Review Article - (2021) Volume 0, Issue 0
1Department of Gastroenterology and Hepatology, Oregon Health and Science University, Portland
2Department of Gastroenterology and Hepatology, Dartmouth-Hitchcock Medical Center, Lebanon, New Hampshire
Received Date: August 19th, 2021; Accepted Date: August 30th, 2021
Cystic fibrosis (CF) is the most common genetic disease among Caucasians; worldwide, it affects up to 80,000 children and adults. Its pathophysiology is due to a mutation of the gene encoding the cystic fibrosis transmembrane conductance regulator (CFTR) channel, which alters the secretory epithelium of organs including lungs, intestines, sinuses and pancreas. The pancreas in particular can be affected and cystic fibrosis can result in acute pancreatitis, chronic pancreatitis, and pancreatic insufficiency, which can affect all ages with significant morbidity and mortality. Research has explored the interplay of cofactors that contribute to the pathophysiology of pancreatic-related disorders. This has provided clues to early screening and counseling for the cystic fibrosis population, as well as insights treating this disease. While there are currently no targeted therapies for CF-related pancreatic disorders, recent investigation of CFTR modulators and other mechanisms provides future promise.
Cystic fibrosis; Pancreatitis; CFTR; Therapy; Pancreatic insufficiency
We performed a comprehensive literature search using the electronic Pubmed database for relevant studies on cystic fibrosis and pancreatic-related disorders. Relevant MeSH keywords were used (“cystic fibrosis, pancreatitis, CFTR, therapy, pancreatic insufficiency”). We limited the search to English-language articles published from January 1, 2010 to April 1, 2021. Two independent reviewers assessed the articles for eligibility. Additional relevant studies that were identified upon manual search of the bibliographies of the primary articles, though outside the time window, were later included in this review. Studies published before January 2000 were excluded from the review.
Seventy-eight abstracts overall were retrieved from our literature search. After appraisal by two investigators, including addition of relevant studies from bibliographies of the primary articles, fifty-six publications were included in our synthesis and narrative literature review.
Pathophysiology
Pancreatic ductal secretion plays a vital role in healthy exocrine function of the pancreas [1]. The ductal cells produce a bicarbonate-rich secretion that promotes an ideal alkaline environment for zymogens, prevents premature inactivation of digestive enzymes, and washes out potential factors like bile acid from the biliary tree [2]. Specifically, the CFTR protein regulates chloride and bicarbonate transport across epithelial cell membranes into the lumen, as well as the pH of the epithelial secretions [3]. The WNK1 (with-no-lysine kinase 1) regulates permeability of the CFTR via an intracellular chlorideconcentration dependent association [4]. The CFTR channel is either absent or dysfunctional in secretory gland diseases like cystic fibrosis [5], which causes decreased ductal bicarbonate secretion and the formation of viscous plugging, as well as a low pH environment which can precipitate premature enzyme activation, scarring, and eventual pancreatic insufficiency [5]. Moreover, premature activation of trypsinogen into trypsin plays an important role in the decreased ductal secretion via inhibition of Cl-/HCO3- exchangers and CFTR Cl- channels, thereby perpetuating the cycle of ductal obstruction and inflammation.
Abnormal or absent CFTR also has implications for acinar cells. Ductal inflammation, as a consequence of viscous plugging from deficient ductal secretion, has shown to inhibit acinar cell secretion, promote acinar cell degradation, and ultimately lead to parenchymal damage [6]. It is the initial insult to the ducts, then, that may be causing secondary downstream damage to the acinar cells [6]. In addition to the HCO3- mechanism, aquaporins have been investigated for their role in pancreatic fluid formation and pancreatitis. Aquaporins are membrane proteins that mediate transcellular water transport. In particular, aquaporin 1 co-localizes with the CFTR channel in the apical membrane of the pancreatic ductal cells [2]. Damaged CFTR reduces the water permeability of ductal cells via decreased expression of aquaporin, causing reduced ductal fluid and HCO3-secretion overall [2]. Thus, CFTR plays an important role in the pathophysiology of aquaporins and development of pancreatic inflammation, evidenced by the decreased expression of aquaporins in acute and chronic pancreatitis.
CFTR is also responsible for intracellular signaling in pancreatic epithelial cells via calcium. The loss of CFTR has therefore been implicated in excess mitochondrial calcium accumulation, increased permeability of the inner mitochondrial membrane, and subsequent mitochondrial swelling, apoptosis and necrosis [7].
