What is known on this subject
• Sickle cell disease (SCD) is a genetic condition that affects about 2% of babies born in sub-Saharan Africa. Though SCD is common among African populations, governmental recognition of the condition as a public health problem has been slow.
• Healthcare provision for SCD patients requires a multidisciplinary team of well-trained professionals supported by a defined system of social support that caters to their physical, emotional, psychological and financial needs.
• Despite their diverse health and social need for survival, SCD patients have limited access to quality clinical, health educational, social and psychological care in most sub-Saharan countries.
• Screening of newborns for SCD is a relatively new phenomenon in sub-Saharan Africa, with very few countries, such as Ghana, Benin, Burkina Faso and Congo, conducting pilot interventions through heelprick and cord-blood methods. No national programmes for newborn screening so far exist in sub- Saharan Africa.
What this paper adds
• Expensive and technologically advanced methods of care for SCD patients in the UK, the USA and parts of Europe are generally not affordable in Ghana and other sub-Saharan African countries.
• Opportunities exist for the adoption of an affordable intermediate strategy that could be the model for the development of services both in Ghana and in other comparable African countries.
• A proposed strategic intermediate model provides a comprehensive package of basic services that are capable of improving the quality of life of patients.
Key words
African context, challenges, genetic condition, Ghana, healthcare provision, sickle cell disease
Introduction
Sickle cell disease (SCD) is a chronic inherited disease affecting millions of people worldwide. Generally in Ghana, as in most parts of tropical Africa, patients have limited access to quality clinical, health educational, social and psychological care. Clinical care in health facilities at the district level is basic and sometimes lacking, despite the fact that 25% of the population are carriers and 2% of all babies born, that is 1 in 50 live births, have a form of SCD (Dennis-Antwi, 1997; Ankrah-Badu et al, 2007). Furthermore, although statistics from Kumasi, which is the second largest city in Ghana and which has the first newborn screening programme in Africa, show a survival rate of over 95% among enrolled patients, screening of newborns has not been scaled up nationally. Health sector reforms started in the 1990s, with a long-term vision for growth and development that would propel Ghana from a low-income to a middle-income country by 2020. The reforms have largely focused on bridging the gaps in health inequalities, with specific emphasis on (i) reducing significantly infant, child and maternal mortality rates, (ii) increasing access to health services, especially in rural areas, and (iii) establishing a health system that is effectively re-oriented towards delivery of public health services. With the recent introduction of theMillennium Development Goals, critical attention has been given to maternal and child health, regenerative health, and nutrition, with limited attention to chronic and non-communicable diseases (Government of Ghana, 1997; Birungi et al, 2006).
Over the years, SCD has scarcely been regarded as a disease of any public health significance. The health status of children with serious genetic disorders such as SCD has been submerged in statistics on death from themajor childhood diseases in Ghana, such as malaria, acute respiratory infections and malnutrition-related conditions. Of the projected 14 200 babies born each year in Ghana with a form of SCD, most go undiagnosed and probably die young (Ohene-Frempong, 2007).
The purpose of this paper is to present a review of the available literature and reports on services for SCD care, primarily in more affluent countries such as the USA and the UK, and to relate these services to that available currently in a developing country such as Ghana. Ultimately, the authors suggest a model for SCD care in Ghana with a potential for adapting this to other African countries. The authors set out the paper by firstly providing an overview of the SCD condition and its epidemiology. Secondly, we describe the healthcare services accessible to patients in the USA, the UK, Ghana and selected African countries. We also briefly examine the economic context of SCD in Ghana as well as selected African countries. Thirdly, we reflect on optimal treatment and care regimes that have been proposed internationally.We note that, until recently, SCD in Ghana has received little attention from policy makers, and document some of the initiatives that have taken place with regard to service provision in the Ghanaian context. The costs of western models of treatment are set against the suggested priorities for Ghana advanced by international health policy analysts. Furthermore, the potential of health education for professionals, parents and patients is outlined, and the role of social support and support groups is discussed.
We then argue that Ghana, which is currently considering the establishment of a national neonatal screening initiative as part of a proposed national SCD programme, cannot afford to uncritically adopt the high-technological strategies of European and American medicine. Rather, the potential exists for Ghana to strategically model an intermediate strategy based on (i) public education and advocacy, (ii) genetic counselling and testing, (iii) premarital counselling, (iv) neonatal screening, and penicillin prophylaxis through an effective clinical care programme, (v) public and parental education, (vi) continual and specialist health worker training in haemoglobinopathies, (vii) social and financial support for families living with SCD and (viii) school-based teaching on SCD and inheritance patterns.
The review process for this paper takes into account the relevant and available research articles, books, and Ghana Government and partner/projects reports that have been published or compiled and made available electronically or through programme offices. Recent electronic journals dating from 2000–2009 were accessed through online collections of electronic databases such as ASSIA. These sources generated much of the literature on publications, and the time frame selected was to ensure up-to-date references on the subject. Grey literature, such as policy documents, project reports, PowerPoint presentations and research reports, compiled between 1995 when the newborn screening for the SCD project started in Kumasi, and the present, were accessed through the Ghana Health Service programme offices and project reports. Reference books and position statements provided information on SCD policies, genetics and the course of the disease and its sociological perspectives without any date limitations. These were accessed through the search engines of OPAC of De Montfort University Central Library and Newborn Screening for Sickle Cell Disease (NSSCD) local office collections. Countries or geographical areas of specific focus for the search were Ghana,West Africa, Africa, the UK and the USA. Other keywords used in the search process were ‘neonatal screening’, ‘newborn screening’, ‘support group’, ‘genetic counselling’ and ‘patient education’.
Sickle cell disease: definition and epidemiology
Sickle cell disease is caused by a mutation in the gene responsible for the � globin of normal adult haemoglobin A, leading to the production of sickle haemoglobin. SCD is a term embracing all disease states where the beta-S (�s) gene is present. New terminology recommended in the USA and used in the current National Heart, Lung, and Blood Institute’s Manual of Management of SCD lists these conditions as: SCD-SS, SCD-SC, SCD-SD, SCD-S�8 thalassaemia andSCD-S�+ thalassaemia(NHLBI,2002).Twenty- four per cent, nearly one in four, of the total population in sub-Saharan Africa carries either haemoglobin S trait or haemoglobin C trait (Konotey-Ahulu, 1991; Serjeant and Sergeant, 2001).
