- (2004) Volume 5, Issue 2
Linda Tillmar, Nils Welsh
Department of Medical Cell Biology, Uppsala University. Uppsala, Sweden
Received: 20 August 2003 Accepted: 30 December 2003
Angiopoietins; Anoxia; Gene Expression; Platelet Factor 4; Receptors, TIE; Serpins; Thrombospondins; Transplantation; Vascular Endothelial Growth Factors
aFGF/FGF-1: acidic fibroblast growth factor; Ang: angiopoietin; bFGF/FGF- 2: basic fibroblast growth factor; BMP: bone morphogenetic protein; c-MET: tyrosine kinase receptor encoded by the MET protooncogene; ECGF: endothelial cell growth factor; Flt-1: flm-like tyrosine kinase-1; HGF/SF: hepatocyte growth factor/scatter factor; HIF-1: hypoxia inducible factor 1; KDR/Flk-1: kinase insert domain-containing receptor 1/ fetal liver kinase-1; PAI2: plasminogen activator inhibitor 2; PDGF: platelet-derived growth factor; PEDF: pigment epithelium-derived factor; PF-4: platelet factor 4; PTN/HB-GAM: pleiotrophin/heparin-binding growth associated molecule; SERPIN: serine protease inhibitor; Smad5: sma- and MAD-related protein 5; TGF: transforming growth factor; THBS/TSP: thrombospondin; Tie: the tyrosine kinase with immunoglobulin and epidermal growth factor homology domains; uPA: urokinase plasminogen activator; uPAR: urokinase plasminogen activator receptor; VEGF: vascular endothelial growth factor
Considering the present lack of transplantable tissue, islet transplantation is reserved for only a few diabetic patients. To improve the situation, the number of islets needed to cure one recipient needs to be reduced. After collagenase isolation, islets are considered to be avascular. The outcome of the transplantation is in part dependent on a sufficient revascularization of the graft. Lately, it has become evident that, at least in animal models, transplanted aggregated islets contain a final vascular system which differs somewhat from that of endogenous islets in vascular density, blood flow and partial oxygen pressure (reviewed by Jansson and Carlsson [1]). This leads to a state of hypoxia [2, 3] as evidenced by the increased lactateto- pyruvate ratio found in grafted islets as compared to cultured islets [4].
To find methods of improving and accelerating the vascularization process, the factors governing the growth, maturation and remodeling of the islet vasculature need to be identified. Indeed, some of the molecular factors involved in islet angiogenesis have already been recognized. Vascular endothelial growth factor (VEGF) is an elementary regulator of angiogenesis [5]. The expression of VEGF, and its receptors KDR/Flk-1 and Flt-1, is known to increase in devascularized islets (i.e. isolated and cultured islets), as well as in cultured rat islets exposed to hypoxia [6, 7, 8, 9]. Transplanted rat islets demonstrate an elevated expression of VEGF, Flt-1 and KDR/Flk-1 mRNA [9]. However, the notion of a principle role of VEGF in islet revascularization has been challenged. For instance, in a transplantation study comparing fetal and adult islets, twice as much VEGF was released from adult as compared to fetal rat islets. Yet revascularization was not more pronounced in the adult islets [10]. In addition, pharmacological inhibition of KDR/Flk-1, using the antagonist SU5416, does not have any significant effect on islet revascularization [11].
Expression of hepatocyte growth factor/scatter factor (HGF/SF), its tyrosine kinase receptor encoded by the MET protooncogen (c-MET), urokinase plasminogen activator (uPA), its receptor uPAR and vimentin are all increased about day 3-5 in transplanted rat islets [12]. However, the expression of basic fibroblast growth factor (bFGF/FGF-2) mRNA seems to decrease after islet transplantation [12]. The mRNA expression of transforming growth factor (TGF)-alpha and TGF-beta 1 is increased in islet grafts of diabetic, but not of healthy, recipients [12]. Unfortunately, the importance of these changes in gene expression patterns for the vascularization process is unknown.
