Claudine le Vaillant* and Mathilde Vital
Centre Hospitalier Universitaire de Nantes, Nantes cedex, France
*Corresponding Author:
Claudine le Vaillant
Centre Hospitalier Universitaire de Nantes, Nantes cédex, France
Tel: +33240083333
E-mail:claudine.le.vaillant@gmail.com
Received date: March 18, 2016 Accepted date: April 18, 2016 Published date: April 21, 2016
Citation: Le Vaillant C, Vital M. How it is Possible to Diagnose in Prenatal Low Form of Anorectal Malformations? Gynecol Obstet Case Rep. 2016, 2:1.
Keywords
Anal atresia; Prenatal diagnosis; Ultrasound; Magnetic resonance
Introduction
Anorectal malformations (ARMs) of all (high and low) forms concern about 1/5000 births. Low form is difficult to diagnose prenatally. Isolated forms are rare; they are often present in the context of multiple malformation syndromes. From a rare case of prenatal diagnosis, ultrasound findings will be discussed as well as the role of three-dimensional ultrasonography (3D) nuclear magnetic resonance (MRI) and biochemical study of the amniotic fluid.
Case Report
A 35-year-old woman, primigravida, was referred at 22 weeks of gestation for suspected multiple malformation syndromes with right renal abnormality, spinal lumbosacral abnormality right clubfoot and doubt as to the presence of an imperforated anus. The fetus male was eutrophic, with no abnormal external genitalia. There was no family history of congenital malformations or consanguinity. Ultrasound examination at 12 weeks of gestation has noted a nuchal translucency of 1.7 mm for a craniocaudal length of 53 mm with a combined risk of trisomy 21 estimated at 1/2777. The ultrasound examination confirmed abnormalities with hemivertebrae L4 and L5 (Figure 1and Figure 1b, and a horseshoe kidney to the left (Figure 1c and Figure 1d).
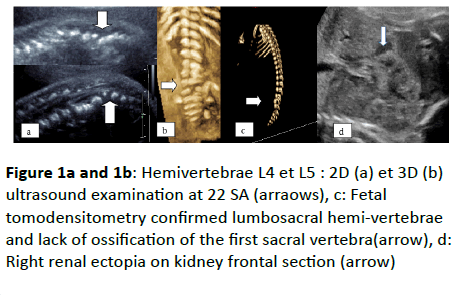
Figure 1a and 1b: Hemivertebrae L4 et L5 : 2D (a) et 3D (b) ultrasound examination at 22 SA (arraows), c: Fetal tomodensitometry confirmed lumbosacral hemi-vertebrae and lack of ossification of the first sacral vertebra(arrow), d: Right renal ectopia on kidney frontal section (arrow)
No cardiac abnormalities were noted. At 24 weeks of gestation, the anal canal was partially visualized; the anus was ectopic located in front under the scrotum (Figure 2) without classic ultrasonographic appearance of circular rim. Spinal cord appeared low implanted (Figure 3).
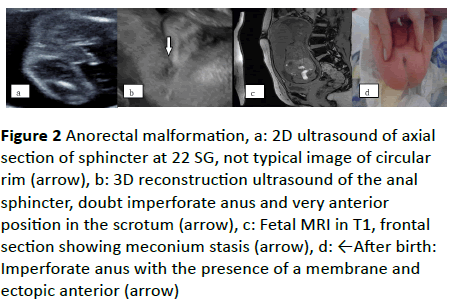
Figure 2 Anorectal malformation, a: 2D ultrasound of axial section of sphincter at 22 SG, not typical image of circular rim (arrow), b: 3D reconstruction ultrasound of the anal sphincter, doubt imperforate anus and very anterior position in the scrotum (arrow), c: Fetal MRI in T1, frontal section showing meconium stasis (arrow), d: ←After birth: Imperforate anus with the presence of a membrane and ectopic anterior (arrow)
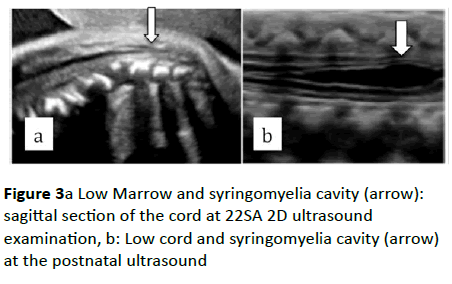
Figure 3a Low Marrow and syringomyelia cavity (arrow): sagittal section of the cord at 22SA 2D ultrasound examination, b: Low cord and syringomyelia cavity (arrow) at the postnatal ultrasound
Karyotype was 46 XY, measurement of digestive enzymes was normal in the amniotic fluid. Fetal tomodensitometry at 30 weeks of gestation confirmed lumbosacral hemi-vertebrae and a lack of ossification of the first sacral vertebra.
Fetal MRI showed a low terminal cone ending L2-L3, and cross right renal ectopia. A stasis meconium was visualized in the rectosigmoid without dilated bowel loops or signs for a rectovesical fistula, eliminating high anorectal malformation.
The patient delivered at term a boy weighing 3000 g. The clinical child examination showed a right clubfoot and an anterior ectopic imperforate anus under the scrotum. Ultrasound examination of the sacral spine showed a significant syringomyelia cavity facing L4 spinal cord was confirmed by a spinal MRI. A simple anoplasty was performed.