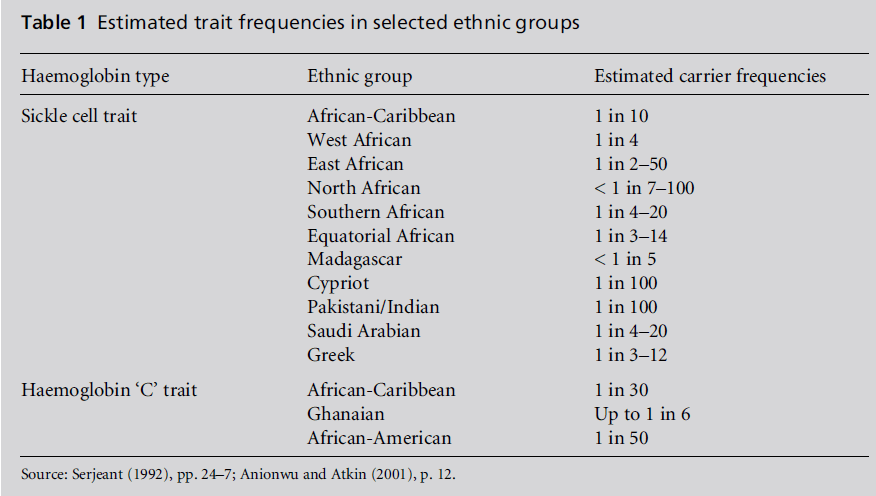
Historical accounts of the spread of the disease from tropical Africa to most parts of Europe, the Americas and the Caribbean attribute it to the Atlantic slave trade in the years 1451–1870, as well as the economic migration of Africans to these parts of the world. However, the journey of the �s gene is a little more complex and has given rise to five genetically distinct �s haplotypes: four African haplotypes – Senegal, Benin, Bantu, andCameroon – and the Arab-Indian haplotype. In Eastern Saudi Arabia, India and Sri Lanka, the Arab-India haplotype is predominant. An assessment of the prevalence of the disease worldwide shows that up to 300 000 affected children are born each year and that three-quarters of these are born in Africa (Okpala et al, 2002). In comparison, there are approximately 1 in 2500 live births with SCD in the USA (Therrell, 2006) and 1 in 2000 in the UK (Dick, 2006), annually. These comparisons clearly establish that the highest proportions of affected babies are to be found in Africa. The current estimates for Africa are probablymuchhigher than stated in the literature cited because in Nigeria, like Ghana, 2% of babies have SCD. With approximately 5 million births per year, Nigeria alone would have 100 000 babies with SCD a year. If the 2% rate of SCD is extrapolated over the 16 countries in the Economic Community of West African States (ECOWAS), with roughly 10 million births per year, West Africa alone would have 200 000 babies with SCD annually. Central Africa and parts of East Africa have an incidence of SCD similar to that of West Africa, while the northern and southern regions have lower incidence rates (Serjeant, 1992). In the year 2000, there were 29.6 million babies born in Africa (US Census Bureau in Ohene-Frempong, 2004). If only 1% of them had SCD, that would amount to 296 000 babies; if the overall SCD incidence rate for Africa was 1.5%, which is probably closer to the truth, thenumber of SCD babies would be 444 000 (United Nations Population Fund (UNFPA), 2008). Table 1 describes gene frequencies in selected ethnic groups, and marks out the distinct position of Africa as a continent that is epidemiologically and socially important in developing models of health and social care provision for SCD in view of the volume of births of children with SCD in Africa (Livingstone, 1976).
To date, the natural history of SCD has not been well studied in Africa, and accounts are subject to the reporting bias that those less affected are less likely to have contact with formal health services. Moreover, very few longitudinal studies have been carried out to assess SCD’s impact on public health in Africa (Ohene- Frempong and Nkrumah, 1994). The life expectancy of SCD patients in Africa remains largely unknown, although a study by Powars (1994) suggests that 90% of children born after 1980 in developing countries should reach at least 30 years of age based on observations made on a cohort study in Jamaica.Onthe other hand, the USA and Jamaica have survival estimates among patients indicating a range between 42 and 60 years in men and 48 and 60 years in women (Dick, 2006). In the UK, the lack of a comparable network of neonatal screening or major cohort studies has made such studies difficult to conduct (Ohene-Frempong and Nkrumah, 1994; Serjeant and Sergeant, 2001;Dick, 2006).
Table 1: Estimated trait frequencies in selected ethnic groups
Furthermore, in his study of a rural community in Africa, Fleming reported that less than 2% of the expected number of children with SCD survived beyond the age of 5 years (Fleming, 1989). This finding is attributable to two factors. First, in most parts of the developing world, including sub-Saharan Africa, not all deaths and births are recorded. Second, most of the population in countries such as Nigeria has limited access to formal systems of medical care, thereby limiting access to prompt and quality life-saving care (Orji et al, 2002). This subsequently leads to missed opportunities for recording the magnitude of disease incidence and prevalence. Consequently, vital statistics in Africa are mainly guesstimates derived from extrapolations, models and common sense, and are constrained by the need to avoid conflictwith previously published estimates. This makes it difficult to make a strong case for a disease like SCD to be recognised as one of public health importance.
Healthcare provision
In the USA and the UK, optimal management of SCD requires a holistic approach involving a multiplicity of professionals including paediatricians, physicians, haematologists, nurses, health educators, genetic counsellors, social workers, psychologists and many more. The management of sickle cell complications is centred on the administration of medicines such as antibiotics (to prevent or fight infections), analgesics and infusions. Daily administration of prophylactic penicillin, specifically for children between 2 months and 5 years, orally, twice daily, has been shown to significantly reduce infection due to Streptococcus pneumoniae (Dick, 2006). Patient and parental education coupled with genetic counselling are also pivotal in effective healthcare maintenance and future decision making related to childbearing.
As a starting point to extensively address the issues related to healthcare provision for SCD in Africa as well as a perspective on Ghana, it is worth reiterating that in western countries comprehensive care of SCD involves a model of healthcare based on interaction between medical and non-medical services and the affected people. This raises questions about the extent to which healthcare services in Ghana, or indeed Africa as a whole, could and should follow the same model. To address this question, we examined the healthcare system in Ghana and,where possible,Nigeria, Burkina Faso, Benin and the Democratic Republic of Congo, with respect to the facilities established for SCD management.
The Ghanaian context
Policies on SCD management
Since the inception of Ghana’s five-year programmes of work (POW) in the 1990s, the country has observed marked progress in the control of vaccine-preventable diseases such as poliomyelitis, measles, diphtheria and tetanus, while communicable diseases such as malaria, tuberculosis, HIV/AIDS and guinea worm are still prevalent, maternal, infant and child mortality are still very high, and non-communicable diseases are on the increase due to changes in lifestyle and nutrition (Aikins and Arhinful, 2006). The current and the third POW spanning 2007–2011 has adopted a preventive paradigm shift that seeks to address these challenges by creating wealth through health (Ministry of Health, 2007). A four-pronged strategy including promoting healthy lifestyles and healthy environments and increasing access to quality health and sustainable financing has been described to guide programme implementation. Though SCD management is implied in the strategy of improving quality health, it is not overtly discussed.
