Original Article - (2016) Volume 17, Issue 6
Division of 1Medical Oncology, Cancer Research Institute, Department of 2Radiology, Kanazawa University, Kanazawa, Japan
Department of 3Internal Medicine, Fukui Saiseikai Hospital, Fukui, Japan
Received Date: July 13th, 2016; Accepted Date: August 19th, 2016
Objectives The concept of early chronic pancreatitis was initially described in the 2009 Japanese diagnostic criteria for chronic pancreatitis. However, the clinical features of non-alcoholic early chronic pancreatitis have not been defined. The aim of this study is to elucidate the clinical features of non-alcoholic early chronic pancreatitis. Methods Thirteen patients, two men and 11 women were diagnosed with non-alcoholic early chronic pancreatitis by revised 2009 criteria. Their clinical features, including their radiologic and endoscopic ultrasonographic findings and clinical courses, were retrospectively reviewed. Results Ten (77%) patients presented with back pain in addition to upper abdominal pain. Pancreatic enzymes were elevated in the sera of seven (54%) patients. EUS findings included lobularity with honeycombing in one (8%), lobularity without honeycombing in seven (54%), hyperechoic foci without shadowing in 10 (77%), strands in 13 (100%), and hyperechoic main pancreatic duct margin in 11 (85%). Neither contrast-enhanced computed tomography nor magnetic resonance imaging/ magnetic resonance cholangiopancreatography showed definite abnormalities in the pancreatic parenchyma. However, magnetic resonance cholangiopancreatography showed evidence of pancreaticobiliary maljunction and pancreatic divisum in one patient each. Camostat mesilate and high titer pancreatic enzyme pancrelipase were effective for relief of pain in all the patients except for two. However, the nine patients followed up for several years showed no improvement in endoscopic ultrasonographic findings. Conclusions Endoscopic ultrasonography may be useful in diagnosing non-alcoholic early chronic pancreatitis, even though computed tomography and magnetic resonance imaging could not detect abnormalities. Medication was effective for relief of pain, but did not improve endoscopic ultrasonographic findings for non-alcoholic early chronic pancreatitis. Further investigations are necessary to identify methods to prevent non-alcoholic early chronic pancreatitis progression.
Pancreatitis, Chronic; Endosonography; Cholangiopancreatography, Magnetic Resonance; Camostat; Pancrelipase
CM camostat mesilate; FD functional dyspepsia; MPD main pancreatic duct; NAECP non-alcoholic early chronic pancreatitis; PBM pancreaticobiliary maljunction; PD pancreatic divisum
Chronic pancreatitis (CP) is considered an irreversible progressive chronic inflammatory disease. In addition to pain, patients subsequently develop exocrine and endocrine insufficiencies.
Major risk factors predisposing to CP have been categorized using the TIGAR-O classification as (1) toxicmetabolic (e.g. alcohol consumption, hypercalcemia, and dyslipidemia), (2) idiopathic, (3) genetic, (4) autoimmune, (5) recurrent and severe acute pancreatitis, and (6) obstructive (e.g. pancreatic divisum (PD) and sphincter of Oddi disorders) [1]. Main etiology of non-alcoholic CP includes idiopathic, genetic, and obstructive. In Japan, alcohol consumption (69.7%) was the most frequent cause of CP, followed by idiopathic CP (21.0%), although the latter is the most frequent cause of CP in Japanese females [2].
Although advanced stage CP can be diagnosed easily, the clinical diagnosis of early stage CP remains difficult. The concept of early chronic pancreatitis (ECP) was initially defined in the 2009 Japanese diagnostic criteria for CP [3]. These criteria include the use of endoscopic ultrasonography (EUS) as the diagnostic modality to detect ECP. Early diagnosis and therapeutic intervention may improve the long-term prognosis of CP patients. The criteria include: (i) characteristic imaging findings, (ii) characteristic histological findings, (iii) repeated upper abdominal pain, (iv) abnormal levels of pancreatic enzymes in serum and/or urine, (v) abnormal pancreatic exocrine function, and (vi) continuous heavy drinking of alcohol equivalent to ≥80 g/day pure alcohol [3]. Imaging findings diagnostic of ECP on EUS included more than two of the following seven features, with at least one of features 1–4: (1) lobularity with honeycombing, (2) lobularity without honeycombing, (3) hyperechoic foci without shadowing, (4) strands, (5) cysts, (6) dilated side branches, and (7) hyperechoic main pancreatic duct (MPD) margin, or irregular dilatation of more than three duct branches on endoscopic retrograde cholangiopancreatography (ERCP). ECP was diagnosed in patients with more than two of items iii–vi, above, along with imaging findings, and was suspected in patients with item iii or iv, along with imaging findings, but only after ruling out other pancreatic diseases.
