Keywords
Dopamine; Cyclosporine; Modified carbon paste electrode; Sodium dodecyl sulphate (SDS); Cyclic voltammetry
Introduction
Cyclosporine (CSA) is a neutral compound soluble only in organic solvents or lipids with very low water solubility [1]. It is a powerful immunosuppressant, drug immune system is made of special type of cells like proteins, tissues, and organs defend the body against germs and microorganisms. Cyclosporine is used to prevent the rejection of transplanted organs [2]. Cyclosporine is blocking reversibly, the early events in T cell as well as in B cell and natural killer cell response leads to the alteration of immune response [3,4] and it may also have clinical application in the treatment of autoimmune disorders (Scheme 1). Cyclosporine can also used for transdermal applications [5].
Scheme 1: Structure of Cyclosporine.
Dopamine (DA) is an important monoamine neurotransmitter belongs to catecholamine family. It found in neurons of both the central and peripheral nervous system major dopamine containing area of the brain is the corpus stratum [6]. The DA is also known as 4-(2-Aminoethyl)-1, 2-benzenediol. In 1958 Arvid carlsson and Nilsake Hillap discovered the function of dopamine as a neurotransmitter in the mammalian central nervous system. It plays an important role in controlling emotions, pleasure, movement and incoming information. It facilitates transmission of signals between brain cells or neurons. And it plays very important role in the life of the mammalian central nervous system. The Low level of dopamine causes neurological disorders, such as Parkinson’s disease [7,8] schizophrenia disease [9,10] and Alzheimer’s disease [11]. Dopamine plays a role in drug addiction [12] and some manifestation of HIV [13]. It is an electrochemically active compound hence it can be easily determined by electrochemical methods based on the anodic oxidation [14]. Therefore, it is a challenging task to develop sensitive and simple methods for the determination of dopamine. There are methods were introduced to determine the dopamine such as spectroscopy, chromatography, and electrochemistry [15- 18]. The electrochemical method received a lot of interest due to its simplicity, sensitivity and eco-friendly and this method even suitable for coloured samples [19].
Ascorbic acid is a water-soluble vitamin and plays very important role in the metabolic process of living organisms. Due to its powerful antioxidant property, it protects the body against oxidative stress [20]. It defends the body from cold, infertility, and mental illness [21,22] it is used to prevent the scurvy disease.
Uric acid is one of the non-protein nitrogen constituent of the blood produced by purine metabolism and it is filtered in the kidney and excreted in the urine. The concentration of uric acid plays a very important role. The normal uric acid range for males is between 3.6 mg/dL and female is 2.3-6.6 mg/dL [23]. High concentration of uric acid in the body leads to several disorders such as gout, Lesch-Nyhan syndrome, Hyperuricemia, cardiovascular disease and multiple sclerosis [24,25].
Surfactants are surface active agents and they are in amphiphilic nature. Due to their unique molecular structure surfactants have been extensively used in the fields of cleaning uses, preparation of paints, pigments and manufacture of rubber, resins. Due to adsorption at interface and aggregation in to supramolecular structures, they have ability to modify and control the properties of electrode surface and enhance the rate of reaction therefore they are beneficially used in electrochemistry [26-30].
In the present work the modified carbon paste electrode was prepared by using cyclosporine as a modifier, it was further modified by immobilization of SDS surfactant. The modified Cyclosporine/SDS carbon paste electrode showed a good electrocatalytic activity for dopamine and uric acid. The modified new carbon paste electrode has more advantages, including high sensitivity, rapid response and simplicity.
Experimental Part
Reagents and chemicals
Cyclosporine was kindly supplied by PerkinElmer (Waltham, MA, USA). Dopamine (DA) was obtained from Himedia chemical company with analytical grade used without further puriï¬ÂÂÂÂcation. 25 mm DA stock solution was prepared in 0.1 M Perchloric acid, Sodium dodecyl sulphate, potassium ferro cyanide and KCl was prepared in double distilled water. Graphite powder of 50 mm size was purchased from Loba and silicon oil was purchased from Himedia. The chemicals for preparation of buffer solution were purchased from Merck. Phosphate buffer (0.2 M pH 7.4) was used as supporting electrolyte.
Instrumentation
Cyclic voltammetric experiments were performed using a model CHI-660c (CH Instrument-660 electrochemical workstation). All experiments were carried out with a conventional three electrode cell. The electrode system contained a carbon paste working electrode (3.0 mm in diameter), platinum wire as a counter electrode and saturated calomel as a reference electrode.
Electrode preparation procedure
Preparation of bare carbon paste electrode: The carbon paste electrode was prepared by graphite powder and silicon oil at a ratio of 70:30 (w/w) in an agate mortar by hand mixing to get homogenous paste. The prepared carbon paste was tightly packed into the cavity of a homemade carbon paste electrode then smoothened the surface of a weighing paper.