Generally, trends in healthcare in Ghana have, like those inmost African countries, lagged behind the developed world. However, the average life expectancy has increased from 45 to 57.5 years (Dick, 2006). The slow rate of change has been partly due to the fact that Ghana has a low gross domestic product (GDP) and a per capita income of US$1400 per annum in 2007 (Central Intelligence Agency (CIA), 2007). Ghana is heavily dependent on international financial and technical assistance, thereby making it difficult to institute effective and adequate services including healthcare for SCD patients (CIA, 2007). Understandably, the situation is different in most parts of Europe and the USA, where there are widespread SCD screening programmes on both universal and pilot bases. There are clear guidelines governing the screening and management of SCD. Ghana, likemost sub-SaharanAfrican countries, has no national policy, national guidelines for management, or national statistics on SCD, although nearly 25%, one in four, of the population are carriers and 2% of all babies born, 20 births per 1000 live births, have a form of SCD (Dick, 2006; Ohene-Frempong, 2007). So far, Ghana has initiated discussions about the establishment of a national SCD programme. These discussions began in earnest in 2006 with the selection of a national taskforce including Dr Jemima Dennis- Antwi (lead author) to develop policy guidelines for management. These guidelines were completed in October 2007 and are currently awaiting final adoption as a policy for dissemination and national use (Ankrah-Badu et al, 2007). In 2007, and for the first time, basic information on SCD was included in the national child health growth monitoring plan for mothers to be aware of SCD and the need for testing.
Historically, through the initiative of Dr FID Konotey-Ahulu in the 1960s, the first comprehensive SCD programme was established at Korle Bu Teaching Hospital inAccra to provide specialist care for all patients country-wide. This programme eventually progressed to become the Institute of Clinical Genetics in 1974 (Konotey-Ahulu, 1991). Until 1992, the facility in Accra was the only one established to cater for the healthcare needs of SCD patients. In the majority of Ghanaian children and those in other African states, SCD diagnosis is often delayed and made only after several visits to the hospital or clinic with acute illness, rather than as an early diagnosis through neonatal screening (Ohene-Frempong and Nkrumah, 1994; Rahimy et al, 2003; Tshilolo et al, 2008). Children who are fortunate to be living in urban and peri-urban communities are those who are likely to have access to better diagnostic services which are often based on solubility tests, though these are not sensitive enough to detect high levels of haemoglobin S (HbS). Solubility testing is a simple test often used in sub-Saharan African clinical care laboratories to detect the presence of HbS in blood. A chemical is added to the patient’s blood sample that reduces the amount of oxygen it carries. In those who carry even one sickle cell gene, some HbS will be present. However, these tests give inconclusive results as they do not distinguish between SCD and sickle cell trait (Anionwu and Atkin, 2001; American Association for Clinical Chemistry, 2006; Tshilolo et al, 2008).
Newborn screening for SCD
In 1992, the Komfo Anokye Teaching Hospital in Kumasi established an SCD programme as a requirement for thecommencement of the first-ever newborn screening for SCD programme in Africa. Heel pricks were used instead of the cord-blood sampling favoured in Benin (Rahimy et al, 2003). Efforts to introduce newborn screening in other parts of Africa have been documented in the Democratic Republic of Congo and Burkina Faso, with remarkable results (Tshilolo et al, 2008).
The Ghana project was set up as an international collaborative programme funded through a research grant by the US National Heart, Lung, and Blood Institute of the National Institutes of Health as part of the Comprehensive Sickle Cell Center of the Children’s Hospital of Philadelphia, with Ohene-Frempong (one of the authors) as principal investigator. Screening of newborns in Kumasi began in February 1995, and by December 2006 the project had successfully screened over 225 315 babies born within and around Kumasi and a rural community called Tikrom. Of these, over 4142 (2%) were found to have possible SCD (Ohene- Frempong, 2007). This project has been highly successful in demonstrating that newborn screening can be carried out successfully in a developing sub- Saharan country. Moreover, it has been instrumental in the provision of specialised healthcare to SCD patients in the Ashanti region and the rest of the regions beyond. However, screening is still performed only in Kumasi, thereby benefiting the few rather than the many. Consultative meetings with stakeholders to nationalise newborn screening are ongoing and are aimed at expanding services nationwide using a phased approach. The outcomes achieved so far are corroborated by statements on SCD at the 56th session of the 2006 World Health Organization (WHO) Regional Committee for Africa, and reflect an emerging interest by public health experts in national SCD management within the framework of national programmes for non-communicable disease prevention and control in Africa (World Health Assembly (WHA), 2006).
Facilities for SCD care
In Ghana, medical care for most patients with SCD nationwide is provided through the existing static health facilities found within the national, regional and district healthcare systems. SCD patients are seen as part of general outpatients, reporting daily at the health facilities except for those in Accra, Kumasi and a few public and private treatment centres mostly in the south and found in Tema, Koforidua, Agogo and Sunyani. Ghana and most sub-Saharan African countries are yet to establish holistic services for SCD care (Okpala et al, 2002). An overwhelming majority of these patients attend hospital with acute illness without necessarily having been diagnosed with SCD (Orji et al, 2002). The situation is different in theUKand the USA, where widespread universal or selective newborn screening programmes ensure early diagnosis and comprehensive management, with optimal outcomes (Hilliard et al, 2004).
Healthcare financing
For years after the 1957 independence, healthcare financing in Ghana was a huge burden for individuals and families. The system had for a long time been built on a fee for service and, despite government subvention to reduce the burden, the rising costs of care led to inequalities in healthcare financing and access. In the mid-1980s, Ghana adopted an exemptions policy towards reducing the burden of healthcare costs among the poor. This policy underwent several modifications to accommodate the complexities of institutionalising equal access, but there were many problems and complexities such as unclear implementation guidelines, high unpaid bills, delays in reimbursement, inadequate funding, and poor client perceptions (Government of Ghana, 2001; Aikins and Arhinful, 2006). Unfortunately,most SCDpatients, themajority ofwhom cannot afford their healthcare, were not included in the scheme due to the belief, bordering on fear, that if SCD patients were able to benefit as part of a standing policy, the cost of their care would consume the entire budget (Dennis-Antwi, 2006, 2008b).
According to Mcintyre et al (2006), recent international recognition of the burden of user fees instituted through the health sector reform initiatives of the 1980s and 1990s, and the difficulties of implementing exemption policies, culminated in mounting pressure on developing countries to introduce alternative approaches to healthcare financing, the most popular of which have been health insurance schemes. In 2003, the Ghana Government passed the National Health Insurance Act (NHIA) 650 to meet this international requirement, thereby bringing into law the establishment of various mutual health insurance schemes (Government of Ghana, 2003).
By January 2006, 107 out of 127 registered schemes had become operational. Currently, over 55% (11 million) of the population is covered by the scheme in various districts. Each scheme has a mandate to operate a minimum package that allows beneficiaries to access basic care at contracted healthcare facilities. Once registered, patients are entitled to the minimum package of services including outpatient, inpatient and specific services such as simple dental complaints and dental restoration. There is no special dispensation for SCD patients, and the extent of cover for complications of the disease is unclear. Another unexplained situation is the extent to which SCD patients with no source of income will benefit from the insurance facility and whether or not they will be considered for treatment under an exemption scheme. Reflecting on the situation in other sub-Saharan African countries, Ndiaye (2006) states that countries such as Guinea, Mali and Senegal have implemented mutual health insurance schemes at various levels, but these have invariably been on pilot and small-scale bases.