CT shows stones in the pancreatic ducts, pancreatic calcification, MPD dilatation, and parenchymal atrophy in patients with advanced stage CP [3, 4, 5], suggesting that CT is useful for the diagnosis of advanced stage CP [4, 5]. MRI has been reported to be more sensitive than CT for the assessment of CP because MRI detects not only morphological changes but the presence of fibrosis [6, 7]. The Cambridge classification, in which CP was graded by pancreatography, was proposed in 1984 [8]. ERCP classifications include side-branch pathology not previously noted on CT, an earlier feature of the disease [9]. However, pancreatography has been reported to provide poor diagnostic accuracy in patients with mild CP as compared with EUS [10, 11].
EUS has been reported useful for the diagnosis of CP [11, 12, 13, 14, 15]. According to traditional criteria, CP was graded by the number of EUS findings and compared with the Cambridge classification of pancreatography results [11, 13, 14]. However, discrepancies in diagnosing CP were observed because of the differences in severity of EUS findings, such as calcification and strands. New EUS-based criteria for the diagnosis of CP (Rosemont classification) were proposed in 2009, in which CP was graded by the severity of EUS findings [10]. Based on the Rosemont classification, ECP was newly defined in the 2009 Japanese diagnostic criteria for CP [3].
Evidence-based 2015 clinical practice guidelines for CP have indicated that the priority in the compensated phase should be to prevent repeated relapses and pain [16]. This includes dietary therapy to prevent excessive stimulation of the pancreas and pharmacotherapy, primarily with protease inhibitors. Elemental diets were also recommended for pain management in patients with CP [16, 17].
The clinical features of non-alcoholic ECP (NAECP) have not been sufficiently known. This study retrospectively investigated clinical features of NAECP.
Among the 818 patients who underwent EUS between 2007 and 2015 in our hospital and were not heavy drinkers, five (i.e. Patients 1, 5, 6, 11 and 13) were diagnosed with NAECP and eight (i.e. Patients 2, 3, 4, 7, 8, 9, 10 and 12) were suspected of having NAECP in the 2009 Japanese diagnostic criteria for CP [3] (Table 1).

The clinical features of these 13 patients were retrospectively reviewed. Baseline factors evaluated included symptoms (upper abdominal pain and back pain); serum levels of pancreatic enzymes (amylase (normal range, 40-113 IU/L), lipase (normal range, 11- 53 IU/L), and Elastase-I (normal range, <300 ng/dL)); imaging findings on EUS, computed tomography (CT), and magnetic resonance imaging (MRI)/magnetic resonance cholangiopancreatography (MRCP). Treatment-associated factors included changes in symptoms, serum levels of pancreatic enzymes and EUS results.
ETHICS
The written informed consent was obtained from each patient and the study protocol conforms to the ethical guidelines of the “World Medical Association Declaration of Helsinki - Ethical Principles for Medical Research Involving Human Subjects” adopted by the 18th WMA General Assembly, Helsinki, Finland, June 1964 and amended by the 59th WMA General Assembly, Seoul, South Korea, October 2008, as reflected in a priori approval by our institutional review committee.
The 13 patients included two men and 11 women, of mean age 50.5 years (range, 26–75 years), with five being <40 years old. All 13 patients denied being heavy consumers of alcohol. The mother of Patient 10 had recurrent pancreatitis, suggesting that Patient 10 had familial pancreatitis, although genetic examination was not performed. MRCP and ERCP diagnosed pancreaticobiliary maljunction (PBM) in Patient 1 and PD in Patient 11 as described below. Etiologically, these factors were regarded as responsible for CP in Patients 1, 10, and 11. In contrast, the remaining 10 patients were suspected of having idiopathic CP (Table 1).
Twelve (92%) patients presented with upper abdominal pain, including 10 (77%) with accompanying back pain. These symptoms were exacerbated by fat diet and overeating. Eleven patients (i.e. all but Patients 2 and 13) presented at the hospital because of upper abdominal and/or back pain. None of the 13 patients presented with steatorrhea due to pancreatic exocrine insufficiency.