Preparation of modified carbon paste electrode: The modified carbon paste electrode was prepared by using 4 mg of cyclosporine, graphite powder and silicon oil at a ratio of 70:30 (w/w) in an agate mortar by hand mixing to get homogenous paste. The modified carbon paste electrode was tightly packed into the cavity of a homemade carbon paste electrode then smoothened the surface on a weighing paper. The modified Cyclosporine electrode was further modified by immobilization by adding an SDS solution (10 μL) on the surface of Cyclosporine MCPE and allowed for 5 min. Later electrode was thoroughly rinsed with distilled water to remove the unabsorbed modiï¬ÂÂÂÂer and dried in air at room temperature.
Results and Discussion
Cyclic voltammetric studies of K4Fe(CN)6 at Cyclosporine/SDS MCPE
The cyclic voltammetric response at Cyclosporine/SDS MCPE was studied using 1mM [K4Fe (CN)6] in 1M KCl as a supporting electrolyte with the scan rate 100 mVs-1 was shown in Figure 1a.
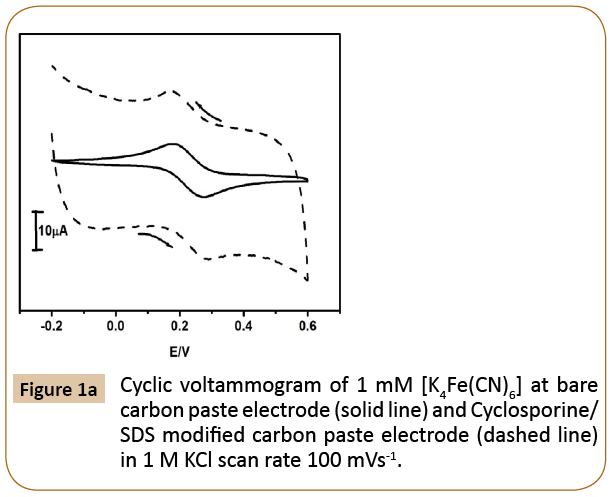
Figure 1a: Cyclic voltammogram of 1 mM [K4Fe(CN)6] at bare carbon paste electrode (solid line) and Cyclosporine/ SDS modified carbon paste electrode (dashed line) in 1 M KCl scan rate 100 mVs-1.
The cyclic voltammogram at modified Cyclosporine/SDS CPE shows excellent enhanced peak current (dotted line) as compared to the bare carbon paste electrode. The trace amount of SDS on the surface of Cyclosporine MCPE alter the electrochemical response of [K4Fe(CN)6] and influence the rate of electron transfer.
Cyclic voltammetric studies of K4Fe(CN)6 at Cyclosporine MCPE
The cyclic voltammetric response at Cyclosporine/MCPE was studied using 1mM [K4Fe(CN)6] in 1M KCl as a supporting electrolyte with scan rate 100 mVs-1 was shown in Figure 1b.
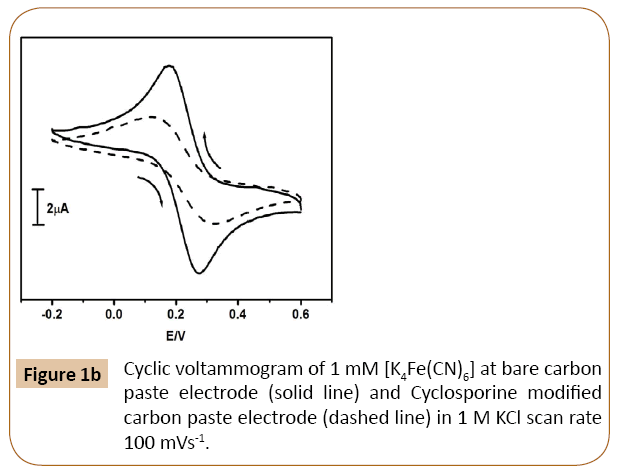
Figure 1b: Cyclic voltammogram of 1 mM [K4Fe(CN)6] at bare carbon paste electrode (solid line) and Cyclosporine modified carbon paste electrode (dashed line) in 1 M KCl scan rate 100 mVs-1.
The cyclic voltammogram at modifying Cyclosporine/MCPE shows decrease in current signal as compared to BCPE (solid line).
Cyclic voltammetric studies of DA at Cyclosporine/ SDS MCPE
The cyclic voltammetric response of DA at Cyclosporine/ SDS MCPE and BCPE was shown in Figure 2a. The cyclic voltammograms of dopamine in 0.2 M phosphate buffer at pH 7.4 and scan rate 100 mVs-1 at Cyclosporine/SDS MCPE showed a good enhancement of peak current (dashed line) as compared to BCPE. In the presence of trace amount (10 μl of SDS) surfactant on the surface of Cyclosporine modified carbon paste electrode greatly influence the enhancement of electrochemical reaction and it was confirmed by the enlargement of peak currents [31].
