the risk of peptic ulcers and gastric cancer [3,32,33]. The cellular and humoral immune response to the infection stimulates the secretion of chemokines, thus contributing to enhance the systemic inflammatory status [34,35]. The inflammatory and immune response may also trigger or exacerbate diseases of organs and systems different and far from the digestive apparatus, concurring to determine acute myocardial infarction [36-38], ischemic stroke of non-cardioembolic origin [39,40] pre-eclampsia [41] (a cerebral ischemia condition of mother and fetus). H. pylori infection is a condition in which autoimmunity is exalted. For instance, patients with H. pylori chronic gastritis frequently develop antibodies to the gastric H +,K+-ATPase, which, in the long run, could lead to an autoimmune gastritis [42]. Autoimmunity is often based on the phenomenon of molecular mimicry between human and bacterial antigens, and it is known that sharing epitopes can work like a trigger for certain autoimmune diseases. Some of us have recently discovered the existence of a linear homology between a main component of sperm flagellum, β-tubulin, and different antigens of H. pylori and have hypothesized that an immune cross reactivity may determine the association between H. pylori infection and male infertility [43].
In this study, we have investigated on the possible association between H. pylori infection and development of SSHL, prompted by the observations that
• H. pylori chronic infection determines an increased, permanent inflammatory status in terms of raised amounts of circulatory cytokines;
• Cytokines could possibly damage the cochlea;
• The gastric H+, K+-ATPase is structurally similar to the cochlear proton pump (PP), which could therefore be damaged by autoantibodies induced by the infection.
To support the conjecture that anti-gastric H+, K+-ATPase antibodies could also injure the cochlear PP through antigen mimicry, we compared the aminoacid (aa) sequence of human H+, K+-ATPases with the sequences of proteins putatively encoded by H. pylori strain J99, whose nucleotidic sequence is available at NCBI. As the cochlear auditive potential is also maintained by a Na+,K+-ATPase, we also aligned the aa sequence of this ionic pump.
Materials and Methods
Case-control study
Thirty-six subjects attending the Clinic of Otolaryngology of the Turin University, Italy, from January to December 2005 and gave their consent to the study were examined.
Patients
Eighteen consecutive patients (11 females and 7 males, mean age 46.3 years), were diagnosed with SSHL. The procedures followed were in accordance with the Helsinki Declaration of the World Medical association. Inclusion and exclusion criteria are reported in Table 1.
| Inclusion Criteria |
Exclusion Criteria |
| Age <70 years |
Congenital hearing loss |
| Clinically normal tympanic membrane and type A tympanogram |
Menière’s syndrome |
| Absence of external and medium ear diseases |
Professional noise exposure |
| Absence of CNS pathologies |
Transmissive hearing impairment |
| Absence of pontocerebellar angle pathologies at NMR |
Metabolic hearing impairment |
| Absence of ototoxic and CNS drug use |
|
| Absence of cardioembolic risk factors |
|
| Absence of sound exposure and trauma |
|
Table 1: Inclusion and exclusion criteria in the studied patients.
Patients’ family history and life style were recorded. All patients received general physical examination, clinical and instrumental investigations necessary for the diagnosis of SSHL. Instrumental examination consisted in pure-tone audiometry (125 Hz, 250 Hz, 500 Hz, 1000 Hz, 2000 Hz, 4000 Hz and 8000 Hz; ISO standard Audiometer A309, Amplifon), and impedance audiometry data (A702 Impedentiometer, Amplifon). To exclude retrocochlear pathologies, each patient underwent brain nuclear magnetic resonance (Siemens Magneton-Expert 1 T, Erlangen, Germany).
The hearing loss degree was classified according to the American Academy of Otolaryngology. Patients were assigned to four groups depending on hearing impairment severity; mild degree=loss higher than 15 dB and less than 40 dB, moderate degree=between 40 dB and 60 dB, severe degree=between 60 dB and 90 dB and deep degree=higher than /90 dB. According to the severity of hearing impairment patients were treated with diuretics and oral or intravenous corticosteroids.
As controls, 18 subjects, 13 females and 5 males, (mean age 42 years) without hear impairment and autoimmune, metabolic and circulatory disorders, who attended the outpatient service of the same Clinic, were enrolled. They were matched for age, sex and social background; they underwent a general physical examination and pure tone audiometry. All patients and controls were investigated for serum antibodies against H. pylori and against H+, K+-ATPase.
