- (2002) Volume 3, Issue 2
Gary W Barone1, Beverley L Ketel1, Sameh R Abul-Ezz2, Meredith L Lightfoot1
1Department of Surgery and 2Department of Medicine, University of Arkansas for Medical Sciences. Little Rock, AR, USA
Received: 16 July 2001 Accepted: 14 November 2001
Context A successful immunosuppression regimen for combined kidney and pancreas transplants is tacrolimus, mycophenolate mofetil, and prednisone. However, not allpatients tolerate these immunosuppressants especially tacrolimus. Objective To evaluate the efficacy of cyclosporine as a rescue agent for tacrolimus toxicity in combined kidney and pancreas transplants. Design Retrospective. Setting :Single center. Patients Thirty-five combined kidney and pancreas transplants were performed between July 1994 and January 1999. All patients were insulin dependent diabetics with end-stage renal disease. Twenty-eight (mean age: 36 years and 57% female) were available with at least 12 month follow-up. Interventions Conversion to cyclosporine following renal (biopsy proven) or pancreaticdysfunction. Main outcome mearsures Toxicity, rejectionrate, and patient/transplant organ survival. Results Nineteen transplant recipients (68%) were continuously maintained on tacrolimus while nine (32%) required conversion to cyclosporine 75±20 days post-transplant. Reasons for conversion included: hyperglycemia (n=2), hemolytic-uremic syndrome (n=1), and severe tacrolimus nephrotoxicity (n=6). By 12 months posttransplant, the 19 patients maintained ontacrolimus had 5 rejections (26%). Three of the 9 patients (33%) converted to cyclosporine had an acute rejection prior to conversion. Seven of these 9 patients (78%; P=0.017 vs. patients maintained on tacrolimus) had rejections an average of 25±4 days post-conversion. Four of the 7 patients had no previous rejections prior to conversion. In spite of increased rejections, the 1- and 2-year patient/graft survivals were unchanged by converting. Conclusions Converting to cyclosporine from tacrolimus was associated with an increased risk of acute rejection especially within the first 30 days post conversion.
Drug Toxicity; Graft Rejection; Immunosuppressive Agents
AZA: azathioprine; CKPT: combined kidney and pancreas transplants; CSA: cyclosporine; MMF: mycophenolate mofetil; TAC: tacrolimus
Combined kidney and pancreas transplants (CKPT) are considered to be the treatment of choice in selected patients with insulin dependent diabetes mellitus and end-stage renal disease requiring a renal transplant [1]. Tacrolimus (TAC), a newer formulation of cyclosporine (CSA), and mycophenolate mofetil (MMF) were all approved for clinical transplantation in the mid 1990's. More recently, sirolimus has been added to the transplant immunosuppression formulary. Improved patient and graft survival and decreased episodes of acute transplant rejection in CKPT have been reported with the combined use of these newer agents [2, 3, 4, 5, 6, 7]. A combination of TAC, MMF, prednisone, and most often antibody induction immunotherapy has become the preferred immunosuppression regimen for patients receiving CKPT. However once started, not all CKPT patients tolerate this combination of immunosuppressive agents especially TAC. Kaufman et al. [7] observed that 78% of their CKPT tolerated the combination of TAC/MMF long-term whereas 22% required switching to alternative medications. When intolerance to TAC occurred, CSA has often been chosen as an alternative immunosuppressant. The longterm efficacy and tolerability of CSA as a rescue agent has not been well documented in many series of CKPT [2, 3, 7]. In this current study, the tolerability of TAC was assessed in CKPT, and the outcome of CKPT recipients who were switched to CSA from TAC was reviewed.
Patients
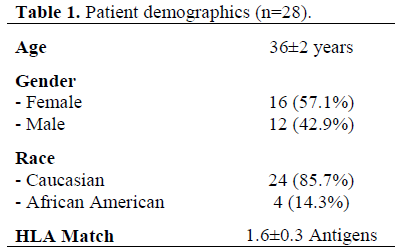
Thirty-five CKPT were performed at the University of Arkansas for Medical Sciences between July 1994 and January 1999 and were retrospectively reviewed. Twenty-eight patients were available with at least a 12- month follow-up. Demographics of these CKP recipient patients are shown in Table 1. The venous drainage for all pancreas transplants was systemic with the portal vein anastomosed to the right external iliac vein.