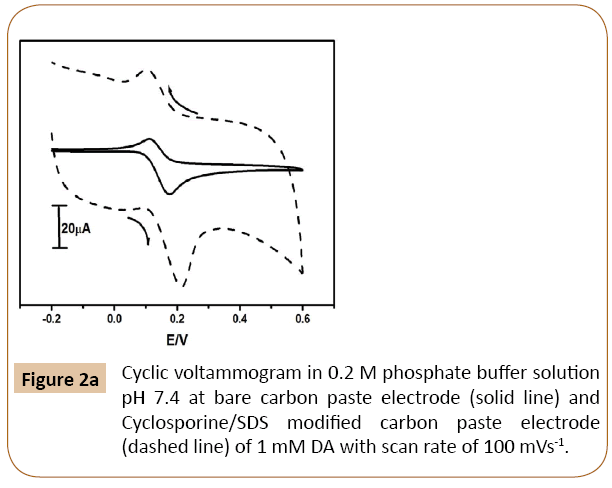
Figure 2a: Cyclic voltammogram in 0.2 M phosphate buffer solution pH 7.4 at bare carbon paste electrode (solid line) and Cyclosporine/SDS modified carbon paste electrode (dashed line) of 1 mM DA with scan rate of 100 mVs-1.
Cyclic voltammetric studies of DA at Cyclosporine MCPE
The cyclic voltammetric response of DA at Cyclosporine MCPE and BCPE was shown in Figure 2b. The cyclic voltammogram of dopamine in the absence of SDS at modifying Cyclosporine carbon paste electrode shows low signal as compared to BCPE (solid line).
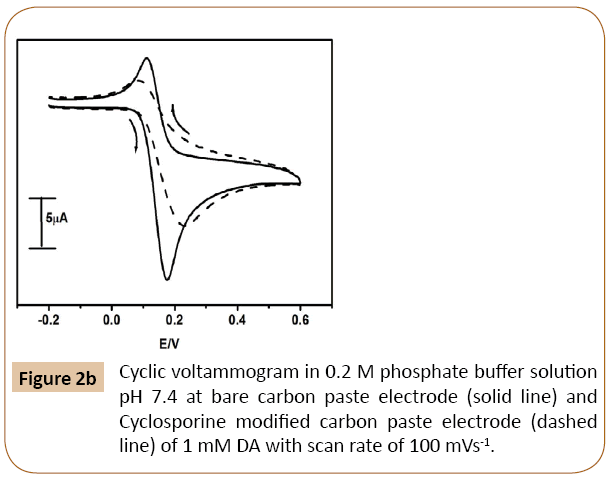
Figure 2b: Cyclic voltammogram in 0.2 M phosphate buffer solution pH 7.4 at bare carbon paste electrode (solid line) and Cyclosporine modified carbon paste electrode (dashed line) of 1 mM DA with scan rate of 100 mVs-1.
Effect of scan rate on Cyclosporine/SDS MCPE
Figure 3a shows the cyclic voltammograms of dopamine recorded for different scan rates in 0.2 M phosphate buffer of pH 7.4 as a supporting electrolyte at Cyclosporine/SDS MCPE. This observation was carried out to investigate the kinetics of the electrode reaction and to verify the mass transport process involved in electrochemical processes, whether diffusion or adsorption-controlled. The scan rate varying study showed that current improved linearly with the square root of scan rate range of 50 to 400 mVs-1 The graph of anodic peak current (Ipa) versus the square root of scan rate showed a linear relationship of Ipa and scan rate. The correlation coefficient was found to be 0.9950 Figure 3b it indicates that the electrode process was diffusion controlled [32-35]. The difference between the anodic and cathodic peak potential (Δ Ep) increasing with increasing the scan rate [36-38].
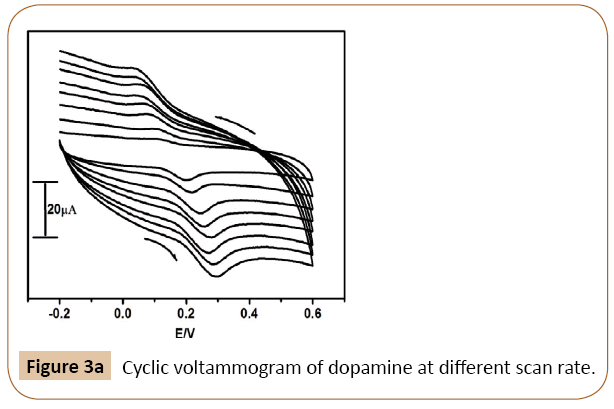
Figure 3a: Cyclic voltammogram of dopamine at different scan rate.
