- (2013) Volume 14, Issue 6
Saurabh Jamdar1, Benoy I Babu1, Mahesh Nirmalan2, Maria Jeziorska4, Raymond FT McMahon3 and Ajith K Siriwardena1*
1Academic Hepatobiliary Unit, 2Critical Care Unit and 3Department of Academic Histopathology, Manchester Royal Infirmary; 4Cardiovascular Research Group, University of Manchester. Manchester, United Kingdom
Received: December 17th, 2012 - Accepted: May 7th, 2013
Context Microvascular thrombosis is a critical event in severe acute pancreatitis. Human recombinant activated protein C(Xigris®, Eli Lilly, Indianapolis, IN, USA) modulates the interplay between pro-inflammatory and pro-coagulant pathwaysand maintains microvascular patency. However, the anticoagulant properties of Xigris® may precipitate bleeding from theinflamed pancreas. Objective This study tests the hypothesis that Xigris® can ameliorate experimental acute pancreatitiswithout causing pancreatic haemorrhage. Methods Sprague Dawley rats were allocated as follows: Group 1: control (n=7);Group 2: acute pancreatitis (n=6); Group 3: administration of Xigris® 500 μg/kg body weight before induction of acutepancreatitis (n=6); and Group 4: Administration of Xigris® 500 μg/kg body weight 30 minutes after induction of acutepancreatitis (n=6). Acute pancreatitis was induced by intraperitoneal administration of L-arginine 300 mg/100 g bodyweight. Animals were sacrificed at 48 hours and biochemical, haematological, and histological markers of pancreatichaemorrhage and inflammation assessed. Results Median lipase in animals with acute pancreatitis was 10 U/mL (range: 7-16 U/mL) compared to 5.5 (range: 3-8 U/mL) in controls (P=0.028). Lipase was also elevated in animals given Xigris® bothbefore (12 U/mL, range: 8-22 U/mL; P=0.031 vs. control group) and after (46 U/mL, range: 9-71 U/mL; P=0.015 vs. controlgroup) induction of acute pancreatitis). Haemoglobin levels were similar among all groups (P=0.323). There was nohistological evidence of pancreatic haemorrhage in animals treated with Xigris®. Pre-treatment with Xigris® was associatedwith a significant reduction in pancreatic injury. This effect was absent when Xigris® was administered after induction ofacute pancreatitis. Conclusion Xigris® did not lead to pancreatic haemorrhage in experimental acute pancreatitis.Administration of Xigris® prior to induction of acute pancreatitis was associated with amelioration of injury. This effect wasnot seen with administration of Xigris® after induction of acute pancreatitis.
Arginine; drotrecogin alfa activated; Pancreatitis, Acute Necrotizing; Protein C; Recombinant Proteins
BAL: bronchoalveolar lavage
Over the last decade, human recombinant activated protein C (Xigris®, Eli Lilly, Indianapolis, IN, USA) became established in the management of severe sepsis. This was primarily based on the publication of the PROWESS study in 2001 [1]. This major world-wide study of Xigris® in severe sepsis randomized 1,690 patients (850 to receive Xigris® and 840 to receive placebo). Non-stratified analysis of results showed 259 deaths (30.8% mortality) in the placebo group compared to 210 (24.7%) in the treatment arm. This difference was highly significant and persisted at 28 days [1]. A likely principal side effect of modulation of the coagulation cascade is haemorrhage and there were 30 serious bleeding events in the treatment arm of the PROWESS study compared to 17 in the placebo group; however, this difference was not statistically significant (P=0.06). Xigris® rapidly became an essential component of management algorithms for the treatment of patients with severe sepsis [2].
Despite widespread use of Xigris® persistent doubts regarding its efficacy and safety remained. In response to these doubts the PROWESS-SHOCK trial was conducted [3]. One thousand, six hundred and ninety-seven patients with infection, systemic inflammation and shock were randomised to receive either Xigris® or placebo. In comparison to placebo, administration of Xigris® did not reduce mortality at either 28 or 90 days following the commencement of treatment. Publication of the findings of this study earlier this year provided robust data against the continued use of Xigris® in severe sepsis and as a consequence the manufacturer withdrew the agent from the marketplace as a treatment for sepsis.