In addition to protein channels, pancreatitis-inducing factors and toxins also modulate CFTR expression and activity (Table 1). Ethanol and fatty acids have demonstrated a dose-dependent inhibition of CFTR and decreased fluid and HCO3- secretion as a result [3]. Moreover, cigarette smoke increases intracellular calcium and causes downstream CFTR channel internalization and down-regulation of CFTR expression, as well as increased sweat chloride levels [8, 9]. Some studies speculate that constituents of smoke like acrolein, cadmium, and manganese -and specifically the free radical-induced damage by smoke-may also directly be responsible for inhibition of CFTR [10].
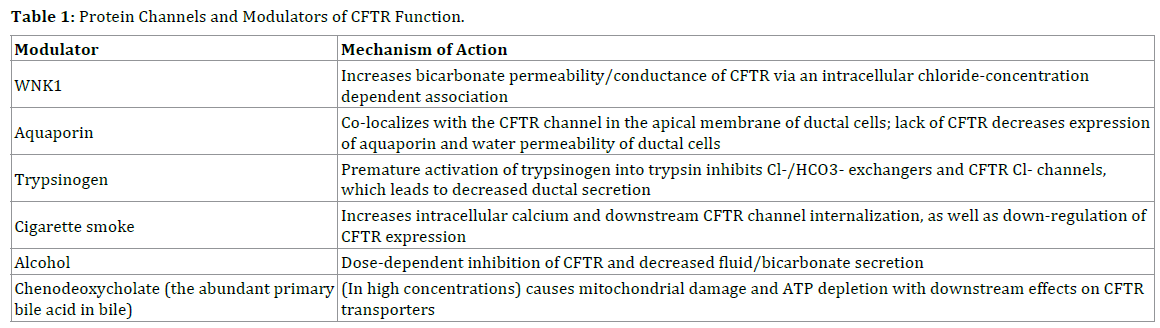
In regard to biliary pancreatitis, chenodeoxycholate, the abundant primary bile acid in bile, in high concentrations has shown to cause mitochondrial damage and ATP depletion with downstream effects on CFTR transporters [11, 12]. Thus pancreatic ductal secretion of bicarbonate is strongly inhibited [3].
Pancreatic insufficiency
Historically, pancreatic insufficiency was associated with significant early childhood mortality before the advent of pancreatic enzyme replacement therapy and improved nutritional care. The vast majority (80%) of cystic fibrosis patients are pancreatic-insufficient [13], largely due to ductal plugging and autodigestion of the pancreas throughout the embryonic stage [14]. Once patients have sufficient pancreatic damage to the extent of losing exocrine function, the pancreas becomes functionally inept and lack the acinar reserve to develop pancreatitis (Figure 1).
Pancreatic exocrine sufficiency is affected by the presence of either severe or mild mutation on each respective CFTR allele [3]. Patients with the mild CFTR genotype are often pancreatic sufficient but have increased risk for pancreatitis [15]. Pancreatic-sufficient patients also are at increased risk for developing exocrine insufficiency, once they have had an index episode of pancreatitis [15]. Patients with pancreatic insufficiency typically develop malabsorption symptoms of foul-smelling stools, abdominal discomfort and weight loss. Combined with the highly catabolic process from pancreatic inflammation and autodigestion, pancreatic insufficiency when left untreated can progress to severe malnutrition and growth failure.
Malnutrition in pancreatic insufficiency is multifactorial: malabsorption from deficient pancreatic enzymes, bile salt precipitation impairing lipid solubilization, altered gut motility and mucosal uptake, inadequate energy intake, and increased energy requirements [14]. In the appropriate clinical context, indirect pancreatic function tests like a pancreatic fecal elastase help make a diagnosis of pancreatic insufficiency with symptoms and signs suggestive of malabsorption. A 72 hour fecal fat test remains the gold standard test to diagnose pancreatic insufficiency, though this can be cumbersome to carry out in clinical practice.
Pancreatic enzyme replacement therapy in cystic fibrosis
Standard of care therapy for pancreatic insufficiency is pancreatic enzyme replacement therapy (PERT) with fat-soluble vitamin supplementation. PERT contains the exogenous lipase, amylase, and protease that digests their respective macromolecules, which is vital in patients with cystic fibrosis for normal growth and nutrition. Without PERT, patients are at risk for severe lung disease, distal intestinal obstruction syndrome, and decreased life expectancy. PERT formulations are porcine-derived and either enteric-coated (which are protected against denaturing by gastric acid) or non-enteric coated. Microsphere formulations, which allow for smaller size preparation and more stable delivery, theoretically hasten delivery of the enzymes into the duodenum, but clinical trials have failed to demonstrate substantial improvement in bowel symptoms with microsphere formulations [16]. Moreover, the type of preparation has not shown significant differences on bowel symptoms, quality of life, or lung disease outcomes [16].