Blood chemistry
The other tests performed included hemocytometry and platelets count; blood levels of fasting glucose, fibrinogen, cholesterol, C-reactive protein (CRP), albumin, ferritin, uric acid, folic acid, vitamin B12, and erythrocyte sedimentation rate. All values were obtained by standard laboratory methods.
Serology
A commercial test was used to determine serum antibodies to the following pathogens: Cytomegalovirus, Herpes simplex virus (IgM), Epstein-Barr virus (IgM), influenza virus A and B, parainfluenza virus 1-3, Echo virus, Coxsackie virus, Hepatitis B and C Viruses, Chlamydia pneumoniae, Mycoplasma pneumoniae and H. pylori. We also determined the presence of anti-nuclear, anticardiolipin and anti-H+, K+-ATPase serum antibodies (by a commercial ELISA kit, Eurimmune, Germany). Anti-H. pylori antibodies were investigated using a commercial ELISA kit (Eurospital, Trieste, Italy). To test the concept of molecular mimicry in relation to the origin of autoantibodies reacting with cochlear ATPase, the amino acid (aa) sequence of human gastric H+, K+-ATPase was “blasted” in the protein databases of Helicobacter pylori strains J99 at the National Center for Biotechnology Information (NCBI), Bethesda, MD, USA [44]. The identical sequences (at least five consecutive amino acids) were recorded and aligned with the sequences of the corresponding bacterial proteins. In some cases, sequences longer than five amino acids were also included, even if the alignment was interrupted by one or two nonmatching amino acids.
Statistical Analysis
A logistic regression analysis and the one-tailed Fisher exact test were performed to calculate the odds ratio using the statistical software SPSS v.13.0 Software (SPSS Inc., Chicago, IL). P-values<0.05 were considered statistically significant.
Results
Nine patients out of the eighteen enrolled in the present study developed unilateral and nine bilateral SSHL. Disease severity in unilateral cases was mild or moderate in seven patients, severe in one patient and deep in the other one; in bilateral cases of hearing impairment, disease severity was mild or moderate in one patient, severe in another one and deep in the other seven patients. Four patients presented hearing impairment at all frequencies; six had impairment for high frequencies only, one patient for low frequencies and seven for high and low frequencies (U shaped hearing loss). All patients showed normal vestibular reflexes.
Potentially confounding variables among patients and controls were analyzed (Table 2). None of patients and controls had elevated systolic blood pressure; three patients (16.6%) and no control had a diastolic blood pressure higher than 90 mmHg. Hypercholesterolemia was present in 6 patients (33.3%), elevated levels of LDL in three patients (16.6%), high CRP and erythrocyte sedimentation rate levels in four patients (22.2%). All the patients were negative for antinuclear and anti-cardiolipin antibodies and for high titer serum antibodies to Cytomegalovirus, Herpes simplex virus, Epstein-Barr virus, Influenza virus A and B, Parainfluenza virus 1-3, Echo virus, Coxsackie virus, hepatitis B and C viruses, viruses causing sexually-transmitted diseases, C. pneumonia and M. pneumoniae.
| Variables |
Sex |
Mean Age |
Smokers (%) |
Previous drug therapy |
HID/Stroke Familiarity |
Diastolic Hypertension |
High total Cholesterol |
High CRP & ESV |
| M |
F |
| No |
Yes |
Ex |
| Patients |
7 |
11 |
46.3 |
68 |
23 |
9 |
22.7% |
36.4% |
16.6% |
33.3% |
22.2% |
| Control |
5 |
13 |
42.1 |
72 |
28 |
0 |
0% |
4.8% |
0% |
0% |
0% |
Table 2:Prevalence of potentially confounding factors in patients and controls.
All patients except one (94.4%) and ten controls (55.5%) tested positive for antibodies against H. pylori (P=0.008, OR=13.6, 95% CI, 1.32-335.70). Sex, age, smoke, hypertension, familiarity for cardiovascular diseases and stroke, previous episodes of ischemic heart disease, stroke and previous antibiotic therapy (Table 2) are known confounding variables. The level of significance remained high even after correction by multivariate analysis for the following confounding variables (data not shown). No correlation between audiometric patterns and the presence of H. pylori infection was observed (data not shown).
Antibodies against H+, K+-ATPase were present in six out of eighteen patients (33.3%) one out of eighteen controls (5.5%) (P=0.043, OR=8.5, 95% CI 0.80-213.00), all of whom infected. The prevalence of autoantibodies among the H. pylori infected patients and controls were 35.3% (six of seventeen) and 10% (one of ten), respectively (P=0.161, OR=4.9, 95% CI: 0.42-129.70).
