- (2002) Volume 3, Issue 5
Åke Andrén Sandberg, Anders Borgström
Department of Surgery, Malmö University Hospital, University of Lund/Malmö. Malmö, Sweden
One out of ten cases of acute pancreatitis develops into severe acute pancreatitis which is a life threatening disorder with a high mortality rate. The other nine cases are self limiting and need very little therapy. The specificity of good clinical judgement on admission, concerning the prognosis of the attack, is high (high specificity) but misses a lot of severe cases (low sensitivity). The prediction of severity in acute pancreatitis was first suggested by John HC Ranson in 1974. Much effort has been put into finding a simple scoring system or a good biochemical marker for selecting the severe cases of acute pancreatitis immediately on admission. Today C-reactive protein is the method of choice although this marker is not valid until 48-72 hours after the onset of pain. Inflammatory mediators upstream from CRP like interleukin-6 and other cytokines are likely to react faster and preliminary results for some of these mediators look promising. Another successful approach has been to study markers for the activation of trypsinogen such as TAP and CAPAP. This is based on studies showing that active trypsin is the initial motor of the inflammatory process in acute pancreatitis. In the near future a combined clinical and laboratory approach for early severity prediction will be the most reliable. Clinical judgement predicts 1/3 of the severe cases on admission and early markers for either inflammation or trypsinogen activation should accurately identify 50-60% of the mild cases among the rest, thus missing only 2-4% of the remaining severe cases. One problem is that there is no imple and fast method to analyze any of these parameters .
Biological Markers; C-Reactive Protein; Cytokines; Diagnostic Techniques, Digestive System; Diagnostic Techniques, Endocrine; Health Status Indicators; Pancreatitis, Acute Necrotizing; Peptides (diagnostic use); Severity of Illness Index; Trypsin (diagnostic use)
CAPAP: carboxypeptidase B activation peptide; ROC: receiver operator characteristic
When the late John HC Ranson designed his prognostic signs for early identification of patients with severe pancreatitis in 1974, he did this to evaluate the role of early operative intervention. After evaluating 100 consecutive cases of acute pancreatitis he selected the nowadays well-known eleven “Ranson prognostic signs“ [1]. In retrospect, perhaps the most important result of his studies was not the finding that early operative intervention with wide pancreatic drainage did not influence the outcome in severe cases of acute pancreatitis but that the prediction of severe cases was possible.
These prognostic signs found no place in clinical practice, partly due to the lack of specific treatment for severe cases of acute pancreatitis at that time. Despite this, severity prediction has gained consistent attention over the years. One reason is that the scoring system has become an important tool in providing stratification of the degree of severity by comparing different patient populations in studies of different treatments or cby omparing the treatment results between different centres. Since the Ranson score was introduced there have been several other multifactorial scoring systems presented, such as the Glasgow score [2] and the APACHE II score (Acute Physiology and Chronic Health Evaluation) [3], to mention just a couple.
Recent reviews of biochemical markers in acute pancreatitis have emphasized the multitude of tests used for disease evaluation [4]. A simpler system - if possible one single reliable prognostic marker - is an important requirement.
`One good reason for early assessment of severity in acute pancreatitis is that this could discount the majority of cases that are mild and have a self limiting course and do not require any treatment other than general support and parenteral fluid for a few days. In the past few years there has been some progress in the treatment of severe acute pancreatitis. Among the emerging new modalities are cytokine inhibitors and different anti-inflammatory regimes. Furthermore, there are randomized studies showing that patients with severe acute pancreatitis benefit from early prophylactic treatment with potent antibiotics [5, 6]. It is also suggested that patients with severe acute gallstone-induced pancreatitis should undergo endoscopic sphincterotomy as early as possible (within 24 or 48 hours after admission) [7]. It can also not be ruled out that treatment with protease inhibitors could re-emerge as a treatment option. It has been shown to be of value in a meta-analysis [8], provided that the therapy can be started early enough, and also as a prophylactic treatment to prevent ERCP-induced acute pancreatitis [9]. All these possible treatments must be instituted at a very early stage in the disease, as the chances for success diminish rapidly with time. Moreover, these therapies are expensive and not without complications, and most importantly, the vast majority of patients with acute pancreatitis will not benefit as their pancreatitis is mild and self-limiting. Therefore, there is today an even greater need for a simple biochemical or clinical marker for severity which can already provide reliable information on admission.