An important issue is whether isolated and cultured islets express endothelial cells with angiogenic capacity, or if they are lost during the process. The majority of the new vessels found in islet grafts are reported to originate from the recipient [13]. However, in a recent study one third of the endothelial cells present within, or in the vicinity, originated from the transplanted islet, and not from the host. Cultured islets were also shown to contain dedifferentiated endothelial cells, capable of sprouting endothelial cells containing cordlike structures [14].
The aim of the present report was to further analyze the expression of genes with possible relevance for the islet vascularization process.
Islet Isolation and Culture
Twenty-five adult Sprague Dawley rats, from a local colony, were killed by cervical dislocation after an intraperitoneal injection of sodium pentobarbital. The pancreases were taken out and the islets isolated by collagenase digestion [15]. The islets were cultured in 5 mL RPMI-1640, supplemented with 10%, Fetal Clone II serum (Hyclone Europe Ltd, Cramlington, U.K.), 2 mM Lglutamine, 100 U/mL benzyl penicillin and 0.1 mg/mL streptomycin (RPMI-WS). The islets were cultured free-floating for 5-8 days, with the medium changed every second day [15]. To reduce experimental variations due to insufficient diffusion of oxygen and/or nutrients, only small or medium sized islets (approximately with a diameter of less than 200 μm) were used. To obtain hypoxic conditions, 300 islets/5 cm2 were cultured under a medium depth of 4-6 mm, in flasks containing 5 mL of culture medium, which had been gassed with 1% O2/94% N2/5% CO2 for at least 20 min prior to the addition of the islets. The flasks were incubated under airtight conditions.
Gene Expression Array
Isolated islets, 300 per group, were exposed to normoxia (21% O2) or hypoxia (1% O2) for six hours; thereafter, the mRNA expression of 37 angiogenesis-related and two housekeeping genes (glyceraldehyde 3- phosphate dehydrogenase, and beta-actin) was detected using the pathway-specific GEArrayTM system Mouse Cancer/Angiogenesis 1 and 2 (SuperArray Inc., Bethesda, MD, USA). This is an array system that allows the determination of mRNA expression with both high sensitivity and accuracy. The expression of the genes was measured by densitometry and normalized against the housekeeping gene glyceraldehyde 3-phosphate dehydrogenase.
Semi-Quantitative RT-PCR
The three genes regarded as upregulated by hypoxia were further evaluated by RT-PCR. After the incubation of 100 islets per group, at 100, 21 or 1% O2 for 6 hours, total RNA was isolated using the Ultraspec TM Total RNA Isolation System (Biotech Laboratories, Houston, TX, USA). First strand cDNA was synthesized using oligo-dT primers and AMV reverse transcriptase (Promega, Madison, WI, USA). PCR reactions were carried out with the Lightcycler real-time PCR instrument using the Faststart DNA Master CYBR Green I kit (Roche Molecular Biochemical, Mannheim, Germany). The following primers were used:
VEGF forward: 5´-GGTCCCAGGCTGCACCCACG,
VEGF reverse: 5´-TTAGGGGCACACAGGACGGC,
aFGF forward: 5´-CTTTGCAGCCCTGACCGAGAG,
aFGF reverse: 5´-GTGCTGGTCGCTCCTGTCCC,
Tie1 forward: 5´-GCTGCAGAGTGCGAGCCCAG,
Tie1 reverse 5´-CTGGTAGGTGAAGGTATGTCTC,
beta-actin forward: 5´-GCCCTGGCTCCTAGCACC,
beta-actin reverse 5´-CCACCAATCCACACAGAGTACTTG.
PCR reactions using VEGF, aFGF and Tie1 primers resulted in the amplification of more than one PCR product, which made real-time quantifications impossible. Instead, PCR products, after 35 cycles (exponential phase), were separated on agarose gel and the product with the expected size was quantified by densitometric scanning, and normalized against beta-actin.
The use of laboratory animals was approved by the local animal ethics committee according to the “Guide for the Care and Use of Laboratory Animals” prepared by the National Academy of Science (NIH publication 86-23 revised 1985).