Discussion
We distinguish between high and low form of ARM depending on the level of disruption relative to the parietal insertion of the levator ani muscles. Antenatal diagnosis is essential in order to optimize perinatal care [1]. Despite advances in ultrasound technology, diagnosis of the isolated low form is only realized in 15% of the cases. In over 60% of the cases, it will be referred to indirect signs or associated malformations, such as renal and spinal abnormalities in 30% of the cases, and urogenital abnormalities in 40% [2]. RAMs is often associated with major fetal structural defects, for example, in the VACTERL association (vertebral anomalies, cardiac, tracheoesophageal, renal and lower extremity) or with trisomies but it may also be an isolated abnormality [2].
The anal sphincter can be visualized in prenatal ultrasound axial section of perineum from 22 weeks of gestation by a hypoechogenic circular rim with central echogenic center. Its size and its location relative to other pelvic organs, including external genitalia can be analyzed [3]. The sagittal and frontal sections of anal canal permit to estimate its length and its continuity until sphincter [3]. A lower bowel dilatation is an indirect sign of low-ARM, leading to study fetal anal sphincter. His absence or ectopic presence is highly suggestive of anomaly, as in our case. Discovered of enterolithiasis, signs a vesical fistula, that is part of most associated signs of high- MAR [4]. A cystic abdominal mass in a female fetus will have to search cloacal anomaly but also ovarian cysts and duodenal atresia [5].
The study of this clinical case is particularly interesting because of a complete prenatal imaging assessment. Sphincter abnormalities would be better analyzed with threedimensional ultrasonography with good reproducibility for measuring diameters of the sphincter and evaluation of its location by ultrasounds [6,7]. It also allows for the measurement of the length of the anal canal, and to evaluate its continuity but these measures require more experience because the junction area between the anal and rectal mucosa is difficult to see [6,7]. Three-dimensional ultrasonography would more accurately assess the level of stenosis and thus determine the type of ARM [8]. 3D reconstruction is used to specify the location of an ectopic anal sphincter, as in our case report. Fetal MRI is an excellent supplementary examination permitting to eliminate higher forms by identifying the level of anal stenosis, and confirming the presence of bowel dilatation and enterolithiasis [2,9]. It will also conduct a review of associated lesions. Measurement of digestive enzymes in amniotic fluid must be analyzed according to the term of pregnancy [10]. Indeed this rate is higher to 10 weeks of gestation, date of the opening of the anal membrane to the functional implementation of the sphincter to 20 weeks of gestation and then decreases, but in case of High-ARM fistula they will remain high. In our case, the dosage was normal at 25 weeks of gestation which allowed us to eliminate high-ARM. The care of these children is based on a surgical emergency treatment at birth. In low forms a simple anorectoplasty can be realized. Full recovery of continence is often possible unlike higher forms that require complicated surgical with a high risk of sequelae. The antenatal discovery of a high-ARM could discuss a medical termination of pregnancy.
Conclusion
Low-ARMs are good prognosis unlike highest forms whose consequences can cause a major handicap. Accurate prenatal diagnosis is essential in order to inform parents and optimize postnatal care. But screening low form needs a rigorous learning of two-dimensional ultrasonographic appearance of the fetal anal sphincter. His systematic review would increase the experience and therefore the sensitivity of the operator in case of pathological form. Other imaging techniques such as three-dimensional ultrasonography and MRI seem to have their place, only if sphincter anomaly is detected by ultrasound examination.
References
- Cuschieri A, EUROCAT Working Group (2001) Descriptive epidemiology of isolated anal anomalies: a survey of 4.6 million births in Europe. Am J Med Genet 103: 207-15.
- Cretolle C, Rousseau V, Lottmann H, Irtan S, Lortat-Jacob S, et al. (2013) [anorectal malformations] Arch Pediatr 20: 19-27.
- Moon MH, Cho JY, Kim JH, Min JY, Yang JH, et al. (2010) In-utero development of the fetal anal sphincter. Ultrasound Obstet Gynecol 35: 556-9.
- Vijayaraghavan SB, Prema AS, Suganyadevi P (2011) Sonographic depiction of the fetal anus and its utility in the diagnosis of anorectal malformations. J Ultrasound Med 30: 37-45.
- LeBorgne H, Philippe HJ, Le Vaillant C (2011) Contribution of three-dimensional ultrasonography in depicting perineal features in cloacal malformation. Fetal Diagn Ther 30: 239-40.
- Elchalal U, Yanai N, Valsky DV, Sela HY, Erez Y, et al. (2010) Application of 3-dimensional ultrasonography to imaging the fetal anal canal. J Ultrasound Med. 2010; 29: 1195-201.
- Gindes L, Weissmann-Brenner A, Achiron R, Tzadikevitch-Geffen K, Zajicek M, et al. (2012) 3 Dimensional demonstration of fetal anal canal and sphincter Ultraschall Med 33: E25-30.
- Guang Y, Wang X, Cai AL, Xie LM, Ding HL, et al. (2015) Evaluation of the development of the fetal anal sphincter with tomography ultrasonography imaging Ultrasound Med Biol 41: 40-6.
- Lubusky M, Prochazka M, Dhaifalah I, Horak D, Geierova M, et al. (2006) Fetal enterolithiasis: prenatal sonographic and MRI diagnosis in two cases of urorectal septum malformation (URSM) sequence. Prenat Diagn 26: 345-9.
- Bourdelat D, Muller F, Droullé P, Barbet JP (2001) Anatomical and sonographical. studies on the development of fecal continence and sphincter development in human fetuses. Eur J Pediatr Surg 11: 124-30.