The alignment between the aa sequences of the two human ionic pumps and the proteins expressed by H. pylori strain J99 showed significant homologies with several antigens, most of which were ionic pumps themselves (Figure 1). In particular, both the human ATPases aligned in a very significant manner with the product of H. pylori gene 889106 copA, a coppertransporting P-type ATPase (E=3 × 10-10 for the H+, K+-ATPase and=2 × 10-11 for the Na+, K+-ATPases, (Figure 1). Homologous tracts spanned two large segments of the entire bacterial copper-transporting pump. The two sections of the H+, K+- ATPase that aligned were 28% and 21% identical and 51% and 42% similar (Figure 1). The two homologous fragments of the Na+,K+-ATPase were 32% and 20% identical and 60% and 44% similar (Figure 1). Similarity refers to identical amino acids and amino acids sharing similar chemical properties.
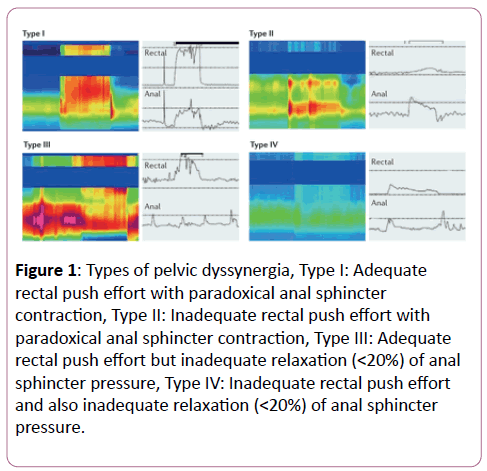
Figure 1: Most significant alignments of human gastric H+, K+-ATPase and Na+, K+-ATPase –transporting-subunit alpha-3 with copper-transporting P-type ATPase (gene id: 889106 copA) from H. pylori. When two proteins are made up by at least five identical (or with the same chemical properties) amino acids in sequence they are considered homologous. Query: tubulin amino acid sequence; Subject: amino acid sequence of the bacterial protein; the numbers in the sequence represent the initial and final positions of amino acid; + indicates that the aligned amino acids are different, although, from an antigenic point of view, they are similar; identities represent the percentage of identical amino acids in the two compared sequences; positives indicate the percentage of amino acids that share the same behavior from an antigenic point of view; gaps mean the number of intervals lacking linear homology between two homologous sequences.
Discussion
Sudden Sensorineural Hearing Loss (SSHL) mostly occurs among young and otherwise healthy active population, impairing an important organ of sensory perception. Every year, about 4000 new cases occur in USA, almost all concerning people aged 30 to 60 years. The potential causes of SSHL are numerous and range from viral infections to ischemic disorders [21-23]. The mechanisms of damage are basically unknown. There is evidence that the full activity of the cochlear H+,K+-ATPase is of basilar importance for a correct function of the organ of Corti. Such a pump is active in fibrocytes of the spiral ligament, in the stria vascularis and in the spiral ganglion [45]. The expression of H+, K+-ATPase in cells of the stria vascularis contributes to maintain the physiological intracellular pH and the endolymph homeostasis and generate the endocochlear potential [27,45].
The observation that increased levels of inflammatory cytokines may damage the cochlea and that the cochlear proton pump (PP) may be altered in cases of SSHL prompted us to investigate a possible association between H. pylori infection and such disease. In fact, 20% to 30% of patients infected by H. pylori present autoantibodies to the gastric H+, K +-ATPase, which shares some epitopes with the bacterial vacuolating toxin VacA [42]. Peptides expressing the same function normally have conserved amino acid sequences, even though they operate in different organs and even in different organisms. We therefore hypothesized that antibodies raised against the VacA protein, which have already been shown to cross react with the gastric H+, K+-ATPase, could target the cochlear ionic pump, with consequent damage of the organ of Corti. Such a hypothesis is reinforced by the observation that the cochlear H+, K+-ATPase and the bacterial ionic pump that regulates the transport of copper, share a wide linear homology (E=3 × 10-10) (Figure 1). The flux of potassium in the cochlear endolymph, however, is also regulated by a Na+, K+- ATPase. The finding that the enzyme shows a highly significant similarity (E=2 × 10-11) with the same type of bacterial ionic pump and with other ATPases further supports our hypothesis that antigen mimicry between bacterial and human epitopes may cause cochlear disorder.