Mean values, standard deviations (SD) and standard errors of means (SEM) were used as descriptive statistics. The RT-PCR results were evaluated by the Student’s paired t-test, using the Statview Student v1.0 software. Two-tailed P values less than 0.05 were considered statistically significant.
We have presently utilized the commercially available pathway-specific cDNA array (GEArray™) system for analysis of basal and hypoxia-induced gene expression in primary rat islets, precultured for one week. Of the 37 genes included in the two systems, the expression of 19 genes was detected in islets in two separate experiments (Table 1 and Figure 1). Of these genes, some code for proteins stimulating angiogenesis, while others code for factors inhibiting angiogenesis and some can either stimulate or inhibit angiogenesis, depending on the conditions.
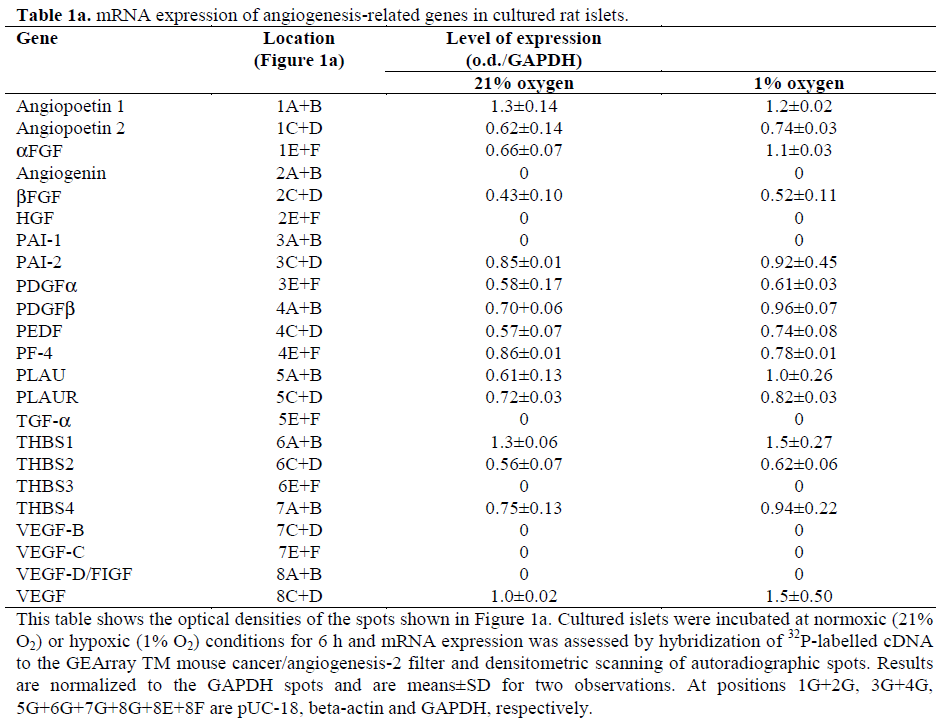
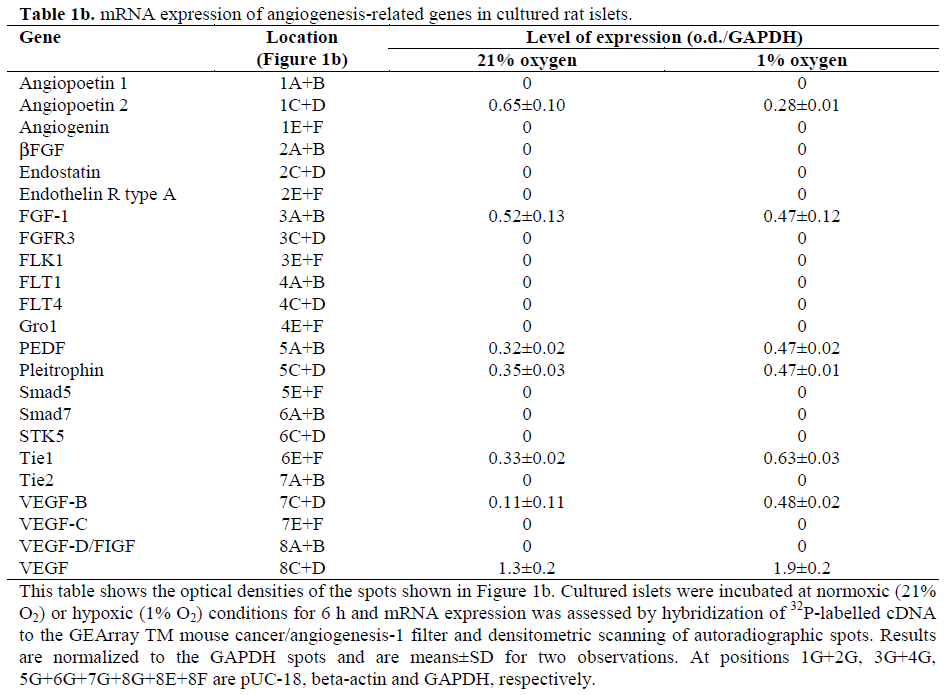
Figure 1. The mRNA expression of angiogenesis-related genes in cultured islets exposed to normoxia or hypoxia. Isolated islets, 300 per group, were exposed to normoxia (21% O2) or hypoxia (1% O2) for six hours and analyzed by the GEArrayTM mouse angiogenesis 1 (Figure 1b) and 2 (Figure 1a) systems. The location of the different genes on the filter is as given in Table 1. This is the result of one of two separate experiments.
The GEArray™ experiments showed possible up-regulation of VEGF, the tyrosine kinase with immunoglobulin and epidermal growth factor homology domains (Tie)1 and acidic fibroblast growth factor (aFGF/FGF-1) mRNA in response to 6 hours of hypoxia (Figure 1). Possible up-regulation was defined as a more than 30% hypoxia-induced increase in both experiments. The possibly upregulated mRNA levels were further analyzed by RTPCR. The VEGF mRNA expression significantly increased when lowering the oxygen level from 21% to 1% oxygen (Figures 2 and 3). For the Tie1 mRNA, there was a significant increase between islets cultured at 100% oxygen versus the islets incubated at 1% oxygen. However, any significant effect of hypoxia on the expression of aFGF/FGF-1 was not found (Figures 2 and 3).