Concentrations of three pancreatic enzymes (amylase, lipase, and Elastase-I) were measured in patient serum as listed in Table 1. At least one enzyme was elevated in seven (54%) patients. None of these 13 patients, however, showed decreased pancreatic enzymes in serum, and none underwent test of pancreatic exocrine function, para-aminobenzoic acid (BT-PABA) test. None of these 13 patients was diagnosed with diabetes mellitus.
EUS findings of normal pancreas, calcified alcoholic pancreatitis, and ECP are shown in Figure 1, respectively. EUS showed lobularity with honeycombing in one patient (8%), lobularity without honeycombing in seven (54%), hyperechoic foci without shadowing in 10 (77%), strands in 13 (100%), and hyperechoic MPD margin in 11 (85%) of 13 patients with ECP in this study. Three patients each had two findings of these EUS criteria, four each had three, and six each had four, respectively. None of these patients had cysts, side branches, hyperechoic foci with shadowing, MPD calculi, irregular MPD contours, or MPD dilatation (Table 2).
Figure 1: Endoscopic ultrasonographic (EUS) findings in pancreatic body/tail. Normal pancreas, showing (a). homogenous and finely reticular pattern in parenchyma, (b). no dilatation of main pancreatic duct (MPD) (arrow) and side branches, and no hyperechoic MPD margin. Calcified alcoholic pancreatitis, showing (c). pancreatic stones (arrowheads) and lobular out gland margin (arrows). Early chronic pancreatitis (ECP), showing (d). lobularity without honeycombing (circle), (e). hyperechoic foci without shadowing (arrows), (f). strands (arrows), and (g). hyperechoic MPD margin (arrows).

None of these 13 patients showed definitive abnormalities in the pancreatic parenchyma and pancreatic ducts on contrast-enhanced CT. Eight patients underwent MRI, which showed normal signal intensity in the pancreas on fat-suppressed T1 and T2-weighted images and uniform enhancement on the capillary phase. Although MRCP showed no irregular dilatation of MPD and side branches, it presented with findings indicative of PBM (Patient 1) and PD (Patient 11) in one patient each, respectively (Figure 2a, b). PBM was confirmed by ERCP in Patient 1 (Figure 2c), who underwent prophylactic cholecystectomy for the risk of gallbladder cancer.
Figure 2: (a). Magnetic resonance cholangiopancreatography (MRCP), showing a pancreaticobiliary maljunction (PBM) (arrow). (b). MRCP showing pancreatic divisum. Also observed was a dominant dorsal pancreatic duct (Santorini duct) crossing the lower bile duct (arrow). (c). Endoscopic retrograde cholangiopancreatography (ERCP) showing abnormal union between the pancreatic and bile ducts (common bile duct flowing to main pancreatic duct), suggesting PBM (arrow).
Twelve patients were initially treated with the protease inhibitor, camostat mesilate (CM; 300~600 mg/ day). Eight patients were secondarily administered the antispasmolytic agent flopropion (120~240 mg/day), but four patients discontinued due to lack of efficacy or adverse events, such as thirst or constipation. Six patients received conventional pancreatic enzymes secondarily; due to lack of efficacy, four patients were switched to the high titer pancreatic enzyme pancrelipase, and two patients were treated with pancrelipase de novo. After medication, numbers of numerical rating scale (NRS) as an indicator of pain (ranging from 0–10) decreased, and upper abdominal and/ or back pain were relieved in nine (82%) of the 11 patients with NAECP who suffered from them (Table 3). Pain relief was observed within 3 months in almost these 11 patients.

Serum levels of pancreatic enzymes were not altered in the six patients with normal levels before treatment. In contrast, of the seven patients with elevated serum levels of pancreatic enzymes, four showed reductions to normal ranges.