Monoclonal antibodies raised against H. pylori antigens react with several different human tissues, including epithelial cells [46] and endothelial ones, as Franceschi et al. [47] have described the existence of an antigenic mimicry between the vascular wall endothelium and H. pylori; some of us have shown a linear homology between human β-tubulin – largely diffused in the cytoskeleton of different cells - and H. pylori CagA, VacA and flagellar proteins [43]. It is therefore possible that the humoral immune response to certain H. pylori antigens may cross react with the vascular endothelial cells and inner ear antigens (such as P0-myelin and β-actin) – in addition to the cochlear ionic pumps - and damage the cochlear fine microstructure, leading to hearing impairment [45].
Other possible mechanisms of cochlear damage associated with H. pylori infection may involve the ability, exhibited by this bacterium, to activate platelets and other coagulation factors through which the infection may concur to cause arterial obstruction with consequent ischemia [48].
Similar mechanisms have previously been reported to explain the increased risk of myocardial infarction and stroke, run by H. pylori infected patients [38,39]. In SSHL it has been shown that systemic vascular and haematological abnormalities may increase the risk of developing sensorineural hearing loss and that disturbances of the cochlear microvasculature are a prominent etiopathological factor in sudden deafness [49].
Another explanation for our finding that H. pylori infection is associated with SSHL, is as a consequence of gastric atrophy. This condition impairs the absorption of vitamin B12 -with consequent hyper-homocysteinemia [50] and a correlation between augmented systemic levels of homocysteine and SSHL development is known.
Conclusion
Finally, it is known that H. pylori infection stimulates immunocytes and the gastric epithelium to produce cytokines, often at high levels [51]. The importance of inflammatory stimuli in the development and progression of SSHL is documented by the observation that proinflammatory cytokines present in the perilymph of organ of Corti may act directly on the vascular endothelial cells. Cytokines may act on the fibrocytes of the spiral ligament to produce various mediators that could activate inflammatory cells and vascular endothelial cells. Activated cells, on their part, may participate in the inflammatory cell trafficking from the circulation to the site of inflammation; this may contribute to cochlear damage in susceptible individuals [52], eventually leading to SSHL [53].
Our results confirm that patients with SSHL are almost invariably infected by H. pylori and may produce antibodies against the H+, K+- proton pump. To the best of our knowledge, this is the first report of an association between H. pylori infection and development of SSHL. Studies with larger number of patients are needed to confirm the association that we have shown and to ascertain the role (if any) of systemic proinflammatory status and antibodies to the proton pump and endothelial cells in the development of sensorineural hearing loss.
Funding
This work was supported by a grant to AP (Rotfil SpA) and University of Siena 2006 grant (“Possible role of Helicobacter pylori infection in the development of autoimmune diseases, such as Hashimoto's thyroiditis, Sjogren's syndrome and systemic sclerosis” to NF).
Conflicts of Interest
None.
References
- Marshall BJ, Warren JR (1984) Unidentified curved bacilli in the stomach of patients with gastric and peptic ulceration. Lancet 1: 1311-1315.
- Rektorschek M, Weeks D, Sachs G, Melchers K (1998) Influence of pH on metabolism and urease activity of Helicobacter pylori. Gastroenterology 115: 628–641.
- Graham DY (2014) History of Helicobacter pylori, duodenal ulcer, gastric ulcer and gastric cancer. World J Gastroenterol 20: 5191-5204.
- Everhart JE, Kruszon-Moran D, Perez-Perez GI, Tralka TS, McQuillan G (2000) Seroprevalence and ethnic differences in Helicobacter pylori infection among adults in the United States. J Infect Dis 181: 1359–1363.
- Malaty HM, Evans DG, Evans DJ Jr, Graham DY (1992) Helicobacter pylori in Hispanics: comparison with blacks and whites of similar age and socioeconomic class. Gastroenterology 103: 813-816.
- Malaty HM, Graham DY (1994) Importance of childhood socioeconomic status on the current prevalence of Helicobacter pylori infection. Gut 35: 742–745.
- Brown LM (2000) Helicobacter pylori: Epidemiology and routes of transmission. Epidemiol Rev 22: 283-297.
- Ponzetto A, Pellicano R, Morgando A, Cirillo D, Marchiaro G, et al. (2001) Seroprevalence of Helicobacter pylori infection among blood donors in Torino, Italy.Minerva Gastroenterol. Dietol 47: 3-7.