Figure 3. The mRNA expression of VEGF, Tie1 and aFGF in islets exposed to hyperoxia, normoxia or normoxia. Isolated islets, 100 per group, were exposed to hyperoxia (100% O2), normoxia (21% O2) or hypoxia (1% O2) for six hours. mRNA expression was analyzed by RT-PCR and agarose gel electrophoresis. The relative mRNA expression was expressed as percentage of control, where mRNA expression at 100% oxygen was used as control. The results are expressed as mean±SEM.
Rat islets were used in the present study because we had previously characterized hypoxic culture conditions, hypoxia-induced beta-cell death and hypoxia-regulated insulin gene expression in such islets [16, 17]. Unfortunately, the lack of expression of some genes may be due to suboptimal hybridization between the sample rat cDNA and the mouse target cDNA sequence immobilized to the filters. Thus, the absence of gene expression should be interpreted with caution.
Angiogenesis Stimulating Factors
A six hour exposure to 1% oxygen resulted in a modest up-regulation of VEGF- and Tie1- mRNA in cultured rat islets. The hypoxiastimulated expression of VEGF mRNA is in line with previous studies [8, 9] and may be due to the activation of the hypoxia inducible factor-1 (HIF-1) [18, 19, 20]. We could not detect any mRNA expression of the two major, endothelial-specific, VEGF receptors KDR/Flk-1 and Flt-1. Vasir et al. have observed expression of the VEGF receptors in isolated rat islets, but, after 48 h of normoxic culture, the expression was strongly reduced [9]. Thus, it is possible that a culturing period of one week eliminates KDR/Flk-1 and Flt-1 mRNA expression. Hypoxia is known to induce expression of these receptors [9], but the capability of the islets to express the receptors might be lost after prolonged culture in vitro, as a result of a gradual depletion of differentiated endothelial cells. Interestingly, VEGF signaling may not be the principle factor in islet revascularization [10, 11]. Indeed, during development of the pancreas, a VEGF-A-induced islet capillary network is not a prerequisite for blood glucose control, although it was shown to be essential for finetuned regulation [21].