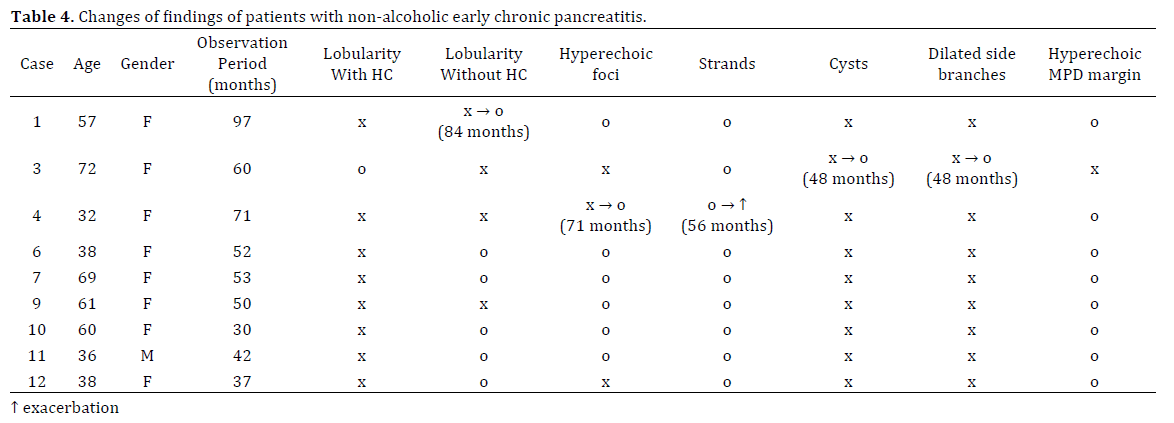
Nine (69%) of the 13 patients were followed-up by EUS for 2 to 8 years. Over this time period, Patient 1 developed lobularity without honeycombing, Patient 3 developed cysts and dilated side branches, and Patient 4 developed hyperechoic foci, as well as strands deterioration. These EUS findings emerged after more than 4 years. Follow-up EUS findings after two or more years showed exacerbation of NAECP in these three patients, equivalent to initial EUS findings in six patients. Improvements in EUS findings were not observed. However, all patients in this study fulfilled the criteria for neither definite nor probable CP during the clinical courses of their disease (Table 4).
Although the clinical diagnosis of advanced stage CP is easy, diagnosis of early stage CP remains difficult. The concept of ECP was first established to improve long-term prognosis in the 2009 Japanese diagnostic criteria for CP [3]. Because the clinical features of ECP have remained unclear, this study retrospectively investigated the clinical features of NAECP.
CT has been reported superior to ERCP in detecting parenchymal changes associated with advanced stage CP [9]. However, CT has insufficient diagnostic ability in early stage CP [5, 18, 19], showing no definitive abnormalities in the pancreas of all 13 patients diagnosed with NAECP by EUS in this study.
Most pancreatologists have considered ERCP the gold standard for morphological diagnosis and staging of CP in the absence of histopathology. ERCP findings were found to correlate with histopathology in 23 (74%) of 31 patients [20]. Pancreatography has been reported to provide poor diagnostic accuracy in patients with mild CP, although it was more accurate in those with moderate and severe CP [10, 11]. Pancreatography alone would be insufficient for diagnosis of CP. In addition, the incidence rates of post ERCP complications (e.g. pancreatitis, bleeding, and infection) have been reported to be 6–7% [21].
EUS has been reported useful for the diagnosis of CP [11, 12, 13, 14, 15]. Rosemont classification was proposed in 2009 [10], and ECP was newly defined in the 2009 Japanese diagnostic criteria for CP [3] based on the classification. These criteria include the use of EUS as the diagnostic modality to detect ECP. EUS findings in ECP are consistent with indeterminate CP according to the Rosemont classification. Excellent agreement was observed between EUS and ERCP in the diagnosis of CP, except for mild changes on EUS [11, 13]. However, this study could not compare the sensitivity of ERCP and EUS in diagnosing ECP, because ERCP was seldom performed. In this study, EUS findings frequently observed included strands (100%), hyperechoic MPD margin (85%), hyperechoic foci without shadowing (77%), and lobularity without honeycombing (54%). Hyperechoic foci have been reported to correspond to focal fibrosis, strands to bridging fibrosis, lobularity to interlobular fibrosis, and hyperechoic MPD margins to periductal fibrosis [22, 23]. These EUS findings are thought to be crucial for diagnosis of ECP, with correlations between EUS findings and histopathology reported in patients with CP [24, 25]. Parenchymal changes are thought to precede ductal changes in CP [26]. Based on their abilities to diagnose ECP and complication rates, EUS should be recommended over ERCP in the diagnosis of ECP.
MRI in patients with CP shows diminished signal intensity on fat-suppressed T1-weighted images, suggesting loss of aqueous proteins within the pancreatic acini. In addition, MRI shows diminished parenchymal enhancement on capillary phase images, suggesting disruption of the normal capillary bed and increased chronic inflammation and fibrosis [6, 7, 27, 28]. However, none of the eight patients with NAECP diagnosed by the 2009 Japanese clinical diagnostic criteria for CP showed definitive abnormalities in the pancreas. These findings indicate that EUS is more sensitive than CT or MRI for the diagnosis of ECP.