Pancreatic insufficiency is frequently underdiagnosed and PERT underdosed, and yet there remains significant heterogeneity between cystic fibrosis centers in regard to management of PERT. Some neonatal screening programs will administer PERT within the first few weeks of a diagnosis of cystic fibrosis; some centers will initiate PERT in clinical scenarios of growth failure in children and adults in failure to thrive, whereas others will rely on pancreatic function testing to guide decision-making on starting PERT [16]. Myriad factors contribute to the efficacy of PERT in each patient: the residual pancreatic function, meal size and fat content, and level of bile acid secretion and intestinal pH [17]. Titration of PERT is largely tailored to each individual patient. Improvement in steatorrhea should be assessed, as should surrogate markers such as fat-soluble vitamin levels, nutrition indices, and quality of life metrics [18]; fecal elastase is unaffected by PERT and should not be used to assess clinical response. Side effects of PERT can include nausea, constipation, diarrhea, abdominal cramping, bloating and less commonly, fibrosing colonopathy with high doses of PERT exceeding 10000 units per kilogram body weight per day [16, 17].
Acute pancreatitis
The prevalence of acute pancreatitis in patients with cystic fibrosis overall is estimated to be 0.5-1.7%, though patients with pancreatic sufficiency have a higher risk (32%) of developing pancreatitis [19]. Mutation of the CFTR genotype is one predisposing factor believed to be responsible for this increased risk. CFTR mutations have been speculated to induce acute pancreatitis in two ways: 1) decrease in pancreatic fluid secretion thereby producing more viscosity and distal obstruction of the duct, and 2) development of excess inflammatory response [20]. The type of CFTR mutation significantly correlates with the risk of acute pancreatitis. And it is the mild CFTR genotype that has a dominant phenotypic affect, compared to that of the severe genotype [15].
It remains unclear why there is such heterogeneity in which pancreatic-sufficient patients develop pancreatitis. Why patients with identical CFTR genotypes can have such variable presentations of pancreatitis is also unclear [19]. One hypothesis is that other non-CFTR genetic variants or environmental exposures (ie, tobacco, alcohol) may be additional predisposing factors for the development of pancreatitis. However, a prospective study of pancreaticsufficient patients [19] showed that while the majority of mild mutations were found in the first transmembrane domain, there was no direct correlation between non- CFTR genotypes (including SPINK1, PRSS1, CTRC, CASR, and SLC26A6) and the prevalence of acute or recurrent pancreatitis. Yet it remains very plausible that multiple players including these genetic variants and environmental exposures work synergistically to contribute to pancreatitis, and further mechanistic studies are needed to better understand and model the interactions between these cofactors [3, 19, 21].
Pancreas Divisum
Pancreas divisum is a variant anatomy of the pancreatic duct resulting from failed fusion of the ventral and dorsal pancreatic buds during embryonic development, leading to pancreatic drainage thru the minor papilla (Figure 2). There is some controversy whether pancreas divisum is a true risk factor for pancreatitis or a nonpathological clinical entity. Although pancreas divisum is surprisingly prevalent (up to 5-10% in autopsy series), only a minority of patients with divisum develop pancreatitis [20]. Studies have failed to consistently show increased frequency of pancreatitis in patients with pancreas divisum, when compared to controls [22]. These findings suggest that pancreas divisum per se may not cause pancreatitis. However, pancreas divisum has shown to be more frequent in cases of genetic pancreatitis, particularly CFTR mutations [22]. CFTR mutations have independently been associated with pancreatic ductal obstruction and inflammation. As pancreas divisum theoretically can lead to increased intraductal pressure by way of drainage through the smaller minor papilla, pancreas divisum may ultimately serve as a cofactor with CFTR in the development of pancreatitis [23]. On the other hand, most cystic fibrosis patients are pancreatic insufficient and are less likely to have develop pancreatitis. It is postulated, then, that pancreas divisum may be protective against pancreatic insufficiency in CFTR carriers, at the cost of increased risk for pancreatitis [24]. Further studies are needed to assess whether or not surgical or endoscopic interventions to enlarge the minor papilla orifice reduces the likelihood of pancreatitis in patients with pancreas divisum and CFTR mutation.