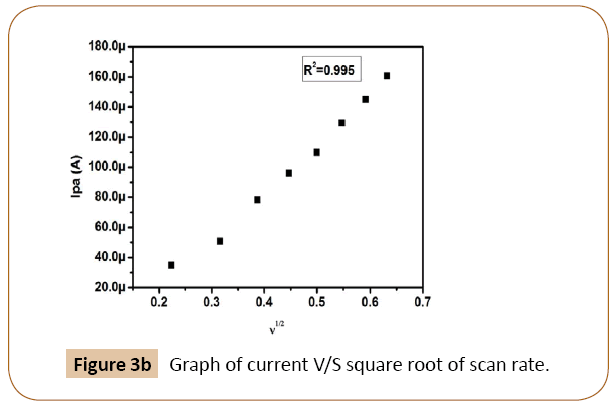
Figure 3b: Graph of current V/S square root of scan rate.
Concentration effects of DA on Cyclosporine/SDS MCPE
The cyclic voltammograms of different concentration of dopamine in the 2M phosphate buffer, pH 7.4 and scan rate 100 mVs-1 at Cyclosporine/SDS MCPE have shown in Figure 4a.
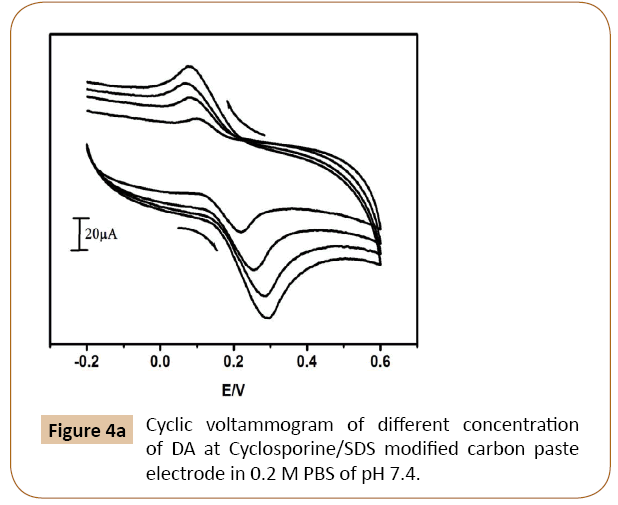
Figure 4a: Cyclic voltammogram of different concentration of DA at Cyclosporine/SDS modified carbon paste electrode in 0.2 M PBS of pH 7.4.
The electro catalytic oxidation of dopamine was carried out by varying the concentrations and it shows anodic current increases with increase of dopamine concentration from 0.1 × 10-3 M to 0.4 × 10-3 M. The electrochemical anodic and cathodic peak currents go on increasing with anodic peak potential shifting towards positive and cathodic peak potential shifting towards negative direction slightly. The Figure 4b shows the Ipa versus dopamine concentration and the figure indicates that the concentration of dopamine is directly proportional to the electrochemical anodic peak current.
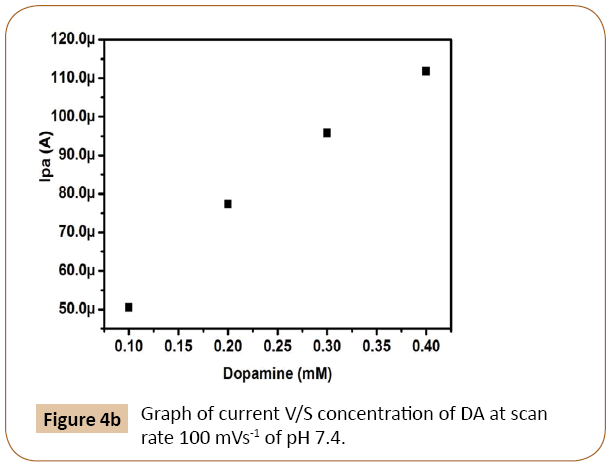
Figure 4b: Graph of current V/S concentration of DA at scan rate 100 mVs-1 of pH 7.4.
Concentration effect of SDS and immobilization time at cyclosporine MCPE
The cyclic voltammograms of dopamine at different concentration of SDS on Cyclosporine MCPE was shown in Figure 5a. The Figure 5b shows that enhancement of anodic peak current of dopamine was closely related to the concentration of SDS on the surface of Cyclosporine modified carbon paste electrode. Anodic peak current of dopamine goes on increases with increasing the concentration of SDS up to 10 μl, after that it shows the decreases in current signal with increasing the concentration of SDS. Similarly immobilization time also influences the anodic peak current of dopamine. The influence of immobilization time was examined and results shown in Figure 5c. The peak current is more with immobilization time 5 min and then decreases. Therefore 10 μl of SDS and immobilization time 5 min was chosen for further electrochemical studies.