To address the various possible biochemical means of assessing severity requires knowledge of the pathophysiology of acute pancreatitis. Even though details of the process still remain to be elucidated, including the pathogenic mechanisms leading from a localized pancreatic inflammation into a systematic inflammatory response, much is known today. It has been well documented that intra-pancreatic activation of trypsin is an important initiating event in most forms of acute pancreatitis [10, 11, 12]. Trypsin is a very potent activator of different pro inflammatory cascade systems in the organism, such as the complement system and the kininogen system [13, 14, 15]. Activation of these systems induces a severe acute inflammatory reaction and the production of several acute phase reactants. Trypsin is also the physiological activator of the other pancreatic proenzymes such as proelastase, chymotrypsinogen, procarboxypeptidase and prophospholipase A2 [16, 17, 18]. The activities of these enzymes add to the tissue destruction (autodigestion) [19].
The locally induced inflammatory reaction also attracts granulocytes early. These cells contain large amounts of proteases and phospholipase and have the ability to produce oxygen-derived free radicals which are released and are potentially harmful.
Therefore, since trypsin is the prime mover of most initial pathophysiological events in acute pancreatitis, variables that measure trypsinogen activation or trypsin-induced events are likely to correlate to the severity of an attack of acute pancreatitis.
During recent years many new laboratory variables have been proposed as early single tests for severity prediction in acute pancreatitis. They can be divided into four categories:
1. Tests that relate to the degree of the inflammatory reaction such as acute phase reactants and other mediators of the inflammatory process. Although these factors probably act in a paracrine or autocrine, rather than endocrine, manner, they can be measured in the circulation. Elevated blood levels are seen in all other severe acute inflammatory conditions. Included in this group are granulocyte elastase [20, 21], tumour necrosis factor (TNF), interleukin 6 (IL-6) and 8 (IL-8) [22, 23, 24, 25, 26], and C-reactive protein (CRP) [3, 27] in serum. Cellular markers of systemic inflammation and immunosuppression also belong to this group. Immunosuppression also relates to the degree of inflammatory reaction and occurs in all types of trauma reaction. Only recently has this been studied in acute pancreatitis [28].
2. Tests that relate to the activation of trypsinogen and other pancreatic proenzymes such as trypsin-alpha1-protease inhibitor complexes in serum [29, 30], trypsinogen activation peptide (TAP) in urine [31, 32], carboxypeptidase B activation peptide (CAPAP) in serum and urine [17, 33, 34].
3. Tests that measure leakage of certain pancreatic enzymes from the pancreatic gland such as trypsinogen 2 [35, 36, 37, 38, 39, 40], lipase [41], and pancreatic elastase in urine and serum [18, 42].
4. Scoring systems
TNF-alpha is a pleiotrophic predominantly macrophage-derived cytokine which is believed to play a major role in mediating many of the pathophysiologic responses of an organism to injury and sepsis [43]. There are reports of a correlation between TNF-alpha and the severity of acute pancreatitis in some publications [44, 45, 46] but not in others [47].
Interleukin-6 is the principal cytokine mediator of the synthesis of acute phase proteins such as fibrinogen and CRP, and can be measured in serum and urine with a commercially available RIA. Interleukins are known to degrade very rapidly in the circulation. For interleukin-6, however, the samples can be stored at –20 °C for up to six months before analysis provided that the samples are centrifuged and frozen immediately after sampling. Interleukin-6 levels in serum are reported to discriminate severe from mild cases on day 1, and maximal levels are seen on day 1 or 2. Most patients with mild disease have undetectable serum levels of interleukin-6 and thus serum levels have been shown to reflect the severity of an attack of acute pancreatitis [23, 24, 26].