Tie1 and Tie2 are specifically expressed by vascular endothelial cells, hematopoietic cells and their embryonic precursors [22, 23, 24, 25, 26, 27, 28]. The receptors regulate the maturation, stabilization and remodeling of the blood vessels [29, 30]. Tie1, but not Tie2, expression was detected. The Tie2 agonist angiopoietin-1 (Ang1) [31] and the Tie2 antagonist angiopoietin- 2 (Ang2) [32] were also present. In adult tissue, both Ang1 and Tie2 are widely expressed [32]. Ang2, on the other hand, is restricted to endothelial cells at sites of vascular remodeling, i.e. in ovaries, uterus, placenta, tumors and in wound healing [32, 33, 34]. Ang1 promotes endothelial cell survival and microvessel stability [35, 36], and suppression of this signal is thought to be required for VEGF-induced microvessel remodeling or growth [36]. So far no ligand has been identified for Tie1. Instead, Tie1 and Tie2 have been shown to form a complex, and one function of Tie1 may therefore be to directly modulate Tie2 signaling [22, 32, 37, 38]. In line with this, VEGF has been shown to change the composition of the Tie2 complex by promoting binding of the full length Tie2 to the endodomain of Tie1. It is not clear why we could not detect expression of Tie2 in cultured rat islets, but Tie2 expression, of donor origin, has been identified in transplanted islets [14]. Thus, the combined findings of the present and previous studies support the view that Tie2-signaling may play an important role in the hypoxiainduced islet vascularization process and that the VEGF pathway may modulate this signaling cascade.
The family of fibroblast growth factors (FGFs) consists of proteins with angiogenic features [39]. Basic FGF (bFGF/FGF-2) is present in cultured islets and is known to be produced by endothelial cells [40]. The expression of bFGF/FGF-2 mRNA in islets seems to be decreased at least on days 3 to 14 after islet transplantation [12]. This may have a negative impact on the microvascular structure of the islet graft. Indeed, in vivo culturing of islets in the presence of aFGF or bFGF has a positive effect of preservation of the capillary bed within the islets [41]. Furthermore, the administration of bFGF concurrently with islet transplantation in rats significantly increases the vascularization at the site of implantation and the development of normoglycemia [42, 43, 44]. The consequence of culturing islets in the presence of endothelial cell growth factor (ECGF) is shown to be even more beneficial, as this initiates a time-dependent proliferation of intra-islet capillaries and endothelial cells [41]. This is interesting because the new microvessels of islet grafts have been observed to predominantly lie in the connective tissue surrounding the individual islets, rather than within the endocrine tissue [45, 46, 47]. Whether this is due to the fact that the new blood vessels are predominantly of host, and not donor, origin is presently unknown [13, 14]. Unfortunately, ECGF was not included in the gene array system presently used.
Platelet-derived growth factor (PDGF) is a growth factor which acts on endothelial cells. The PDGFs (AA, BB and AB) are expressed in vascular endothelial cells, but also in a range of other cell types. The importance of PDGF for angiogenesis is probably higher in some specific organs than in others [48]. Recent findings show that PDGF regulates expression of other angiogenic factors. For instance, PDGF induces VEGF production [49, 50] but inhibits bFGF expression [51]. Although weak, we detected expression of both the PDGF A- and B-chains in the cultured islets.