- Putsep K, Branden CI, Boman HG, Nomark S (1999) Antibacterial peptide from H. pylori. Nature 398: 671–672.
- Meichers K, Weitznegger T, Steinhilber W, Sachs G, Schäfer, KP (1995) A novel P type ATPase cloned from Helicobacter pylori. Gastroenterology 108: A165.
- Andersen LP, Blom J, Nielsen H (1993) Survival and ultrastructural changes of Helicobacter pylori after phagocytosis by human polymorphonuclear leukocytes and monocytes. Apmis 101: 61–72.
- Goni E, Franceschi F (2016) Helicobacter pylori and extragastric diseases. Helicobacter 21: 45-48.
- Hwang MS, Forman SN, Kanter JA, Friedman M (2015) Tonsillar Helicobacter pylori colonization in chronic tonsillitis: systematic review and meta-analysis. JAMA Otolaryngol Head Neck Surg 141: 245–249.
- Wilson WR, Byl FM, Laird N (1980) The efficacy of steroids in the treatment of idiopathic sudden hearing loss. Arch Otolaryngol 106: 772–776.
- Byl FM (1984) Sudden hearing loss: An eight years' experience and suggested prognostic table. Laryngoscope 94: 647-661.
- Adams PF, Hendershot GE, Marano MA (1999) Current estimates from the National Health Interview Survey 1996. Vital Health Stat 10: 1-203.
- Mc-Cabe BF (1979) Autoimmune sensorineural hearing loss. Ann Otol Rhinol Laryngol 88: 585-589.
- Yehudai D, Shoenfeld Y, Toubi E (2006) The autoimmune characteristics of progressive or sudden sensorineural hearing loss. Autoimmunity 39: 153-158.
- Wilson WR (1986) The relationship of the herpes virus family to sudden hearing loss. a prospective clinical study and literature review. Laryngoscope 96: 870-877.
- Fukuda S, Furuta Y, Takasu T, Suzuki S, Inuyama Y, et al. (1994) The significance of herpes viral latency in the spiral ganglia. Acta Otolaryngol Suppl 514: 108-110.
- Mom T, Avan P, Gilain L (2002) Idiopathic sudden deafness. Rev Med Interne 23: 292-307.
- Nakashima T, Naganawa S, Sone M, Tominaga M, Hayashi H, et al. (2003) Disorders of cochlear blood flow. Brain Res Brain Res Rev 43: 17-28.
- Morizane I, Hakuba N, Shimizu Y, Shinomori Y, Fujita K, et al. (2005) Transient cochlear ischemia and its effects on the stria vascularis. Neuroreport 16: 799-802.
- Wangemann P (2006) Supporting sensory transduction. Cochlear fluid homeostasis and the endocochlear potential. J Physiol 576: 11-21.
- Marcus DC, Wu T, Wangemann P, Kofuji P (2002) KCNJ10 (Kir4.1) potassium channel knockout abolishes endocochlear potential. Am J Physiol Cell Physiol 282: C403-407.
- Schulte BA, Adams JC (1989) Distribution of immunoreactive Na+, K+-ATPase in gerbil cochlea. J Histochem Cytochem 37: 127-134.
- Stankovic KM, Brown D, Alper SL, Adams JC (1997) Localization of pH regulating proteins H+ATPase and Cl-/HCO3- exchanger in the guinea pig inner ear. Hear Res 114: 21-34.
- Carlson NG, Wieggel WA, Chen J, Bacchi A, Rogers SW, et al. (1999) Inflammatory cytokines IL-1 alpha, IL-1 beta, IL-6, and TNF-alpha impart neuroprotection to an excitotoxin through distinct pathways. J Immunol 163: 3963-3968.
- Nietsch HH, Roe MW, Fiekers JF, Moore AL, Lidofsky SD (2000) Activation of potassium and chloride channels by tumor necrosis factor alpha. Role in liver cell death. J Biol Chem 275: 20556-20561.
- Xia JH, Liu CY, Tang BS, Pan Q, Huang L, et al. (1998) Mutations in the gene encoding gap junction protein beta-3 associated with autosomal dominant hearing impairment. Nat Genet 20: 370-373.
- Parsonnet J (1998) Helicobacter pylori the size of the problem. Gut 1998; 43: S6-9.