Within the PROWESS study 62 patients with acute pancreatitis and severe sepsis received Xigris®. Subgroup analysis of these patients demonstrated a trend towards a reduction in mortality (24% vs. 15%) in those treated with Xigris® [1]. It is important to emphasise that the decision to withdraw Xigris® was made on the basis of a lack of efficacy and not of a lack of safety and whether treatment with Xigris® could modify the disease course in acute pancreatitis remains an unanswered question.
Early acute pancreatitis is characterized by an intense systemic inflammatory response rather than intra-abdominal sepsis [4]. There is evidence both from experimental models [5, 6] and from clinical studies [7, 8] that microvascular thrombosis in the pancreatic vascular bed is a mediator of pancreatic parenchymal necrosis and is also involved in the endothelium-inflammatory cell interplay. Although these findings would support (in theory) the use of activated protein C “early” in the disease course of acute pancreatitis, one of the major causes of death in the severe form of this disease is peri-pancreatic haemorrhage [9], raising concerns about the safety of a drug with anticoagulant properties.
Integration of current knowledge of the pathophysiologic changes early in acute pancreatitis with clinical information from large-scale trials of Xigris® in sepsis highlight a potential novel indication for this drug (as a disease modifier early in the course of acute pancreatitis). Given the risks of pancreatic haemorrhage in pancreatitis and the limited information regarding the effect of this agent during pancreatic inflammation, it is necessary to evaluate the effect of Xigris® in an experimental model of acute pancreatitis. The present study thus tests the hypothesis that Xigris® will reduce pancreatic injury in a well-validated experimental model of acute pancreatitis without causation of pancreatic parenchymal or peripancreatic haemorrhage. In order to test this hypothesis, we have selected the L-arginineinduced experimental pancreatitis model as this does not involve physical manipulation of the pancreas. The principal aim of these preliminary studies is to assess whether there is any evidence of exacerbation of pancreatic haemorrhage induced by Xigris®.
L-arginine-Induced Acute Pancreatitis Model
The intra-peritoneal L-arginine-induced model of acute pancreatitis was first described by Mizunuma [10]. Tani et al. reported that 500 mg/100 g body weight of intraperitoneal L-arginine reliably induces pancreatitis with 70-80% pancreatic necrosis [11]. Their report, however, does not publish mortality data in animals receiving this concentration of L-arginine. Unpublished data from our group have examined concentrations of 200 mg/100 g body weight through to 500 mg/100 g body weight. A high mortality is associated with concentrations above 400 mg/100 g body weight (JJ Powell; personal communication). We selected a concentration of 300 mg/100 g body weight to attain severe pancreatic injury without associated high mortality.
Animal Care
Twenty-five adult male Sprague-Dawley rats with a median weight of 280 g (range: 230-340 g) were allowed access to food and water ad libitum. Prior to the commencement of the experiment the animals were housed in metabolic cages with controlled temperature and 12-hour light-dark cycles for at least 1 week. During the course of the experiment animals were monitored in compliance with current animal care regulations and analgesia and supplemental fluids administered.
Group Allocation
Rats were randomly allocated to four groups: one control groups and 3 induced acute pancreatitis groups. One acute pancreatitis group had no Xigris® treatment (acute pancreatitis with sham intervention), while Xigris® was administered before or after induction of pancreatitis in the other two groups, respectively (Table 1).
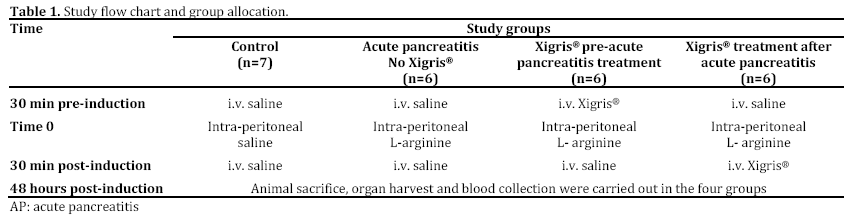
Induction of Pancreatitis
A single intraperitoneal injection of L-arginine diluted in sterile 0.15 M saline was administered to unanaesthetised rats to induce pancreatitis. A concentration of 300 mg per 100 g body weight prepared as a 30% (weight/volume) solution was given. The volume of injection administered was limited to 10 mL/kg body weight.