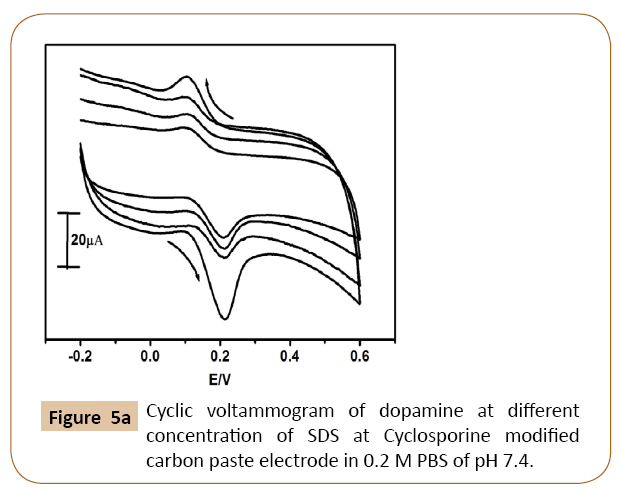
Figure 5a: Cyclic voltammogram of dopamine at different concentration of SDS at Cyclosporine modified carbon paste electrode in 0.2 M PBS of pH 7.4.
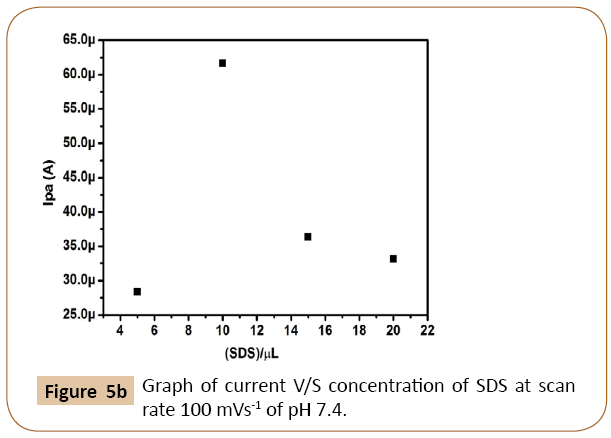
Figure 5b: Graph of current V/S concentration of SDS at scan rate 100 mVs-1 of pH 7.4.
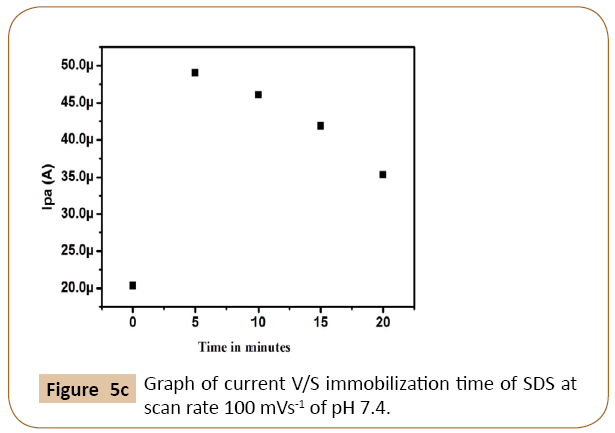
Figure 5c: Graph of current V/S immobilization time of SDS at scan rate 100 mVs-1 of pH 7.4.
Cyclic voltammetric response of UA at Cyclosporine/ SDS MCPE
The Figure 6 shows cyclic voltammograms of uric acid in 0.2 M phosphate buffer at pH 7.4 with a scan rate of 100 mVs-1 at Cyclosporine/SDS MCPE. The cyclic voltammogram of uric acid in the absence of SDS at cyclosporine modified carbon paste electrode shows low signal. However, in the presence of SDS at cyclosporine modified carbon paste electrode showed a good increment in the peak current. The cyclic voltammograms at modifying Cyclosporine/MCPE shows decrease in current signal (dotted line) as compared to bare carbon paste electrode (solid line) the modified Cyclosporine/SDS carbon paste electrode showed a good enhancement of peak current (dashed line) as compare to bare carbon paste electrode.
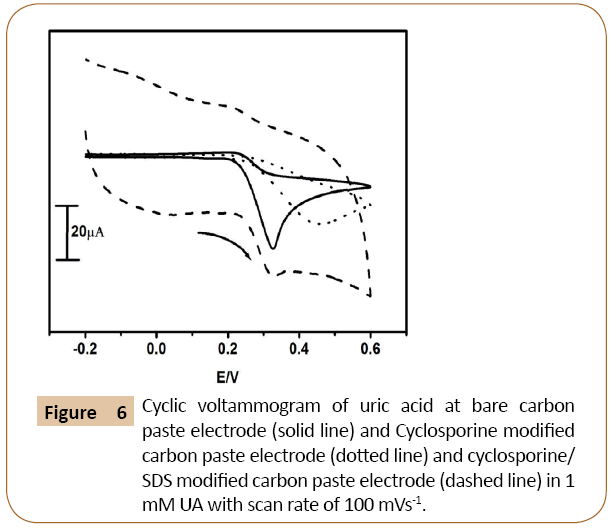
Figure 6: Cyclic voltammogram of uric acid at bare carbon paste electrode (solid line) and Cyclosporine modified carbon paste electrode (dotted line) and cyclosporine/ SDS modified carbon paste electrode (dashed line) in 1 mM UA with scan rate of 100 mVs-1.