Interleukin-8 is thought to be the principal secondary mediator of TNF-alpha-induced neutrophil activation [43]. Serum interleukin- 8 has been shown to be elevated in the course of acute pancreatitis in several studies [24, 25] and to correlate to the clinical course [25].
C-reactive protein (CRP) is an acute phase reactant which is synthesized by the hepatocytes. This synthesis is induced by the release of interleukin 1 and 6. Thus the CRP peak in serum is usually not maximal until about day three after the onset of pain, and is always later than the peak of these interleukins. CRP is the most popular single test severity marker used today. The problem is that CRP is a rather late severity marker (day 2-4) as compared to the other markers in this overview. Cut-off levels have been discussed in the literature, and levels between 120 and 210 mg/L have generally been agreed upon as distinguishing between the mild and the severe disease [3, 27, 32]. Levels above 120 mg/L after one week also distinguish severity well but this is much too late for an early marker of severity [27, 43].
Cellular markers for systemic inflammation include a proportion of HLA-DR positive monocytes and CD11b expression level on neutrophils and monocytes [28].
Markers for inflammation in general have one disadvantage and that is that they are not always present on admission but develop later during the course of the disease. They are also not specific or diagnostic for acute pancreatitis of course. One big advantage is that these markers can be used to monitor the clinical course of the disease. Some markers, such as CRP assay, are suitable for rapid analysis and also some of the cellular markers are also suitable for immuno-suppression.
Trypsin-alpha-1-protease inhibitor complexes. Levels of trypsin-alpha-1-protease inhibitor complexes in serum correlate to severity in several reports [29, 30]. Levels of this complex are always mostly elevated very early during the disease (from less than 24 to 48 hours). However, high levels of this complex have also been reported in serum from patients with perforated ulcers and other diseases associated with a damaged gastrointestinal barrier [48].
Trypsinogen activation peptide (TAP). This is a small peptide (eight amino acids, molecular weight approximately 900 daltons), which is cleaved from the amino-terminal end of trypsinogen during activation. Among the markers for trypsinogen activation TAP is the most thoroughly studied variable [32]. Elevated levels are seen early after onset of symptoms and are usually maximal within 24- 48 hours.
Carboxypeptidase B activation peptide (CAPAP) in urine and serum. This is the largest activation peptide (molecular weight approximately 10,000 daltons) released from any pancreatic proenzyme. This peptide is very stable in serum and urine. Three retrospective studies have been published [17, 33, 34].
Markers for trypsinogen activation have one major advantage as markers for severity, that is that they appear very early during the disease. They are maximal 1-2 days after onset of pain and then decrease very quickly irrespective of the course of the disease. After 3-4 days these variables are not useful. This rapid decrease give these markers a limited “diagnostic window”. Many patients with acute pancreatitis are admitted rather late and they usually appear at tertiary referral centers long after the 3-4th day. It is also important to realize that these markers are not suitable to use as monitors of disease activity during the course of the disease. The tests for these markers are usually immunological and are not completely suitable for rapid simple analysis although rapid strip tests are starting to emerge for these kind of markers.
Amylase. It is generally accepted that the degree of elevation of the amylase levels in serum and urine shows little correlation with disease severity and prognosis. If anything, amylase levels may have an inverse relationship with severity in that some patients with severe disease have normal or only modestly elevated amylase values when first seen [49, 50].
Trypsinogen 2 (anionic trypsinogen) in serum and urine. Trypsinogen 2 increases considerably more than trypsinogen 1 (cationic trypsinogen) in serum in acute pancreatitis [40]. High serum levels of trypsinogen 2 have been shown to predict complications in patients with acute pancreatitis [36, 37] and also to predict severity after ERCP-induced pancreatitis [38]. In acute pancreatitis, trypsinogen 2 is also excreted into the urine in large amounts. A rapid dipstick method for the measurement of trypsinogen 2 in urine has been developed [39]. This can be used as a screening test for acute pancreatitis.