- Hooi JKY, Lai WY, Ng WK, Suen MMY, Underwood FE, et al. (2017) Global prevalence of Helicobacter pylori infection: Systematic review and meta-analysis. Gastroenterology (Article in press).
- Blaser MJ, Atherton JC (2004) Helicobacter pylori persistence. Biology and disease. J Clin Invest 113: 321-333.
- Robinson K, Argent RH, Atherton JC (2007) The inflammatory and immune response to Helicobacter pylori infection. Best Pract Res Clin Gastroenterol 21: 237–259.
- Ruggiero P (2010) Helicobacter pylori and inflammation. Curr Pharm Des 16: 4225-4236.
- Mendall MA, Goggin PM, Molineaux N, Levy J, Toosy T, et al. (1994) Relation of Helicobacter pylori infection and coronary heart disease. Br Heart J 71: 437-439.
- Morgando A, Sanseverino P, Perotto C, Molino F, Gai V, et al. (1995) Helicobacter pylori seropositivity in myocardial infarction. Lancet 345: 1380.
- Danesh J, Youngman L, Clark S, Parish S, Peto R, et al. (1999) Helicobacter pylori infection and early onset myocardial infarction. case-control and sibling pairs study. BMJ 319: 1157-1162.
- Markus HS, Mendall MA (1998) Helicobacter pylori infection: a risk factor for ischaemic cerebrovascular disease and carotid atheroma. J Neurol Neurosurg Psychiatry 64: 104-107.
- Ponzetto A, Marchet A, Pellicano R, Lovera N, Chianale G, et al. (2002) Association of Helicobacter pylori infection with ischemic stroke of non-cardiac origin. The BATMAN project study. Hepatogastroenterology 49: 631-634.
- Ponzetto A, Cardaropoli S, Piccoli E, Rolfo A, Gennero L, et al. (2006) Pre-eclampsia is associated with Helicobacter pylori seropositivity in Italy. J Hypertens 24: 2445-2449.
- Claeys D, Faller G, Appelmelk BJ, Negrini R, Kirchner T (1998) The gastric H+,K+-ATPase is a major autoantigen in chronic Helicobacter pylori gastritis with body mucosa atrophy. Gastroenterology 115: 340-347.
- Figura N, Piomboni P, Ponzetto A, Gambera L, Lenzi C, et al. (2002) Helicobacter pylori infection and infertility. Eur J Gastroenterol Hepatol 14: 663-669.
- Altschul SF, Madden TL, Schäffer AA, Zhang J, Zhang Z, et al. (1997) Gapped BLAST and PSI-BLAST. a new generation of protein database search programs. Nucleic Acids Res 25: 3389-3402.
- Boulassel MR, Deggouj N, Tomasi JP, Gersdorff M (2001) Inner ear autoantibodies and their targets in patients with autoimmune inner ear diseases. Acta Otolaryngol 121: 28-34.
- Ko GH, Park HB, Shin MK, Park CK, Lee JH, et al. (1997) Monoclonal antibodies against Helicobacter pylori cross-react with human tissue. Helicobacter 2: 210-215.
- Franceschi F, Sepulveda AR, Gasbarrini A, Pola P, Silveri NG, et al. (2002) Cross-reactivity of anti-CagA antibodies with vascular wall antigens. possible pathogenic link between Helicobacter pylori infection and atherosclerosis. Circulation 106: 430-434.
- Elizalde JI, Gomez J, Panes J, Lozano M, Casadevall M, et al. (1997) Platelet activation in mice and human Helicobacter pylori infection. J Clin Invest 100: 996-1005.
- Shikowitz MJ (1991) Sudden sensorineural hearing loss. Med Clin North Am 75: 1239-1250.
- Shuval-Sudai O, Granot E (2003) An association between Helicobacter pylori infection and serum vitamin B12 levels in healthy adults. J Clin Gastroenterol 36: 130-133.
- D’Elios MM, Amedei A, Benagiano M, Azzurri A, Del Prete G (2005) Helicobacter pylori, T cells and cytokines. the "dangerous liaisons". FEMS Immunol Med Microbiol 44: 113-119.
- Adams J (2002) Clinical implications of inflammatory cytokines in cochlea. Otol Neurootol 23: 316-322.
- Ichimiya I, Yoshida K, Hirano T, Suzuki M, Mogi G (2000) Significance of spiral ligament fibrocytes with cochlear inflammation. Int J Pediatr Otorhinolaryngol 56: 45-51.