Chronic pancreatitis
Chronic pancreatitis is characterized by progressive inflammation of the pancreas and ductal plugging [25], which leads to acinar loss, lobular and periductal fibrosis, and irreversible pancreatic damage [26]. These changes are due to dysregulation of HCO3- secretion. However, morphometric analysis of pancreatic ductal mucosa has also shown increased mucus content in the small pancreatic ducts in chronic pancreatitis, as well as increased expression of secreted mucins MUC5B and MUC6 [25]. Whether the mucus hypersecretion via differential expression of mucin proteins is a result or cause of the increased mucus content and associated plugging in the pancreatic ducts remains unclear [25]. Therapies that increase ductal hydration and improve mucus clearance, therefore, may play a vital role in washing out the digestive enzymes and toxins in the early stages of chronic pancreatitis, before progressive and irreversible inflammation develop [27]. Genetic variants including PRSS1, SPINK1, CTRC, and CFTR have long been associated with the development of chronic pancreatitis, presumably to their effect of premature trypsinogen activation [3, 28, 29]. Although CPA1 has previously been implicated in the pathogenesis of chronic pancreatitis [30], Hegyi et al. [31] was the first in-vivo study to demonstrate CPA1 as a disease-modifying factor for chronic pancreatitis, via enzyme misfolding and induction of endoplasmic reticulum stress. This pathway may ultimately have therapeutic implications which are independent of the trypsinogen pathway. There are also functional CFTR variants with impaired HCO3- (but not Cl-) permeability which can predispose patients to chronic pancreatitis, but not cystic fibrosis [5, 32].
Pancreatic cystosis
Pancreatic cystosis in cystic fibrosis is relatively uncommon, and large cysts spanning greater than 1 centimeter are even more rare, occurring in only 8% of patients with cystic fibrosis [33]. Little is known regarding the natural history and clinical outcomes of pancreatic cystosis. A recent systematic review showed that symptoms on initial presentation were noted in only 42% of total reported cases, with no strong correlation between size of cyst and symptoms [34]. There is neither proven risk of progression to malignancy with pancreatic cystosis [35] nor copious information on the natural history of cytosis, so it is reasonable to hold off on treatment if patients are asymptomatic. Decisions on therapy, therefore, are predicated on factors such as severity of pain symptoms, quality of life, obstructive symptoms, as well as comorbidities such as recurrent pulmonary complications [34].
CF related diabetes
Cystic fibrosis-related diabetes is a pancreatic complication that increases in prevalence with age, up to 40-50% of patients older than 40 years of age [36]. Progressive damage to the pancreatic islet cells leads to insulin deficiency, contributing to diabetes. Other factors that can impair glucose tolerance include infection, malnutrition, concomitant liver disease, and bacterial overgrowth [14]. Diabetes in turn can also have a multifactorial effect on pancreatic duct secretion: decreased or absent endogenous insulin impairing secretion at both the ductal (decreased duct morphology) and acinar level (tissue atrophy), autoantibodies against carbonic anhydrase, autonomic neuropathic effect on ductal secretion, negative feedback of elevated blood glucose on sodium-dependent glucose transporter and downstream bicarbonate secretion, and decreased Na+/ K+-ATPase activity [5]. Cystic fibrosis-related diabetes is associated with increased mortality, decreased pulmonary function and malnutrition [36]. Moreover, screening and detection can be challenging particularly in the early stages of cystic fibrosis-related diabetes, as these patients may be at risk for microvascular complications well before traditional tests like HbA1c detect diabetic disease [36]. Typically, once a diagnosis of cystic-fibrosis related disease is confirmed via oral glucose tolerance test, basal insulin and adequate caloric/energy intake is recommended with the supervision of a multidisciplinary team with a cystic fibrosis dietician.