MRCP findings in CP include biliary and pancreatic ductal dilatation, strictures, irregularities in MPD, sacculation, and ectasia of side branches [9]. MRCP in patients with early stage CP often show a normal MPD with dilated and irregular side branches [6, 9, 27, 29]. In this study, MRCP showed normal MPD and side duct branches in all 8 patients with NAECP diagnosed by the 2009 Japanese diagnostic criteria. However, MRCP yielded results suspicious for PBM and PD in one patient each. PD is known to cause pancreatitis [2, 30, 31]. MRCP shows a dominant dorsal pancreatic duct (Santorini duct) crossing the lower bile duct and draining to the minor papilla, with no communication between ventral and dorsal pancreatic ducts, indicating PD. Furthermore, PBM has been reported to be the cause of pancreatitis [32, 33, 34]. MRCP was found to show an anomalous union between the common bile duct and the pancreatic duct, as well as the presence of a long common channel [34, 35, 36]. In the cases suspicious of having NAECP, MRCP may be recommended to detect the etiology of ECP.
In evidence-based 2015 clinical practice guidelines for CP, dietary therapy and pharmacotherapy, primarily with protease inhibitors in addition to elemental diets were also recommended for pain management in patients with CP [16, 17]. CM has been reported to attenuate dibutyltin dichloride-induced pancreatic fibrosis in rats by inhibiting the activity of monocytes and pancreatic stellate cells [37]. CM was also found to be effective against dyspepsia associated with non-alcoholic mild pancreatic disease [38]. Based on these findings, our patients with NAECP were initially treated with CM; however, CM alone had an insufficient effect on pain relief in most patients. Subsequently, these patients were treated with flopropion and/or pancreatic enzymes. Flopropion was discontinued due to insufficient efficacy or adverse effects in 50% of patients treated with this agent. Although pancreatic enzyme replacement therapy has been reported effective for pain relief in patients with CP [39, 40], conventional pancreatic enzymes were ineffective in 50% of patients treated with these agents.
Pancrelipase is an enteric-coated, delayed-release pancreatic enzyme, with 6-to 9-fold greater enzymatic activity than conventional pancreatic enzymes. Pancrelipase is used to treat patients with CP and those who have undergone pancreatic surgery [41, 42]. In this study, pancrelipase was effective in reducing pain in almost all patients treated with this agent. These findings suggest that combinations of pancrelipase and CM may be recommended for pain reduction in patients with NAECP.
Although medication was effective for pain relief, EUS findings were unchanged or worsened in all nine patients with NAECP followed up by EUS for several years. In all three patients with exacerbated EUS findings, they emerged after more than 4 years. Abstinence from alcohol is expected to be effective for both pain relief and improvement of EUS findings in patients with alcoholic ECP. Our findings showed that medication alone may not improve EUS findings in patients with NAECP, although the small population of NAECP was investigated. Further investigations are necessary to elucidate the cause of dissociation of EUS findings from pain relief, and to identify methods to prevent the progression of NAECP.
Rome III diagnostic criteria for functional gastrointestinal disorders (FGID), including functional dyspepsia (FD), were formulated in 2006 [43]. CM has been reported more effective than famotidine in patients diagnosed with FD [44]. In addition, CP may not be completely excluded in patients with FD [45]. ECP may be misdiagnosed as FGID if EUS is not performed. Indeed, 12 (92%) of the 13 patients with NAECP in this study fulfilled the diagnostic criteria of FD, if CP diagnosed by EUS is not considered an organic disease. Early stage CP is not readily diagnosed by CT and MRI, making EUS essential for diagnosing ECP. First-line diagnostic evaluation of patients with upper abdominal pain should include EUS plus upper endoscopy, primarily to detect CP [46]. Patients with upper abdominal pain accompanied by back pain, and abnormal pancreatic enzyme levels in serum or urine should be evaluated by EUS to detect ECP.
In conclusions, EUS is useful in diagnosing NAECP, as CT and MRI cannot detect abnormalities. Although medication was effective in relieving pain due to NAECP, it had no effect on EUS findings. Further investigations are necessary to clarify whether patients with NAECP progress to definite or probable CP, and their risk of pancreatic carcinogenesis, and to determine treatments effective in preventing the progression of NAECP.
The authors declare that there is no conflict of interests regarding the publication of this paper.