Health priorities and SCD
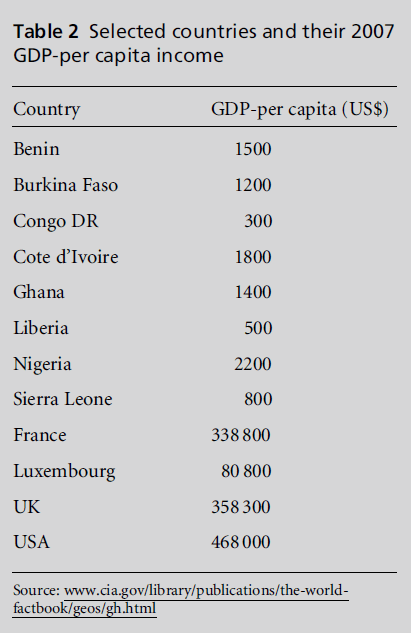
Overall, resource allocation for healthcare in most African countries is very low and highly skewed towards communicable diseases and reproductive health conditions that attract international support. Thus SCD resourcing is highly compromised, whereas the USA and Europe are globally reported to expend the most health resources on SCD (Karnon et al, 2000). A study by Karnon et al (2000), aimed at calculating the cost to the UK National Health Service (NHS) for patients with SCD, found that lifetime treatment costs ranged from about US$184 646 (£92 323) or US$15 387 per year to US$371 228 (£185 614) or US$30 935 per year. In comparison, in the USA costs were estimated at US$250 000 for a 60-year life-span, or over US$4000 a year (Anionwu and Atkins, 2001). It is evident from these findings that the high cost of the western model of care involving high-technology treatments such as bone marrow transplants is not an appropriate model for the African context, where GDPs and per capita incomes are generally low in comparison to the UK, the USA and other European countries, as shown in Table 2.
In isolation, a modest $4000 per year seems like an unaffordable amount for a developing country like Ghana. However, the relative costs of delivering the same healthcare are vastly different between Ghana and the UK or USA. According to Rahimy et al (2003), the most frequent reasons for hospital/medical consultation by SCD patients in Africa are fever/infection and pain. The management of the febrile patient in most African countries is intended to rule out bacterial infection and cover for possible malaria. This is now an entirely outpatient or home-based procedure for the majority of patients who do not appear ill (WHO, 2004). Patients are given two doses of ceftriaxone (Rocephin) and a three-day course of antimalarials which could cost less than $20 in Ghana and is now covered through the health insurance scheme (Ansong and Osei-Akoto, 2006). A 72-hour admission for pain management with intravenous hydration and opioid analgesics costs approximately $6000 at the Children’s Hospital of Philadelphia (Ohene-Frempong, 2006, unpublished data). The same care delivered at Komfo Anokye Teaching Hospital may cost less than US$50, as opioids are not the drugs of choice. These lower costs do not mean that they are affordable to the average Ghanaian patient, but they imply that the cost to the public health system or a national insurance health plan may not be as high as expected in theUKor theUSA. InGhana, the management ofSCDis primarily a low-tech affair. The routine drugs, folic acid and prophylactic antimalarials are quite cheap and easily accessible. The high technology comes in the new evaluation tools, such as magnetic resonance imaging and transcranial doppler ultrasonography (TCD), used to screen patients for the risk of stroke, that are recommended for most SCD treatment centres. Even TCD, apart from its initial capital cost of around $45 000 for the equipment and technical training, may cost less than $5 per test when administered in Ghana and other African countries.
Table 2: Selected countries and their 2007 GDP-per capita income.
Blood transfusion is another critical service that influences the prognosis of patients, especially those diagnosed with acute chest syndrome, aplastic crisis and acute cerebrovascular events (Dick, 2006). Comprehensive sickle cell programmes in most of the UK and the USA are structured to ensure that quality and prompt services are available to save patient lives (Anionwu and Atkin, 2001). The situation in most of sub-Saharan Africa is quite different. Blood transfusion services may be compromised with respect to their availability, the quality of the stocked blood and delays in blood typing and matching. This situation is further complicated by possible exposure to malarial and other blood-borne viral infections such asHIV and hepatitis (Tshilolo et al, 2008). Stem cell transplantation, though a proven cure for SCD, is not likely to be amajormodeof therapy in the near future unless there are major breakthroughs in the prevention of graft rejection and graft-versus-host disease. According to Mentzer et al (1994), although Africans tend to have larger families and a higher fullsibling compared to half-sibling rate, and therefore higher frequency of HLA-matched siblings, bone marrow transplantation will still be an unlikely prospect in the management of SCD in Africa in years to come.
The above considerations usually lead health policy analysts for developing countries to propose that it would be inappropriate for countries in Africa to try to import western high-tech-style health services (Mentzer et al, 1994; Ohene-Frempong, Children’s Hospital of Philadelphia, unpublished data, 2006). They suggest that countries in Africa should concentrate on those initiatives that epidemiologically contribute most to preventing premature deaths, such as vaccinations against communicable diseases, water and sanitation projects, reproductive health and supplementary feeding programmes for the lowest-income groups, etc. (UNICEF, 1987; Botha and Hendricks, 2008). For the most part we concur with such analyses. However, there are certain points, apart from its endemicity in Africa, that make SCD something of an exception, and these are discussed below.
Training of healthcare workers in SCD management
Regular training for health staff is paramount to quality management of SCD in Ghana. An important dimension is the extent to which healthcare workers in Ghana and other African countries are trained to care for SCD patients and their families. Healthcare workers are introduced to SCD as a haemoglobinopathy during their professional training as students. Experience in SCD management is often gained as part of on-thejob training. Continued medical and specialist training is limited to a few cities (Accra and Kumasi) and organised on an ad hoc basis. In a survey by Dennis- Antwi et al (1995) to assess the educational needs of health workers and consumers in Ghana prior to the institution of newborn screening for SCD in Kumasi, 22 doctors and 35 nurses in clinical care services were interviewed as part of the total sample. Findings indicated gaps in knowledge on SCD and its management, thereby necessitating the organisation of continued medical education for doctors and nurses to enable them to effectively manage patients and support the newborn screening programme. Training of health workers is therefore an integral and critical component of an SCD programme of care.
Moreover, SCD is a chronic condition for which there is no affordable cure applicable for the majority of patients. Providers of comprehensive care for people with SCD in the UK have stressed the benefit of educational information being provided in simple language by specially trained nurses. Such provision enables this crucial interaction to take place in a more relaxed environment than would be possible for a doctor to carry out. Unfortunately,mostpatients, whether inGhana or elsewhere in Africa, do not have access to such services because the majority of nurses do not have that special training. Specialist haemoglobinopathy training is not available in Ghana, but Nigeria is known to periodically organise short genetic counselling courses for nurses in SCD programmes (Sickle Cell Foundation, Nigeria, 2007; Dennis-Antwi, 2008a).