Advances in CFTR modulators and other therapies
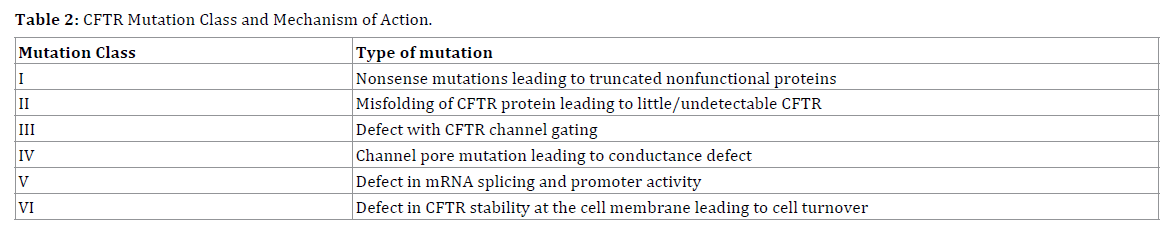
Initial therapies for cystic fibrosis were largely supportive including inhaled mucolytics, antibiotics, and pancreatic enzyme replacement therapy. Ideally to halt disease progression, treatments that target the underlying CF mutation present the most promise. There are over two thousand known CFTR mutations, though most have no clinically significant phenotype. The mutations that are most typically associated with disease are categorized class I-VI mutations (Table 2). Class I mutation results in truncated nonfunctional proteins and Class II non-expressed CFTR protein at the plasma membrane [37]. F508del is in class II and the most common CFTR mutation (up to 80-90% in patients with cystic fibrosis) [38]. For classes III and IV, the CFTR protein is fully synthesized and expressed, but there are defects with CFTR channel gating (III) and conductance via channel pore defect (IV). Class V mutation is a defect in mRNA splicing and promoter activity, whereas Class VI mutation deals with defective CFTR stability at the cell membrane and resultant cell turnover [37].
Therapeutic efforts in cystic fibrosis therefore have been directed at these specific mutations (Table 3). Ivacaftor was first identified as a CFTR potentiator that increases the CFTR channel opening at the cell surface and thereby enhances chloride ion transport [39]. It went on to demonstrate normalized fecal elastase and decreased number of pancreatitis episodes in patients with CFTR dysfunction [40]. Moreover, ivacaftor has demonstrated in vivo evidence of improved proximal small intestinal pH, which supports the hypothesis of CFTR modulator’s effect on bicarbonate secretion, gut hydration, and gut biosis as well as downstream effects on weight gain [41]. However, ivacaftor has not shown to be effective in a significant portion of the cystic fibrosis population, namely the F508del homozygous patients [42]. CFTR correctors were also explored, which increase trafficking of the CFTR protein to the cell surface. The corrector lumacaftor, though shown to improve CFTR function in the sweat duct, failed as monotherapy in demonstrating significant improvement in outcomes like lung function [43].
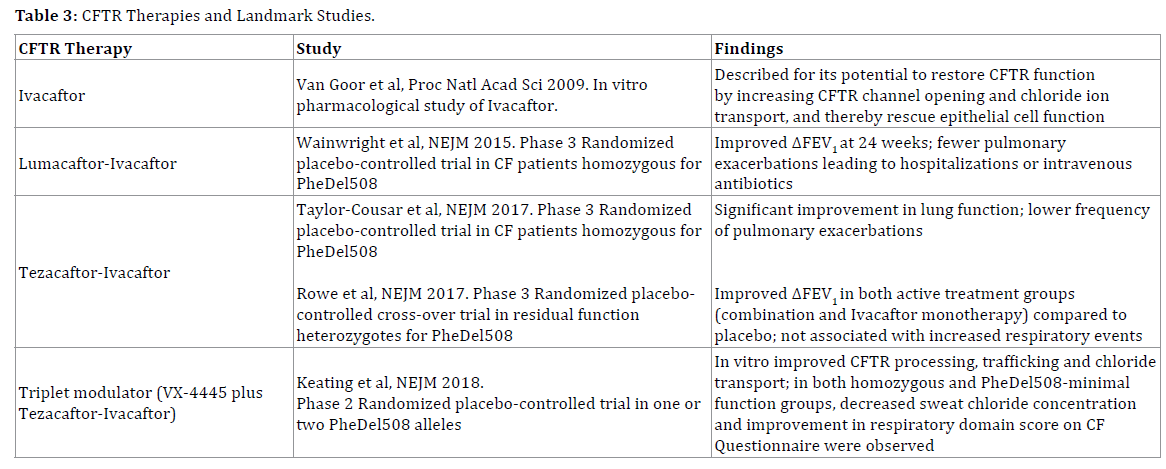
The question then arose whether combining a CFTR corrector with a potentiator may provide added benefit. CFTR correctors and potentiators have been investigated in combination in mouse models for their role in rescuing ductal CFTR. In mouse models of Sjogren’s and chronic pancreatitis, CTFR potentiator VX770 and corrector C18 restored ductal CFTR, as well as increased calcium signaling, aquaporin expression, and fluid secretion in acinar cells [6]. Lumacaftor-ivacaftor was first approved for combination use after demonstrating modest improvement, compared to placebo, in FEV1 (forced expiratory volume during first second) of patients with cystic fibrosis homozygous for PheDel508 CFTR [44]. Because of the modest effect, potential drug interaction between lumacaftor and ivacaftor, and side effects of dyspnea and liver damage, another combination tezacaftor-ivacaftor was investigated and found to have similar effect size with lung function, albeit improved side effect profile [45, 46].