Immunosuppression
All CKPT received Nashville/Rabbit Antithymocyte (Applied Medical Research, Inc. Nashville, TN, USA) induction therapy on investigational protocol daily for 7-14 days post-transplantation. The first dose was given the day of transplant. Dosage was adjusted according to absolute CD3 lymphocyte counts (target: less than 150 lymphocytes/mL). Oral TAC (Prograf. Fujisawa USA. Deerfield, IL, USA) was started on post-transplant day 1 via a nasogastric tube at 0.1 mg/kg twice daily maintaining 12 hour trough levels of 10-15 ng/mL (IMx System: Tacrolimus II. Abbott Laboratories. Abbott Park, IL, USA). CSA (Neoral. Novartis Pharmaceuticals. Basel, Switzerland), after conversion from TAC, was started at 5 mg/kg twice daily with target 12 hour trough levels of 300-350 ng/mL (TDxFlx System: Cyclosporine Monoclonal. Whole Blood Abbott Laboratories. Abbott Park, IL, USA). All patients received a total of 1 g intravenous methylprednisolone in divided doses peri-operatively followed by an oral prednisone taper. Prednisone dose at one year was 5-10 mg/day. Ten patients received oral azathioprine (AZA) (Imuran. Glaxo Wellcome Research. Triangle Park, NC, USA) at a 2 mg/kg total daily dose while the remaining patients received oral MMF (Cellcept. Roche Pharmaceuticals. Nutley, NJ, USA) at 1,000 mg twice daily.
The induction therapy was administered in accordance with the ethical standards of the 1964 Declaration of Helsinki after receiving written informed consent.
Data are reported as mean values ± standard deviation (SD) and ranges. Survival rates and creatinine values were compared between patients continuously receiving TAC and those converted to CSA by means of the Fisher’s exact and the Mann-Whitney Wilcoxon tests, respectively. Data were analyzed by running the SPSS/PC+ package on a personal computer. A two-tailed P value of 0.05 was chosen to define the statistical significance.
Tolerability of TAC Immunosuppression
Nineteen CKPT recipients (68%) were continuously maintained on TAC whereas nine recipients (32%) required conversion to CSA. Conversion occurred at an average of 75±20 days (range 14-188 days) posttransplant. Renal transplant biopsies were performed in all recipients at the time of conversion. Recipients were converted to CSA because of hyperglycemia (n=2), hemolytic-uremic syndrome (n=1), and severe TAC-induced nephrotoxicity refractory to TAC dose reductions (n=6). Patients with TAC-induced toxicity had TAC levels slightly elevated ranging from 13 to 17 ng/mL.
Rejection
A total of 5 rejections occurred in the 19 patients maintained continuously on TAC resulting in an overall incidence of rejection at 12 months of 26%. All cases of rejection were confirmed by renal transplant biopsy. All rejection episodes were successfully reversed. Two patients were on MMF while 3 were on AZA. These 3 patients converted to MMF post-rejection. Four rejections occurred within 30 days post-transplant while the other rejection occurred at four months posttransplantin a patient who suffered a protracted episode of emesis with resultant low TAC levels. Therefore, no acute rejection episodes occurred after 4 months. Three of the 4 early rejection episodes required antibody treatment.
Three of the nine patients (33%) who were converted to CSA had acute rejection episodes prior to conversion to CSA. These rejections occurred early in the post-transplant course (average 28±5 days post-transplant), and all were successfully reversed.
Importantly, 7 patients (78%; P=0.017 vs. patients maintained on tacrolimus) had rejection episodes after conversion from TAC to CSA with 4 on MMF and 3 on AZA (these 3 patients converted to MMF post-rejection). These rejections occurred early with an average of 25±4 days after conversion. All the 3 patients who had had acute rejection episodes prior to conversion to CSA had also rejection episodes after conversion; therefore, 4 patients (57%) had had no previous rejection episodes prior to CSA conversion. Two of the 7 patients who had rejection episodes, eventually lost renal transplant function and returned to dialysis.
Two patients had no rejection episodes either before or after conversion.
The overall incidence of rejection in the CSA converted CKPT at 12 months was 78%.
Patient and Graft Survival
Patient, kidney, and pancreas allograft survival rates are shown in Table 2. In patients continuously maintained on TAC the 1- and 2-year patient survival rates were 100% and 95%, respectively. One patient died of intestinal infarction 15 months posttransplant with normal allograft function. Patient survival was 100% at 2-years in the 9 patients converted to CSA. The 1- and 2-year kidney allograft survival rates were 100% and 89% respectively, in the TAC group and 89% and 78%, respectively, in the CSA conversion patients.