Parent and patient education
The success of any comprehensive care programme for SCD patients and their families is heavily reliant on parent and patient education. Several projects and programmes attest to this assertion (Anionwu and Atkin, 2001; Rahimy et al, 2003; Kafando et al, 2005; Tshilolo et al, 2008).Educational programmes are largely responsible for encouraging affected persons to be fully engaged with their care and to positively take control over their lives. Unfortunately, most of the SCDrelated information received by affected people in an organised and consistent way in Ghana is from the centres in Accra and Kumasi. In Kumasi, a comprehensive educational programme to support the newborn screening programme was established.Mothers of newborns identified as havingSCDare enrolled for continual education and counselling on healthcare maintenance for their babies. Public educational programmes are also organised to increase public awareness of SCD. This strategy is similar to that used in Benin, where a comprehensive education programme was set up to support parents as part of the newborn screening programme (Dennis-Antwi, 1997; Rahimy et al, 2003). However, in other parts of Ghana, limited programmes may be organised on an ad hoc basis at schools and in churches as part of individual or collective initiatives by health workers or community members. Annual campaigns involving the mass media and interpersonal interactive sessions are also held in the month of May to celebrate Africa Day for SCD, but these are one-off events with a limited and short-lived impact.
Social support services
So far, this paper has suggested that there is a limited provision for SCD management in Ghana and other African countries. Certainly the socio-economic conditions in most African countries restrict the allocation of resources to the health sector. The clinical picture of SCD tends to be much worse than that seen in the technologically developed countries (Ohene-Frempong and Nkrumah, 1994). Patients are reported to experience a lot of emotional and psychological stress in coping with SCD. This is particularly the case among adolescents who experience delays in physical growth and menarche. Victim blaming, ridiculing, dependence, labelling, isolation, hopelessness and helplessness, not to mention the stigmatising effects of some lay beliefs about the disease, are common (Telfair and Gardner, 2000; Dennis-Antwi, 2007a,b). It is also not uncommon to find that many SCD patients are poorly educated, unemployed and therefore, in the African context, unable to afford their care (Konotey-Ahulu, 1991; Anionwu and Atkin, 2001).
The role of social support groups in mitigating the social, psychological and emotional stress experienced by patients and families has been documented in the literature (Telfair and Gardner, 2000). Some support groups have been reported to play pivotal roles in seeking better services for patients, and are inseparable components of comprehensive care programmes in the UK and America (Anionwu and Atkin, 2001). In Africa, support associations exist in Ghana, Nigeria, Cameroun and Benin, and the West African group of associations has formed a Federation of African Associations of SCD popularly called FALDA (Fe´de´ration des Associations de Lutte contre la Dre´panocytose en Afrique). The Sickle Cell Association of Nigeria was reportedly resuscitated by Professor Akinyanju, a professor of medicine in 1984, starting in Lagos, and followed by the establishment of the Sickle Cell Foundation of Nigeria in 1994 (Sickle Cell Foundation Nigeria, 2007).
The Sickle Cell Association of Ghana (SCAG), a patient support group formed more than 20 years ago, was formally inaugurated as a non-governmental organisation in May 2004. Currently there are five branches of SCAG. The Sickle Cell Foundation of Ghana (SCFG), led by the third author (Ohene-Frempong) and founded in April 2004, aims to support the development of resources and services to improve the health and quality of life of people with SCD and related conditions. It has been involved in international and local initiatives to raise funds and increase focus on the disease. SCFG and SCAG are leading the effort to influence the government and the wider health sector to commit itself to the cause of people with SCD.
Sponsorship for SCD programmes
SCD is not on the priority list of the United States Agency for International Development, the UK Department for International Development, theDanishAgency for International Development or the Japanese Agency for International Cooperation, and therefore receives negligible international funding. Though the WHO has issued a statement urging governments to pay increasing attention to SCD by establishing national programmes, there has so far been minimal drive to translate such statements into policy and practice (World Health Assembly, 2006). While it is largely true that in poor developing countries the government is often the originator of all public health activities, there seems to be no reason why the provision of SCD services could not be initiated and possibly driven by a nongovernmental socially active organisation in consultation and collaboration with governments. In fact recent trends in healthcare delivery in Ghana would tend to suggest a de-emphasis of the role of government to some extent. Currently the concept of decentralisation guides national, regional, district and community initiatives through a sector-wide approach to healthcare (Aikins and Arhinful, 2006) driven more by the agenda of international funders. International funding for SCD can only occur if governments and partner organisations recognise SCD as a public health problem and advocate the involvement of international funding agencies as a means to reduce infant mortality in African countries.
SCD education in the formal sector
Another important social service worth commenting on is the lack of sickle cell education in the formal education sector. Nigeria has a population of over 120 million, with over 40% being of school-going age (United Nations, 2003). Ghana’s population, though small compared with that of Nigeria, is over 22 million, with over 12 130 primary schools, 5450 junior secondary schools and 503 senior secondary schools (Ghana Embassy in Japan, 2008). Benin and Burkina Faso have populations of around 6.7 million and 13 million, respectively (Encyclopedia of theNations, 2003). These statistics alone show that sub-Saharan Africa’s education sector presents as a critical sector with the opportunity for reaching young people and their teachers with basic information on SCD. Early exposure of African and Ghanaian youngsters and their teachers to SCD education will go a long way towards promoting better care for patients at school, publicinformed decisions about genetic testing and counselling, choice of partners, marriage, inheritance of SCD and SCD care (Dyson et al, 2007a). As part of her work with the NSSCD project in Kumasi-Ghana, Dennis-Antwi identified about 50 primary school teachers in Kumasi metropolis, assessed their SCD knowledge, and found that though they knew about SCD and had encountered pupils with the condition in their classes, their limited medical knowledge of the disease inhibited their ability to be supportive of such children in times of need. This finding informed the development of a brochure entitled Notes for the Teacher on the SCDChild at School,whichisavailablethroughthe project’s health education office. The notes guided the in-service training of primary school teachers of children identified, through the NSSCD project, as having SCD. Parents, through the support groups, were also encouragedto have personal copies to guide their interactions as the children changed classes and had new teachers. These activities served to develop a cohort of teachers who understood SCD and supported such children at school, with remarkable results, although they were limited to Kumasi. Lessons learnt on such a programme offer opportunities for upscaling nationally and regionally in Africa.
Genetic counselling and testing
A study of supportive services available for sickle cell management in most parts of theUSA, theUKand other parts of Europe suggests antenatal screening, neonatal screening, prenatal diagnosis, genetic counselling and testing are among pertinent services required for optimal living among affected families (Etorre, 2002). Antenatal and neonatal screening and prenatal diagnosis potentially offer the pregnant woman or nursing mother the opportunity to assess her sickle cell status, know that of her child, and make an informed decision about the baby or fetus (Dyson et al, 2007b). Nigeria has been able to conduct prenatal diagnostic services as part of a research-based study, but this is yet to be translated into a routine service for the public (Duronsimni et al, 1995). However, Nigeria has established regular genetic counselling courses for health workers to upgrade their knowledge (Sickle Cell Foundation, Nigeria, 2007). In Ghana, routine genetic testing for SCD was established through a study by Dennis-Antwi to be an unfamiliar concept to Ghanaian couples, though it has the potential to succeed if culturally acceptable approaches, such as recognition of significant others in genetic decision making, are employed (Dennis-Antwi, 2002, unpublished report). Genetic counselling and testing is widespread in western countries and is perceived as a private interaction by the couple involved, and it is considered to be a ‘good thing’ for carrier status to be widely known by people in the community before having a child or entering into marriage (Dyson et al, 2007b). Experiences gained in such programmes offer lessons for adaptation to the African setting.