Further studies have since investigated triplet CFTR modulator therapy: two correctors with distinct binding sites and mechanisms are hypothesized to increase PheDel508 processing and trafficking to the cell surface, which can then be potentiated by a third agent like ivacaftor [47]. Tezacaftor-ivacaftor has served as the foundation for combination therapy with next-generation correctors such as VX-440, VX-152, and VX-659 [48], with randomized trial studies demonstrating significant improvement in FEV1 [47, 49].
Although triplet modulator therapy with novel correctors show promise for the PheDel508 patients which make up the bulk of the CF population, up to 8-10% remaining have nonsense mutations resulting in premature termination of CFTR protein synthesis [50]. Additional therapies requiring ongoing study include eluforsen, an antisense oligonucleotide that repairs the genetic defect in RNA, which has demonstrated safety and efficacy with respiratory symptoms in phase 1 trials [51]. Ataluren had been studied for its potential benefit in nonsense mutation cystic fibrosis via reading through premature termination codons and producing functional CFTR proteins—but results in phase 3 have failed to show significant pulmonary benefit from placebo [52]. Gene editing strategies have been explored with recent studies using Cas9 and adenoassociated virus 6 to correct CFTR mutation in upperairway basal cell stem cells ex vivo [53]. These corrected basal cells can then be embedded onto a scaffold for engraftment while retaining their differentiation capacity. Other approaches being investigated include derivation of donor-specific induced pluripotent cells with CFTR correction and directed differentiation to airway basal cells, as well as direct in vivo editing of airway cells [54]. Such gene editing and replacement therapy may ultimately hold promise for the patients who do not respond to triplet modulator therapy. An additional challenge will be the selection of more sensitive clinical endpoints, as traditional outcomes of FEV1 and number of pulmonary exacerbations [1] have significantly improved with triplet modulator therapy. Future clinical trials will need endpoints that better encompass pancreatic function and end-stage complications.
TPIAT in CF patients
TPIAT (total pancreatectomy with islet autotransplantation) is an appropriate treatment option for severe chronic pancreatitis and recurrent acute pancreatitis. It can relieve pain as well as preserve betacell mass to reduce the risk of post-operative diabetes [55]. Moreover, when compared to controls without CFTR mutations, patients with CFTR homozygotes and heterozygote mutations who underwent TPIAT had similar post-operative Hgb A1c, C-peptide, islet yield, and post-operative complications [55]. TPIAT may be a viable treatment option in cystic fibrosis patients with medically refractory chronic pancreatitis and recurrent acute pancreatitis. Also TPIAT has demonstrated substantive pain relief and improved quality of life for pancreatic cystosis in cystic fibrosis [56]. Provided that islet cells are preserved, and cysts drained prior to digestion to ensure a high islet yield, TPIAT can provide a surgical option for these difficult cases. Overall further studies are needed to risk stratify patients undergoing TPIAT with concomitant lung and pancreas involvement of cystic fibrosis.
CFTR mutation in cystic fibrosis leads to ductal plugging, pancreatic inflammation and downstream pancreatic insufficiency which can cause severe malnutrition and growth failure. PERT is vital in the management of pancreatic insufficiency, nutrition and overall morbidity in patients with cystic fibrosis. Trypsinogen and aquaporins may play an important role in the pathophysiology of pancreatic ductal secretion. The development of acute pancreatitis depends on cofactors of genetic variants, environmental exposures, and severity of CFTR genotype. Other pancreatic complications in cystic fibrosis include cystic fibrosis-related diabetes and, less commonly, pancreatic cystosis; pancreatic divisum may be a cofactor for pancreatitis rather than an independent risk factor per se. Therapies for cystic fibrosis have evolved with triplet modulator therapy demonstrating the potential to alter disease trajectory in the majority of cystic fibrosis patients. However, ongoing investigation with gene replacement therapy and antisense oligonucleotide therapy will be necessary to meet the needs of patients with nonsense mutations, who may not respond to triplet modulator therapy.
The authors received no financial support for the research, authorship, and/or publication of this article. The authors have no competing financial interests or personal relationships to disclose.
The authors declare no competing interest.