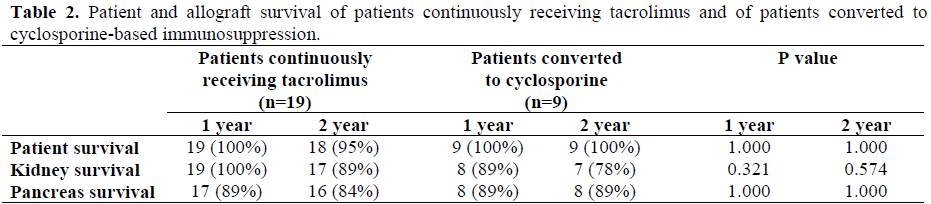
A total of four kidneys were lost, two from each group. In the group receiving TAC, one kidney was lost at 15 months because of patient death; and one kidney failed at 22 months due to polyoma virus infection. In the patients converted to CSA, one kidney failed at 3 months because of irreversible rejection; and a second kidney failed at 14 months from chronic allograft nephropathy. The creatinine in the remaining 7 CSA converted renal transplants at 2 years averaged slightly high at 1.9±0.14 mg/dL (normal range: from 0.4 to 1.0 mg/dL) which was not significantly different from the 1.7±0.17 mg/dL in the group maintained on TAC (P=0.120).
Pancreas allograft survival was defined as normal fasting glucose and normal HgbA1C without exogenous insulin administration. The 1- and 2-year pancreas allograft survival was 89% and 84% respectively in the TAC group, and 89% at both time points in the 9 patients converted to CSA. Three pancreas allografts were lost in the group receiving TAC. Two were due to irreversible rejection in the first 6 months post-transplant, and one was due to patient death at 15 months. One pancreas allograft was lost in patients converted to CSA in a recipient who had severe vascular rejection after plasmapheresis treatment for recurrent hemolytic uremic syndrome. The only one pancreas transplant converted from TAC for hyperglycemia had the HgbA1C decrease from 6.8 to 6.2% (normal range: from 4.4 to 6.4%) post conversion to CSA.
The combination of TAC, MMF, and prednisone for immunosuppression in CKPT recipients has proven to be very efficacious with excellent patient and graft survival and associated with a low incidence of acute cellular rejection episodes [2, 3, 4, 5, 6, 7]. However, some CKPT recipients do not necessarily tolerate all these immunosuppressive agents especially TAC. The best alternative immunosuppressive regimens for CKPT patients intolerant to TAC has not been clearly defined. In our series, 32% of our CKPT patients required discontinuation of TAC because of drugrelated toxicity during the first six months post-transplant. CSA was chosen as an alternative calcineurin inhibitor in these patients. Seven of nine patients experienced an episode of acute rejection within one month of CSA conversion (average 25±4 days). However, 1- and 2-year patient and graft survival were not significantly different in the patients converted from TAC to CSA compared to the group of patients maintained continuously on full dose TAC. While our numbers are small, 60% of the patients on AZA suffered rejections compared to 33% in the MMF group.
Several other reports [2, 3] and one especially by Kaufman et al. [7] support our findings. Stegall et al. [2] did report a 16.7% conversion rate (3 patients in 18 CKPT) from TAC to CSA between 3 to 6 months posttransplant. Conversions were performed not for renal toxicity but for systemic and infections indications. They did not report any specific follow-up in these patients. Peddi et al. [3] noted that 37% of their series of CKPT on TAC developed TAC nephrotoxicity, but all initially responded to reduction in TAC doses. In the series by Kaufman et al. [7], TAC intolerability was observed in 18% of their CKPT recipients. Similarly, conversion to CSA was associated with an increased 12 month post-transplant rejection rate of 56% compared to 20% for the CKPT who remained on TAC. As in our series, 1- and 2- year patient and graft survival was not affected by the conversion.
We conclude that the substitution of CSA for TAC may be associated with an increased risk of acute rejection especially within the first 30 days post conversion. Therefore, patients should be closely monitored during this time period. Two-year results are not worse in CKPT converted from TAC to CSA but longer-term outcomes of allograft function may be adversely affected. Patients who experience acute transplant rejection episodes have lower graft survival than those who remain free of rejection [2]. Because CSA conversion appears to be associated with an increased rate of rejection, the recent introduction of sirolimus may provide another choice. Besides changing to CSA in CKPT recipients who do not tolerate full-dose TAC especially with associated TAC nephrotoxicity, sirolimus with or without low dose TAC may be a better option. However, this combination needs further investigation in CKPT.