Cyclic voltammetric response of AA at Cyclosporine/ SDS MCPE
Figure 7 shows cyclic voltammograms of ascorbic acid in 0.2 M phosphate buffer solution of pH 7.4 and scan rate 100 mVs-1 at Cyclosporine/SDS modified carbon paste electrode. The electrochemical response of ascorbic acid at modifying Cyclosporine carbon paste electrode showed decrease in the current signal in the absence of SDS and showed an increase in the current signal in the presence of SDS as compared to the bare carbon paste electrode. The presence of trace amount (10 μl of SDS) surfactant on the surface of cyclosporine modified carbon paste electrode has deeply influenced the enhancement of electrochemical responses. The cyclic voltammograms at modifying Cyclosporine carbon paste electrode shows decrease in current signal (dotted line) as compared to bare carbon paste electrode (solid line). The modified Cyclosporine/SDS carbon paste electrode showed a very good enhancement of peak current (dashed line) as compared to bare carbon paste electrode.
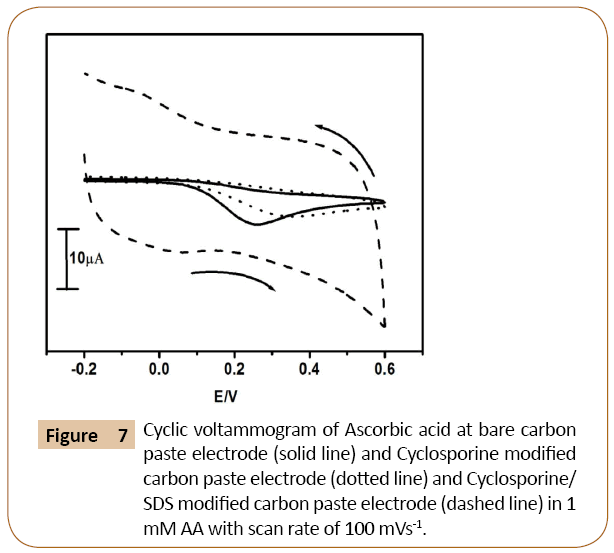
Figure 7: Cyclic voltammogram of Ascorbic acid at bare carbon paste electrode (solid line) and Cyclosporine modified carbon paste electrode (dotted line) and Cyclosporine/ SDS modified carbon paste electrode (dashed line) in 1 mM AA with scan rate of 100 mVs-1.
Simultaneous detection of dopamine, uric acid and ascorbic acid
The main objective of the proposed fabricated electrode was detection of dopamine, uric acid and ascorbic acid simultaneously, because they are co-exists in physiological sample. The Figure 8 shows the cyclic voltammograms of mixture sample having 1 mM dopamine, 2 mM uric acid and 1 mM ascorbic acid in 0.2 M phosphate buffer solution of pH 7.4 at bare carbon paste electrode (dashed line) Cyclosporine/SDS modified carbon paste electrode (dotted line). The bare carbon paste electrode resulting overlapped peak and it fails to locate individual anodic peak potential for dopamine, uric acid and ascorbic acid but the Cyclosporine/SDS modified carbon paste electrode clearly shows three well distinct peaks for dopamine, uric acid and ascorbic acid with great enhancement of anodic peak current. The observed result reveals the selectivity of the Cyclosporine/SDS modified carbon paste electrode towards dopamine, uric acid and ascorbic acid.
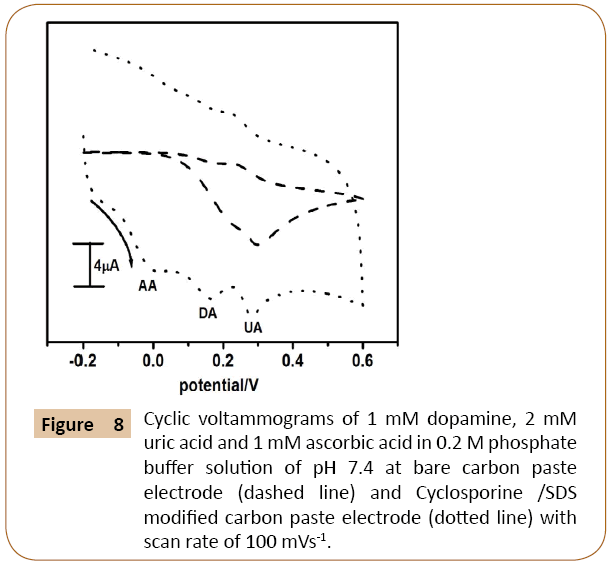
Figure 8: Cyclic voltammograms of 1 mM dopamine, 2 mM uric acid and 1 mM ascorbic acid in 0.2 M phosphate buffer solution of pH 7.4 at bare carbon paste electrode (dashed line) and Cyclosporine /SDS modified carbon paste electrode (dotted line) with scan rate of 100 mVs-1.