Lipase is more pancreas-specific than amylase, and it has therefore been advocated to be a more specific marker of acute pancreatitis, especially as it stays elevated for a longer time after the onset of pancreatitis than amylase does [41]. However, the levels bear just as little relationship to severity as amylase.
The major advantage for markers of leakage of pancreatic enzymes are hard to define. One major advantage is that many of these variables can be measured with fast and simple methods. The urinary dipstick for trypsinogen 2 determination is an example of a rapid test strip for urinary analysis and is very simple, fast and suitable to use in the emergency room as a screening method. However, it is important to realize that trypsinogen 2 seems to differ from most markers for acinar cell damage studied earlier such as amylase, lipase and trypsinogen 1. The levels of these enzymes do not relate to severity.
Markers for leakage are better to use as diagnostic tests rather than as prognostic tests.
There are several scoring systems [51, 52, 53, 54, 55, 56] described after Ranson’s first attempt [1], many of them, e.g. the scoring systems from Glasgow and Hong Kong are variants of Ranson’s scoring system [56, 57]. Two general types of scoring systems have been applied to pancreatitis: one that correlates laboratory and clinical markers specific to pancreatitis to subsequent outcome, and the other correlating nonspecific physiological variables to outcome. Examples of the latter are the Acute Physiology and Chronic Health Evaluation (APACHE) II and III, which are built up from a more generalized point: the severely ill patient [58]. When comparing the Ranson score to APACHE III prospectively in an appropriate patient population, with 21% mortality, due to acute pancreatitis, the Ranson score remained a valid predictor of outcome [59].
Today, it seems as if there is only little more information to be gained from the scoring system than what was found by Ranson in 1974, which might indicate that more attention should be paid to what is measured rather than how to combine variables.
Of course, the ability to predict the severe outcome of an attack of acute pancreatitis immediately on admission using a biochemical marker or a scoring system has to be compared to that of a good clinical judgement. The ability of the doctor to predict the severe outcome of a case of acute pancreatitis in the emergency room is described by most textbooks to be poor. One study [60] shows that the ability to predict a severe outcome is rather low with a sensitivity of 34%. Although cases considered severe on admission will turn out to be severe 8 out of ten times, still in that study clinical assessment on admission still failed to identify two thirds of the severe attacks.
The methods that can be used to compare the predictive value of different tests have been summarized in a paper by Jaeschke [61]. What is thought to be most important is to what degree the outcome of the test increases the likelihood ratio of a severe or mild outcome. The likelihood ratios are functions of sensitivity and specificity according to the following formulas:
Likelihood ratio for a positive test:
LRpos = Sensitivity/(100-Specificity);
Likelihood ratio for a negative test:
LRneg = (100-Sensitivity)/Specificity.
Likelihood ratios indicate by how much a given diagnostic test result will raise or lower the pre-test probability for the target disorder. The pre-test probability to predict a severe case of acute pancreatitis is the same as the incidence of severe cases among all cases of acute pancreatitis in a population. This figure varies in different materials between 10-20% depending mostly on selection due to referrals. An LRpos greater than 10 or an LRneg less than 0.1 is thought to be necessary to generate conclusive changes from the pretest to post-test probability. Post-test probability can be obtained from a special nomogram. With a pre-test probability of 20% and an LRpos of 10, a positive result of the test will increase the likelihood of a severe outcome to about 75% which means that 3 out of 4 cases predicted as severe will turn out, in fact, to be severe. An LRneg of 0.1 in the same situation will mean that only 2% of the cases with negative tests will still turn out severe. A test with these likelihood ratios will thus be very useful clinically. Using the sensitivities and specificities in one paper which describes the clinicians’ ability to predict severity, the LRpos is 17 and LRneg 0.67. Thus if an experienced doctor predicts a severe outcome, the chance of a severe attack is around 80%. However if the doctor thinks it will turn out to be mild the chance of a severe outcome is still about 13%. Thus what we need is an immediate test with a low LRneg.