Concentration of Xigris®
Xigris® used in this study is produced by recombinant techniques using human tissue. When used in animals cross-reactivity leads to antibody formation if used for more than 48 hours (Charles Brigden, Eli Lilly, Indianapolis, IN, USA; personal communication). In rats, Xigris® should be given by intravenous bolus injection and exhibits a biphasic half-life (t1/2 alpha is 13 minutes and t1/2 beta is 30 minutes) with 90% eliminated by hepatic metabolism within 2 hours. In man, the beneficial effects on coagulation cascades and microcirculation are sustained after a single bolus dose.
In a randomised controlled trial of intravenous Xigris® in severe sepsis [1], a dose of 24 μg/kg/h over a 96-hour period (576 μg/kg per day) was found to reduce mortality. A review of published literature examining the role of Xigris® in various experimental animal models of acute inflammation reported a range of bolus intravenous concentrations (from 100 μg to 2 mg per kg body weight) given both prophylactically (10-30 minutes pre-insult) and post-insult (up to 60 minutes after), which ameliorated the injury [8]. Consequently a bolus concentration of 500 μg per kg body weight from the mid-range of reported efficacy has been adopted in this study.
Animal Sacrifice and Tissue Collection
Animals were sacrificed at forty-eight hours after induction of pancreatitis under terminal anaesthesia with halothane. Immediately prior to sacrifice (under anaesthesia) a laparotomy was performed to assess for evidence of peri-pancreatic haemorrhage. Death was confirmed by exsanguination. Blood samples were collected immediately after sacrifice by cardiac puncture for biochemical and haematological analysis. Pancreata and lungs were excised immediately. The head of the pancreas and apex of the lung were buffered in 4% formalin at 4oC for 24 hours prior to haematoxylin-and-eosin (H&E) staining.
Assessment of Blood Samples
Samples were analysed for full blood profile, amylase and lipase at a regional veterinary laboratory (Central Diagnostic Services, The Queen's Veterinary School Hospital, Cambridge, United Kingdom) using an AutolabTM system (Metrohm AG, Utrecht, The Netherlands).
Assessment of Lung Injury
Bronchoalveolar lavage (BAL) protein content
Bronchoalveolar lavage of the right lung was performed with 2 mL saline containing 0.07 mol/L ethylenediamine tetra-acetic acid. The procedure was repeated three times to produce a combined lavage of approximately 4 mL which was centrifuged at 1,500 g for 20 min, frozen at -20oC, and subsequently assayed for protein concentration.
Histological Evaluation
Haematoxylin and eosin (H&E) stained slides were prepared from formalin fixed lung. A pathologist blinded to group allocations assessed slides qualitatively.
Assessment of Pancreatic Injury
H&E stained slides were prepared from formalin fixed pancreas. A pathologist blinded to group allocations assessed slides using the histological scoring system reported by Schmidt et al. (Table 2) [12]. The composite histological injury score was calculated as sum of the 4 scores (oedema score; acinar necrosis score; haemorrhage and fat necrosis score; inflammation and perivascular infiltrate score).

The study was conducted under a home office license with ethics committee approval and conformed to University of Manchester ethical guidance on animal welfare during experimentation. All animals received humane care according to the criteria outlined in the “Guide for the Care and Use of Laboratory Animals (1996)” prepared by the National Academy of Sciences.
Data are expressed as median (range) unless otherwise stated. Statistical analyses were undertaken by Kruskal-Wallis with post-hoc analyses using unpaired nonparametric test (Mann- Whitney) unless otherwise stated. Statistical significance was accepted at the two-tailed P<0.05 level, but for post-hoc analyses Bonferroni adjustment was applied. The GraphPad Instat statistics package (GraphPad Software, San Diego, CA, USA) was used.
RESULTS
Biochemical Analyses
Plasma Lipase (Figure 1a)
Plasma lipase differed significantly among groups (P=0.011; Kruskal-Wallis). The lipase in animals with acute pancreatitis was 10 U/mL (range: 7-16) U/mL compared to 5.5 U/mL (range: 3-8 U/mL) in control animals (P=0.028; Mann-Whitney). Lipase was also elevated in animals given Xigris® prior to induction of acute pancreatitis (12 U/mL, range: 8- 22 U/mL; P=0.031 vs. control group) and in the group receiving Xigris® after induction of acute pancreatitis (46 U/mL, range: 9-71 U/mL; P=0.015 vs. control group. The lipase in animals treated with Xigris® after the induction of acute pancreatitis was significantly different from the acute pancreatitis group (P=0.033) while no significant difference was found for animals treated with Xigris® before the induction of acute pancreatitis (P=0.629).