Ghana has yet to institute a national policy to guide the establishment of genetic counselling and testing services, though recently an increasing number of church members in charismatic and Pentecostal denominations have opted to undertake premarital testing without the requisite formal genetic counselling to guide reproductive decision making (Etorre, 2002). These churches, with a perceived obligation to protect their congregations, have instituted premarital testing as part of their marriage counselling sessions. It is believed that the upsurge of HIV/AIDS in several countries has strongly influenced and contributed to the institution of mandatory testing as part of measures to curb its spread (Luginaah, 2005).
The observations made by Dennis-Antwi (2006, 2008b) through her study of fathers of SCD children and work with the NSCCD project in Kumasi have shown that often there is limited opportunity for the prospective couple to receive the appropriate interpretation of test results, as there is no structure within the healthcare system for supporting such initiatives. This is further complicated by the prospective couple’s decision to get married. At that point, information on their SCD genotypic status becomes irrelevant, or in extreme cases destructive to their emotional status and relationship. Some prospective couples who have had a previous experience with SCD have suspended marriage upon discovering their SCD status during their marriage counselling period. Married couples who have one or more childrenwith SCD would have wished for information on theirSCDstatus and genetic counselling during their courtship period, and this would have greatly influenced their choice of partner. Genetic counselling and testing therefore will be most beneficial to prospective couples if accessible programmes are instituted for young people during their school-going years, to ensure maximum relevance.
Conclusions and recommendations
From the experiences so far discussed in this paper, it can be said that though there is a high prevalence of SCDinmost parts of sub-SaharanAfrica,SCDis generally not perceived by African governments as a disease of public health importance. The management of patients so far has been basic. Special healthcare services for SCD patients are provided by few centres, leaving a large proportion of affected families to attend general outpatient services in district hospitals and health centres with limited knowledge about specialist SCD care. There are limited support groups to provide the much needed psychosocial support to affected patients and families. Genetic counselling services are generally lacking, although charismatic churches have policies for premarital testing, which raises certain challenges. SCD education for the public and within the formal education sector is generally lacking, thereby leaving the population largely uninformed. Healthcare financing is extremely limited outside the provision of the health insurance schemes, which do not cover the entire populace. However, public health professionals in both the public and private sectors have the potential to lead the effort to change the present trend towards a service that provides essential healthcare and basic social support to SCD patients and their families, if adequately trained. The concluding part of this paper suggests the setting up of a model for SCD provision in Africa, with a focus on Ghana.
Ghana: a model for SCD provision in Africa?
The reluctance to consider SCD as a priority may be due to it being falsely assumed that service developments for genetic diseases will necessarily be expensive, based on high technology, and hence ill advised in the Ghanaian context. However, if SCD management in Ghana and Africa received consideration and recognition for specialised clinical services, improved access to care and comprehensive social support services, people with SCD could lead relatively normal lives to achieve their highest potential, just like those without the condition.
SCD is a sizeable issue for Ghana and sub-Saharan Africa. We strongly suggest that it is possible to make considerable improvements in life expectancy and life quality by relatively modest investments in care. These include public education about SCD services and the establishment of facilities for genetic counselling and testing for young people, to guide reproductive decision making. The Ghana HIV/AIDS programme has established several counselling centres for HIV/AIDS. Culturally sensitive lessons learnt from their experiences and from other parts of Africa could inform the SCD effort. Alongside this, premarital genetic counselling and testing would assist prospective couples in making informed decisions about their marriage. Neonatal screening, coupled with penicillin prophylaxis, folic acid supplementation, vaccinations and antimalarial drugs, is essential. These improvements could be combined with patient and parental education about signs of acute illness and emergency care. Community and parental education would help to reduce stigma and improve home-maintenance skills. This education could be augmented by improved social support through a network of patient and family support groups and public education to increase public awareness about SCD and its inheritance patterns. Such networks could encourage people successfully living with SCD to present themselves as living testimonies to the fact that they can contribute to the economic and social well-being of Ghana. Research in the UK has stressed that people with SCD undertake work caring for others as well as being cared for themselves (Anionwu and Atkins, 2001).
These initiatives could be sponsored and coordinated, within the budget of the medium-income country that Ghana is aspiring to become, with the adoption and implementation of the drafted policy guidelines (Ankrah-Badu et al, 2007) as part of a national plan for health service provision for SCD. At the time of writing this paper, Ghana Health Services has initiated efforts through stakeholder consultations to establish a national SCD programme, with effect from 2009, using a gradual scale-up approach to newborn screening and comprehensive management based on experiences from the Kumasi project.
Recommendations for a model service
There are real solutions to the problems of people with SCD that are within the means of developing nations such as Ghana, Nigeria, Benin, Burkina Faso and Congo. The solutions can come neither from government alone, nor from the actions of individuals, but only through the combined efforts of stakeholders such as governments, healthcare providers, non-governmental organisations (NGOs), faith-based organisations, civil society organisations, community support groups, families and households. These proposed solutions are set out below.
1 Consistent and continual advocacy and educational programmes led by NGOs such as SCFG and supported by the Ghana Health Service could support and facilitate public education. This could provide recognition of the achievements of those living with SCD and help to change the public perception of SCD by demonstrating that individuals with the condition can become self-supporting and contribute to the social and economic life of Ghana. Advocacy at higher governmental levels is also critical in mobilising the necessary resources and political will to establish a comprehensive programme for people with SCD.
2 Through use of careful record keeping, stakeholders could also initiate media- and community-based advocacy campaigns that disseminate the success of the Kumasi-Ghana-based screening programmes. These could emphasise that 25% of all Ghanaians are in need of SCD services. These campaigns could also contrast the current estimated attrition of up to 98% of those born with SCD, with the 5% loss of life of those who have been enrolled in the Ghanaian screening programme. The screening programme as at December 2006 had diagnosed 4142 newborns with possible SCD, and enrolled 2914 (87.4%) of them in the clinic; 5% of the latter were known to have died. The remaining figure represents 2768 (95% of the 2914 babies with SCD enrolled), young babies who have been helped to survive to school age (Dennis-Antwi, 2006, 2008b). This represents 2768 parents who have not had to grieve for the loss of a child, and 2768 potential future contributors to theGhanaian economy. Benin, Burkina Faso and Congo could also use the data emerging from their screening programmes as the basis for advocating a national SCD programme.
3 Newborn and antenatal screening and follow-up services for SCD should be incorporated into the basic package of the Ghana health insurance scheme and integrated into the ongoing immunisation programmes in the public health service as a formal service. Currently, the Ghana Health Service, SCFG and the NHIS are discussing the establishment of such regulations. Moreover, providing a newborn screening service as an adjunct to immunisation services will ensure maximum success, as uptake of immunisation services in most African countries is high. However, effective systems of tracking and defaulter tracing will need to be instituted to ensure that all newborns identified as having SCD are enrolled into a clinical care programme. This would have far-reaching beneficial results for SCD services. Current SCD programmes in other African countries could assess the success so far of their existing health insurance schemes and determine appropriate mechanisms for using these as a platform for funding screening activities.