Analysis of real sample
Inorder to estimate the analytical efficacy of the fabricated Cyclosporine/SDS carbon paste electrode, it was used for quantitative determination of dopamine in pharmaceutical sample. The dopamine injection was purchased from Sterile Specialities India Pvt. Ltd with a specified content of dopamine of 40 mg/mL, the dopamine injection sample was diluted with 0.2 M phosphate buffer solution for suitable concentration. The results were shown in Table 1 it demonstrates a good performance of the fabricated Cyclosporine/SDS carbon paste electrode with acceptable recovery. The proposed drug based modified carbon paste electrode could be efficiently used for the determination of dopamine in injections with recovery in the range from 98-99.5%.
| Sample |
DA added (M) |
Found (M) |
Recovery (%) |
| 1 |
1×10-4 |
0.98×10-4 |
98.0 |
| 2 |
2×10-4 |
1.99×10-4 |
99.5 |
| 3 |
3×10-4 |
2.98×10-4 |
99.3 |
Table 1: Detection of DA in injection samples (n=3).
Conclusion
In the present work Cyclosporine/SDS modified carbon paste electrode was prepared by immobilization of SDS on the surface of Cyclosporine carbon paste electrode. The modified Cyclosporine/SDS carbon paste electrode shows increasing electrochemical sensitivity towards dopamine, ascorbic acid, uric acid as compare to the bare carbon paste electrode, it resolved the overlapping anodic peaks of dopamine, ascorbic acid and uric acid. The fabricated electrode can successfully used for the detection of dopamine in real sample. The developed method can also be used as other bioactive molecules in developing the modified sensor.
References
- Chen M, Kumar S, Anselmo AC, Gupta V, Slee DH, et al. (2015) Topical delivery of Cyclosporine A into the skin using SPACE-peptide. J Control Release 199: 190-197.
- Bierer BE, Hollander E, Fruman D, Burakoff ST (1993) Cyclosporin A and FK506: molecular mechanisms of immunosuppression and probes for transplantation biology. Curr Opin Immunol 5: 763-773.
- Borel JF, Weisinger D (1979) Studies on the mechanism of action of cyclosporin A. Br J Pharmaco I 66: 66-67.
- Kahan BD (1989) Medical Intelligence Cyclosporine. N Engl J Med 321: 1725-1738.
- Mahesh B, Pai KV, Gowda DV, Srivastava A, Raghundan HV, et al. (2015) Enhanced permeability of Cyclosporine from a transdermally applied nanoemulgel. Der Pharmacia Sinica 6: 69-79.
- Vishwanath CC, Swamy BEK, Sathisha TV, Madhu Anal GM (2013) Electrochemical Studies of Dopamine at LithiumZirconate/SDS Modified Carbon Paste Electrode: A Cyclic Voltammetric Study. Bioanal Electrochem 5: 341-351.
- Marceglia S, Foffani G, Bianchi A, Baselli G, Tamma F, et al. (2006) Dopamine-dependent non-linear correlation between subthalamic rhythms in Parkinson's disease. J Physiol 15: 579-591.
- Shankar SS, Swamy BEK, Chandra U, Manjunatha JG, Sherigara BS (2009) Simultaneous Determination of Dopamine, Uric Acid and Ascorbic Acid with CTAB Modified Carbon Paste Electrode. Int J Electrochem 4: 592-601.
- Wightman RM, May LJ, Michael AC (1988) Detection of dopamine dynamics in the brain. Anal Chem 60: 769A-793A.
- Guohua Z, Mingfang L, Mingli L (2007) Differential pulse voltammetric determination of dopamine with the coexistence of ascorbic acid on boron-doped diamond surface. C E J C 5: 1114-1123.
- Kemppainen N, Marjamaki P, Roytta M, Rinne JO (2001) Different pattern of reduction of striatal dopamine reuptake sites in Alzheimer's disease and ageing. J Neural Transm 108: 827-836.
- Shankar SS, Swamy BEK, Chandra U, Manjunatha JG, Sherigara BS (2009) Simultaneous Determination of Dopamine, Uric Acid and Ascorbic Acid with CTAB Modified Carbon Paste Electrode. Int J Electrochem Sci 4: 592-601.
- Scheller C, Sopper S, Jassoy C, Meulen T, Riederer VP, et al. (2000) Dopamine activates HIV in chronically infected T lymphoblasts. J Neural Transm 107: 1483-1489.
- Zheng J, Zhou X (2007) Sodium dodecyl sulfate-modified carbon paste electrodes for selective determination of dopamine in the presence of ascorbic acid. Bioelectrochemistry 70: 408-415.