Pleiotrophin (PTN, also known as the heparin-binding growth associated molecule, HB-GAM) is an angiogenic factor that promotes both proliferation and migration of endothelial cells [52, 53]. In the adult, PTN mRNA and protein are normally expressed at low levels, but are up-regulated in endothelial cells, macrophages and astrocytes, at sites of injury-induced angiogenesis [54]. Smad5 is a mediator of TGF-beta and bone morphogenetic protein (BMP) signaling, leading to endothelial cell migration and tube formation [55]. Smad5 deficient embryos have enlarged blood vessels surrounded by decreased numbers of vascular smooth muscle cells and they are unable to direct angiogenesis in vitro [56]. Expression of both PTN and Smad5 mRNA was found at low levels in the islets.
An important event in angiogenesis is the degradation of the extracellular matrix (ECM), since this is necessary for endothelial cell migration [57]. This process involves the urokinase type plasminogen activator (uPA/PLAU)/uPA receptor (uPAR/PLAUR) complex which induces the conversion of inactive plasminogen to fibrinolytically active plasmin. We found mRNA expression of both uPA/PLAU and its receptor uPAR/PLAUR in the islets.
Angiogenesis Inhibiting Factors
Pigment epithelium-derived factor (PEDF) and plasminogen activator inhibitor 2 (PAI2) are both members of the serine protease inhibitor (SERPIN) gene family. PAI2 is an endogenous inhibitor of the uPA/PLAU/uPAR/ PLAUR complex [58], and it is expressed in the cultured rat islets. PEDF counteracts the action of several angiogenesis stimulating factors, such as VEGF, aFGF, bFGF and PDGF [59, 60, 61]. The way it prevents angiogenesis may be due to its recently discovered capability of specifically inducing apoptosis, using the Fas/Fas ligand pathway, in endothelial cells which areforming new vessels, while leaving pre-existing vessels intact [62]. PEDF expression, at low levels, was also detected.
PF-4 is a chemokine which exerts inhibitory effects on endothelial cell proliferation, migration and angiogenesis [63, 64]. The mechanism behind this effect is not completely understood, but it is known that PF-4 can inhibit binding of VEGF and bFGF to their respective receptors and that it blocks the progression of endothelial cells into the Sphase [64, 65, 66, 67]. PF-4 was also expressed in the cultured islets, although at low levels.
The thrombospondins (THBS/TSP) are complex glycoproteins, which are located at the cell surface and the extracellular matrix. The most extensively studied members of the family are THBS/TSP-1 and THBS/TSP-2, both endogenous inhibitors of angiogenesis [68, 69, 70]. Neither THBS/TSP-1 nor THBS/TSP-2 appears to have a major impact on developmental angiogenesis [71, 72]. Instead, the proteins are known to inhibit endothelial cell proliferation and reduce both the size and the number of blood vessels during wound healing and in tumors [68, 71, 72, 73]. The anti-angiogenic activity of THBS/TSP-1 is conferred by the conserved amino acid sequence known as type I repeats [74]. These repeats are also found in THBS/TSP-2 but not in THBS/TSP-3 and THBS/TSP-4 [73]. Interestingly, expression of THBS/TSP-1, -2, -3 and -4 was detected in the islets, with the expression of THBS/TPS-1 and THBS/TSP-4 being more pronounced. The expression of the anti-angiogenic THBS/TPSs, PEDF, PAI2 and PF1-4 could possibly explain the lack of an intra-islet capillary network, which is characteristic of a transplanted islet graft [45, 46, 47].
In summary, it can be envisaged that the balance between different pro-angiogenic and anti-angiogenic factors controls the de novo formation of islet capillaries. The present findings may be of importance for further exploration, aimed at finding a means of improving the revascularization process of transplanted islet grafts. For example, it may be possible not only to stimulate endothelial cell VEGF- and Tie-signaling, but also, by means of genetically or pharmacologically attenuating the expression of SERPINs, PF-4 and thrombospondins, to establish a satisfying new intra-islets capillary network after transplantation.
The authors thank Ing- Marie Mörsare for the valuable assistance. This research was supported by the Swedish Society for Medical Research, the Swedish Medical Research Council (12X-109, 12X- 11564, 72P-12995), the Swedish Diabetes Association, the Nordic Insulin Fund and the Family Ernfors Fund.