4 The Ghanaian Ministry of Health has in recent times given positive recognition to the existence of SCD, and has initiated stakeholder meetings to develop an active implementation plan for the management of SCD as recommended by the WHO. The SCD policy guidelines developed for the Ghana Health Service should receive optimal attention and quick approval (Ankrah-Badu et al, 2007). Work towards establishing treatment centres for SCD throughout the country should be a priority after the approval of the guidelines, coupled with the development and approval of standard treatment guidelines.
5 The financial costs of treatment for SCD are coupled with an associated lay notion of the condition as a metaphysical disease which is often perceived as spiritually induced by enemies of the couple to bring untold hardship and disgrace. This is currently a powerful tool militating against optimal care of SCD patients by families, as they tend to seek spiritual remedies for complications of the disease, rather than reporting for medical care. The cost of care cannot be borne by many families without help. The costs of frequent visits to healthcare institutions for routine care and acute illness management, frequent hospitalisation, medications, blood and other tests, and transportation far exceed the means of many families and may not be covered by the NHIS. These factors draw attention to the urgent need for a system of financial provision for SCD care that promotes family cohesion and financial commitment to caring for the affected child. As the first element of a strategy to underpin social and financial security for SCD care in Ghana, there is scope for SCAG and SCFG to negotiate with the Ghanaian Government NHIS for special arrangements to ensure maximum cover for frequent hospitalisation and complications of the disease.
A second element of a financial strategy would be for the SCFG to organise access to the existing community-based savings and credit schemes that abound in Ghana, and are called susu in a common local language. Families themselves could informally contribute and support each other as an informal system of credit scheme. Additionally, several savings and loans schemes have recently emerged on the financial market, and present opportunities for families to invest towards emergency healthcare needs outside the NHIS coverage.
A third element is the training or retraining of the unemployed patients or parents in incomegenerating activities to equip them with the skills to venture into self-sustaining economic initiatives. Thiswill help to mitigate the cost of care. The SCAG and SCFG could spearhead strategies aimed at accessing support from local and international organisations such as theOpportunities Industrialisation Centre (OIC) or the National Board for Small Scale Industries.
The development of specialist skills among health workers is an important dimension to optimal healthcare for SCD patients. It is recommended that SCD be the subject of more formal and continuing professional education in addition to basic information provided during initial professional training.
This continuing professional education should include both clinical and social aspects of care. Specialist training in haemoglobinopathies at a post-basic level should be established, and a system for using the specialist expertise set up to ensure effective deployment of their services to sickle cell centres, which should be established nationwide. Lessons from the Nigerian genetic counselling training programme could inform the process of setting up these centres.
7 Ghana’s education sector presents as a crucial opportunity for reaching a large number of young people with basic information on SCD as part of a national policy for genetic counselling and testing in basic formal education. SCD and the concept of genetics should be an integral part of primary education and high school curricula. West Africa adopts the same teaching curriculum for primary and high schools through the West African Examination Council. Therefore, once introduced, all pupils and students in West Africa stand to benefit, and the success stories could be beneficial to other parts of sub-Saharan Africa. Besides, teachers’ updated knowledge on SCD will promote a positive relationship with SCD patients and establish a sense of responsibility to ensure thewelfare ofSCDchildren during peer interactions (Dyson et al, 2007a).
Conclusion
This paper has argued that the quality of life for SCD patients in Ghana and sub-Saharan Africa could be enhanced by the following:
1 A strong social support network is important for SCD programmes in Ghana and Africa. So far, support networks for Ghana and other African countries mentioned are limited in coverage to a few cities and towns.
2 Expanding newborn screening services (NSSCD) in Ghana by the adoption of a gradual scale-up to other regions from Kumasi-Ashanti, coupled with quality patient data management and transfusion services. The development of rollout plans by SCFG to establish more SCD support groups, wherever NSSCD is introduced, will facilitate patient and family uptake of services, as well as management of psychosocial stress.
3 The role of specialist health workers and professional social workers in this effort cannot be overemphasised.
4 Diverse health financing schemes should be employed to facilitate uptake of health services in an affordable manner.
Acknowledgment
We wish to thank the Newborn Screening for Sickle Cell Disease in Ghana, Komfo Anokye Teaching Hospital, Kumasi-Ghana, and the Sickle Cell Association of Ghana – Ashanti Region Branch.
DEDICATION
To all patients and families living with sickle cell disease in Ghana and Africa.
CONFLICTS OF INTEREST
None.
Tables at a glance


Table 1
Table 2
References
- Aikins M and Arhinful DA (2006) Review of the ExemptionPolicy: a report of the Annual Health Sector Review. Accra:Ministry of Health.
- Anionwu EN and Atkin K (2001) The Politics of Sickle CellAnaemia. Buckingham: Open University Press.
- Ankrah-Badu G, Dennis-Antwi JA and Sackey SO (2007)Strategic Guidelines for Sickle Cell Disease Management inGhana. Compiled for the Ghana Health Service. Accra:Ghana Health Service.
- Ansong D and Osei-Akoto A (2006) A report on clinicalservices to SCD patients. Komfo Anokye Teaching Hospital,Kumasi, Ghana (unpublished data)
- American Association for Clinical Chemistry (2006) SickleCell Test. https://labtestsonline.org/understanding/analytes/sickle/glance-2.html
- Birungi HE, Addai G, Addico G et al (2006) Health SectorReforms in Ghana and Implications for Reproductive PrioritySetting. Frontiers Final Report. Washington, DC:Population Council.
- Botha C and Hendricks M (2008) Financing South Africa’sNational Health System Through National Health Insurance:possibilities and challenge. Cape Town: HRSCPress.
- Central Intelligence Agency (CIA) (2007) World Fact Book.www.cia.gov/library/publications/the-world-factbook/geos/gh.html (accessed 25 September 2008).
- Dennis-Antwi JA, Opoku SA, Osei-Amoh B et al (1995)Survey of educational needs of health workers and consumersin Ghana prior to the institution of newbornscreening for sickle cell disease in Kumasi. Health Courier5(4):28–32.
- Dennis-Antwi J (1997) Sickle cell disease in Ghana. AfricaHealth Journal 19(2):14–15.
- Dennis-Antwi JA (2006) The social meanings of sickle celldisease in Ghana: fathers’ reactions and perspectives. UnpublishedPhD thesis. Leicester: De Montfort University.Dennis-Antwi JA (2007a) Sickle Cell Disease in Ghana.
- Presentation made at Sickle Cell Symposium, Kofi AnnanCentre, Accra, Ghana, 25 October 2007.
- Dennis-Antwi JA (2007b) The social meanings of sickle celldisease: changing public perception. Daily Graphic (Ghana)5 July: 7.
- Dennis-Antwi JA (2008a) Choosing a career in nursing.Daily Graphic (Ghana) 19 April: 7, 9.
- Dennis-Antwi JA (2008b) The social meanings of a childwith sickle cell disease in Ghana: fathers’ reactions andperspectives. Disability and Society 23(5):415.