- Sarre S, Michotte M, Herregodts YP, Deleu D, Klippel N (1992) High-performance liquid chromatography with electrochemical detection for the determination of levodopa, catecholamines and their metabolites in rat brain dialysates. J Chromatogr B 575: 207-212.
- Guan CL, Ouyang J, Li QL, Liu BH, Baeyens WRG (2000) Simultaneous determination of catecholamines by ion chromatography with direct conductivity detection. Talanta 50: 1197-1203.
- Salem FB (1987) Spectrophotometric and titrimetric determination of catecholamines. Talanta 34: 810-812.
- Tian FK, Guo L, Ru QY (1997) Voltammetric behaviour of dopamine at nickel phthalocyanine polymer modified electrodes and analytical applications. Analytica Chimica Acta 354: 343-349.
- Hui FU, Yu HC, Yu LS, Zhang K, Wei DZ (2010) Nanotechnology 21: 215601.
- Ashok A, Lucky HS (2010) The role of antioxidant therapy in the treatment of male infertility. Human Fertility 13: 217-225.
- Dursun Z, Pelit L, Taniguchi I (2009) Voltammetric Determination of Ascorbic Acid and Dopamine Simultaneously at a Single Crystal Au (111) Electrode. Turk J Chem 33: 223-231.
- Zhang L, Jiang X (2005) Attachment of gold nanoparticles to glassy carbon electrode and its application for the voltammetric resolution of ascorbic acid and dopamine. J Electroanal Chem 583: 292-299.
- Rajanchawla R (2014) Practical clinical biochemistry. Jaypee Brothers Publishers, New Delhi, India, pp:366.
- Jyh MZ, Jia JJ, Govindasamy I (1998) Selective voltammetric method for uric acid detection using pre-anodized Nafion-coated glassy carbon electrodes. Analyst 123: 1345-1350.
- Elena P, Yoshinobu K, Donald AT, Akira F (2000) Selective Voltammetric and Amperometric Detection of Uric Acid with Oxidized Diamond Film Electrodes. Anal Chem 72: 1724-1727.
- Adamson AW (1990) Physical chemistry of surfaces. 6th edn, Wiley, New York, USA, p: 808.
- Hiemenz PC (1986) Principles of surface and colloid chemistry. 3rd edn, Marcel Dekker, New York, USA, p: 672.
- Heyrovsky J, Kuta J (1966) Principles of polarography. Academic Press, New York, USA.
- Franklin TC, Mathew S (1989) The Use of Surfactants in Electrochemistry. Surfactants in solution. New York, USA, pp: 267-286.
- Fendler JH, Fendler E (1975) Catalysis in micellar and macromolecular systems. Academic Press, New York, San Fransisco, London, USA.
- Manjunatha JG, Swamy BEK, Deepa R, Krishna V, Mamatha GP, et al. (2009) Electrochemical studies of Dopamine at Eperisone and Cetyl Trimethyl Ammonium Bromide Surfactant modified Carbon paste electrode: A Cyclic Voltammetric Study. Int J Electrochem Sci 4: 662-671.
- Chandra U, Swamy BEK, Gilbert O, Pandurangacharya M, Reddy S, et al. (2010) Chin Chem Lett 211490.
- Gilbert O, Swamy BEK, Chandra U, Sherigara BS (2009) Electrocatalytic Oxidation of Dopamine and Ascorbic Acid at Poly (Eriochrome Black-T) Modified Carbon Paste Electrode. Int J Electrochem Sci 4: 582-591.
- Shankar SS, Swamy BEK, Chandra U, Manjunatha JG, Sherigara BS (2009) Simultaneous Determination of Dopamine, Uric Acid and Ascorbic Acid with CTAB Modified Carbon Paste Electrode. Int J Electrochem Sci 4: 592-601.
- Chandra U, Swamy BEK, Gilbert O, Sherigara BS (2010) Voltammetric resolution of dopamine in the presence of ascorbic acid and uric acid at poly (calmagite) film coated carbon paste electrode. Electrochimica Acta 55: 7166-7174.
- Manjunatha JG, Swamy BEK, Mamatha GP, Chandra U, Niranjana E, et al. (2009) Cyclic Voltammetric Studies of Dopamine at Lamotrigine and TX-100 Modified Carbon Paste Electrode. Int J Electrochem Sci 4: 187-196.
- Chitravathi S, Swamy BEK, Niranjana E, Chandra U, Mamatha GP, et al. (2009) Electrochemical Studies of Sodium Levothyroxine at Surfactant Modified Carbon Paste Electrode. Int J Electrochem Sci 4: 223-237.
- Niranjana E, Raghavendra Naik R, Swamy BEK, Bodke YD, Sherigara BS, et al. (2008) Cyclic Voltammetric Investigations of 3-aryl-4-bromo Sydnone and its Derivatives at Glassy Carbon Electrode. Int J Electrochem Sci 4: 980-992.