Another way of comparing tests is by calculating accuracy using a receiver operator characteristic (ROC) curve [62]. This curve is a plot of sensitivity versus 100-specificity. Accuracy can be obtained by calculating the area under this curve. An accuracy of 1.0 is optimal and an area of 0.5 indicates no discriminatory capacity in the entire test. The ROC curve is also a very useful tool for calculating the optimal cut-off value of a given test.
A comparison between different variables used to predict severity is shown in Table 1. There are at least two difficulties in comparing different materials in this situation: first, different methods for severity scoring are used and, second, the time between onset of pain and admission are usually not described in detail. In this comparison most of the severity determinations used are based on systems that relate to the number and grade of complications and they are all similar to the Atlanta classification [63].
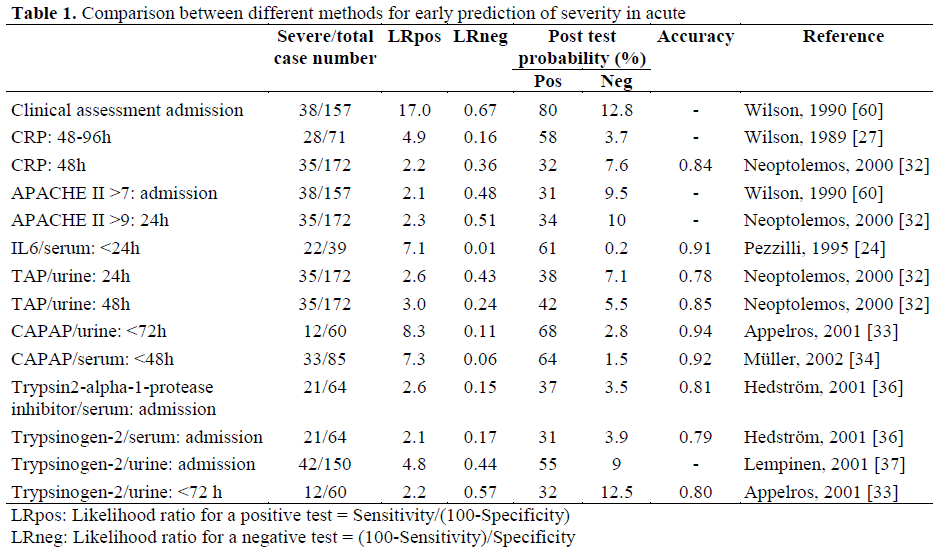
The answer to this question could be both yes and no depending on what you demand of the test. With good clinical judgement, an attack of acute pancreatitis predicted severe on admission, will turn out to be severe 8 out of ten times but you will miss two-thirds of the severe cases. With the best available scoring systems or biochemical methods today only 6 out of 10 cases predicted severe will turn out to be severe but the test will miss only 2-4 severe cases out of 100. For clinical purposes this must be considered to be a large step forward. It may be questionable if this is good enough to recruit severe cases for future therapeutic studies on acute pancreatitis should only severe cases be wanted. A good test will reduce the amount of mild cases in such a study from 80-90% down to 40%.
Only one of the tests available today can be measured using a rapid dipstick method and that is trypsinogen 2 in urine. This test is however best suitable for the diagnosis of acute pancreatitis and less suited for severity prediction. Urinary dipstick methods for an activation peptide like TAP or CAPAP or for a cytokine such as IL-6 or IL-8 would be better suited for this purpose. The methodology is not yet available but it should be possible to develop such methods in the near future.
What we need after this is a large prospective study using a rapid test to differentiate mild and severe cases, to study the effect of more aggressive monitoring and intensive resuscitation therapy in the severe cases and to study the potential risks of undertreating the cases predicted as mild.
This study was supported by Grants from the Swedish Medical Research Council projects no 17-X- 8305-09A, Einar and Inga Nilssons Foundation for Surgical Research and the Foundations for Research at the University Hospital in Malmö.