- Dick M (2006) Standards and Guidelines for ClinicalCare.For the UK Forum for Haemoglobin Disorders. London:NHS Sickle Cell and Thalassaemia Trust and Sickle CellSociety.
- Dyson SM, Atkin K, Culley LA and Dyson SE (2007a) Theeducational experiences of young people with sickle celldisorder: a commentary on existing literature. Disabilityand Society 22(6):581–94.
- Dyson SM, Cochran F, Culley LA et al (2007b) Observationand interview findings from the Ethnicity Questions andAntenatal Screening for Sickle Cell/Thalassaemia (EQUANS)Study. Critical Public Health 17(1):31–43.
- Durosinmi MA, Odebiyi IA, Adediran NO et al (1995)Acceptability of prenatal diagnosis of sickle cell anaemiaby female patients and parents of SCA patients in Nigeria.Social Science and Medicine 41(3):433–6.
- Encyclopedia of the Nations (2003) www.nationsencyclopedia.com (accessed 25 September 2008).
- Etorre E (2002) Reproductive Genetics, Gender and the Body.London: Routledge.
- Fleming AJ (1989) The presentation, management andprevention of crisis in sickle cell disease in Africa. BloodReview 3:18–28.
- Ghana Embassy in Japan (2008) The Educational System inGhana. www.ghanaembassy.or.jp/educational.html (accessed25 September 2008).
- Government of Ghana (1997) Ghana Vision 2020: the firstmedium term development plan 1997–2000. Accra: NDPC.
- Government of Ghana (2001) The Health of the Nation:reflections on the first five year health sector programme ofwork. Accra: MOH/GHS.
- Government of Ghana (2003) The National Health InsuranceAct. Act 650. Accra: Assembly Press.
- Hilliard LM, Maddox MH, Tang S and Howard TH (2004)Development of a regionalised, comprehensive networkfor paediatric SCD to improve access to care in rural state.Disease Management and Health Outcomes 6:393–8.
- Kafando E, Sawadogo M, Cotton F et al (2005) Neonatalscreening for SCD in Ouagadougou-Burkina Faso: a pilotstudy. Journal of Medical Screening 12(3):112–14.
- Karnon J, Zeuner D, Ades E et al (2000) The effects ofneonatal screening for SCD on lifetime treatment costsand early deaths avoided: a modelling approach. Journalof Public Health Medicine 22(4):500–11.
- Konotey-Ahulu FID (1991) The Sickle Cell Disease Patient.London: Macmillan.
- Livingstone FB (1976) Epidemiology of sickle cell disease – aglobal problem. In: Scott RB (ed.) International Aspects ofSickle Cell Disease. Proceedings of the International Conferenceon Sickle Cell Disease. A World Health Problem.Washington, DC.
- Luginaah IN, Yiridoe EK and TaabazuingMM(2005) Frommandatory to voluntary testing: balancing human rights,religious and cultural values, and HIV/AIDS preventionin Ghana. Social Science and Medicine 61(8):1689–1700.
- McIntyre D, Thiede M, Dahlgren G and Whitehead M(2006) What are the economic consequences for householdsof illness and of paying for health care in low andmiddle income country contexts? Social Science andMedicine 62:858–65.
- Mentzer WC, Heller S, Pearle PR et al (1994) Availability ofrelated donors for bone marrow transplantation in sicklecell anemia. American Journal of Pediatric Hematology/Oncology 16:27–9
- Ministry of Health (2007) The National Health Policy:Creating wealth through health. Accra: MOH.National Heart, Lung, and Blood Institute (2002) The Managementof Sickle Cell Disease (4e). NIH: Bethesda, p. 2.
- Ndiaye P (2006) Development of mutual health insurance inAfrica: a comprehensive analysis. Canada: InternationalDevelopment Research Center. Research Matters. www.idrc.ca/uploads/user-S/11539235471Pascal_Ndiaye_-_ Mutuelles_-_juin_2006.pdf
- Ohene-Frempong K and Nkrumah FK (1994) Sickle celldisease in Africa. In: Embury SH, Hebbel RP, MohandasN and Steinberg MH (eds) Sickle Cell Disease: basic principlesand clinical practice. New York: Raven Press Ltd,pp. 423–35.
- Ohene-Frempong K (2004) The Sickle Cell Foundation ofGhana: Mission Statement. Prepared for the Inaugurationof Foundation, Accra, Ghana, April 2004 (unpublished).
- Ohene-Frempong K (2007) Sickle Cell Disease Services inGhana. Presentation to the Joint WHO and TIF Meetingon Haemoglobin Disorders, Hilton Park Hotel, Nicosia,Cyprus, 16–18 November 2007.
- Okpala I, Thomas V, Westerdale N et al (2002) The comprehensivecare of sickle cell disease. European Journal ofHaematology 68:157–62.
- Orji EO, Ogunlola IO and Onwudiegwu U (2002) Broughtin maternal deaths in south-west Nigeria. Informa Healthcare4:385–8.
- Powars DR (1994) Natural history of disease: the first twodecades. In: Embury SH, Hebbel RP, Mohandas N andSteinbergMH(eds) Sickle Cell Disease: basic principles andclinical practice. New York: Raven Press Ltd.
- Rahimy MC, Gangbo A, Ahouignan G et al (2003) Effect of acomprehensive clinical care program on disease course ina sub-Saharan setting. Blood 102:834–8.
- Serjeant GR (1992) Sickle Cell Disease (2e). Oxford: OxfordUniversity Press.
- Serjeant GR and Sergeant BE (2001) Sickle Cell Disease (3e).Oxford: Oxford University Press.
- Sickle Cell Foundation Nigeria (2007) Eighth GeneticCounselling Training Course. https://sicklecellfoundation.com (accessed 25 September 2008).
- Telfair J and Gardner M (2000) Adolescents with SCD:determinants of support group attendance and satisfaction.National Association of Social Workers 25(1):43–50.
- Therrell BL and HannonWH(2006) National Evaluation ofUS Newborn Screening System Components. MentalRetardation and Developmental Disabilities (MRDD) ResearchReviews 12:236–45.
- Tshilolo L, Kafando E, Sawadogo F et al (2008) Neonatalscreening and clinical care programmes for sickle celldisorders in sub-Saharan Africa: lessons from pilot studies.Public Health 122(9):933–41.
- World Health Assembly (2006) Sickle Cell Anaemia. WHA59.20 Document Agenda Item 11.4. 59th Annual Meetingof the WHA, Geneva, 26 May 2006.
- UNICEF (1987) Accra, Ghana. In: Cornia GA, Jolly R andStewart F (eds) Adjustment with a Human Face. Volume 2.Ten country case studies. Oxford: Clarendon Press, pp. 93–125.
- United Nations (2003) Nigeria Population. www.nationsencyclopedia.com (accessed 25 September 2008).
- United Nations Population Fund (UNFPA) (2008) State ofthe World Population. Reproductive facts sheet. www.unfpa.org (accessed 25 September 2008).
- WHO/RBM/UNICEF/UNDP/WB (2004) Scaling up Home-Based Management of Malaria: from research to implementation.Geneva: WHO/HTM/MAL.