Plasma Amylase (Figure 1b)
Plasma amylase levels did not significantly differ among groups (P=0.287; Kruskal-Wallis).
Haematological Analyses
Haemoglobin (Figure 2a)
Haemoglobin levels were similar in all groups. (P=0.323; Kruskal-Wallis). In keeping with this, no evidence of peri-pancreatic or intra-abdominal haemorrhage was identified in any animal at laparotomy.
Haematocrit (Figure 2b)
Haematocrit was significantly different among groups (P=0.017; Kruskal Wallis). Haematocrit in animals receiving Xigris® after induction of pancreatitis was significantly elevated compared to control animals (P=0.009; Mann-Whitney).
Lung Histology
Qualitative histological assessment of lung tissue revealed no significant evidence of injury in any group.
Lung BAL Protein Content (Figure 3)
There was no significant difference in lung BAL protein content among groups (P=0.633; Kruskal- Wallis)
Pancreatic Histology
H&E preparations of pancreatic parenchyma in the control group is shown in Figure 4a. Induction of pancreatitis was associated with diffuse injury characterised by oedema, polymorphonuclear leukocyte infiltration and acinar cell disruption. There were also areas of haemorrhage and necrosis (Figure 4b). Xigris® given prior to induction was associated with a significant reduction in the degree of pancreatic injury (Figure 4c). In contrast, Xigris® administered after induction of acute pancreatitis was associated with similar patterns of injury to the acute pancreatitis group (Figure 4d). There was no associated increase in pancreatic parenchymal haemorrhage in either group receiving Xigris®. Histological injury scores were significantly different among groups (P<0.001; Kruskal-Wallis). Post-hoc analysis with Bonferonni adjustment revealed higher scores in the acute pancreatitis group in comparison to both control and the group receiving Xigris® prior to induction of acute pancreatitis (control vs. acute pancreatitis: P=0.001; acute pancreatitis vs. Xigris® pre-acute pancreatitis: P=0.004; Mann-Whitney) (Figure 5). Scores in the acute pancreatitis group differ significantly from the Xigris® post-acute pancreatitis group (P=0.039; Mann-Whitney). With reference to haemorrhage there was no significant difference among groups (P=0.196; Kruskal-Wallis) (Figure 6).
Figure 4. H&E preparations of pancreatic parenchyma in the four groups. a. Representative control animal (original magnification x10). b. Representative L-arginine-induced acute pancreatitis animal. Note extensive disruption of acinar architecture with necrosis and haemorrhage (original magnification x10). c. Animal pre-treated with Xigris®. Note in particular the preservation of acinar cell architecture and the absence of haemorrhage (original magnification x40). d. Animal treated with Xigris® after the induction of pancreatitis. Note extensive disruption of acinar architecture, oedema and inflammatory cell infiltrate (original magnification x10).
Diminished protein C generation and low levels of protein C are key features of systemic inflammation and in particular clinical severe acute pancreatitis [13]. This has led to the development of human recombinant activated protein C (Xigris®). In patients with acute pancreatitis, the disease-related risk of intra-abdominal haemorrhage has raised specific concerns about the safety of a drug with anticoagulant properties. The present study investigates whether administration of Xigris® ameliorates pancreatic injury in a well-validated experimental model of acute pancreatitis. Crucially, this study also examines whether Xigris® exacerbates pancreatic haemorrhage. The Larginine induced model of pancreatitis in the rat is, in theory, ideal for this purpose as it involves no physical manipulation of the pancreas (and therefore no iatrogenic predisposition to pancreatic haemorrhage), is associated with haemorrhagic necrosis and the rat pancreatic injury correlates well to that seen in man [11]. There is a narrow range of injury with the concentration used in this study giving rise to acute pancreatitis with necrosis but without lung injury. When higher concentrations are used, there is high lethality. This limitation of the L-arginine experimental acute pancreatitis model is now more widely recognised (Z Raconzay; personal communication) but was accepted for the present study, in particular as there was no pancreatic manipulation.
The administration of L-arginine in this study resulted in hyperlipasemia and characteristic histological features of acute pancreatitis with oedema and patchy necrosis at forty-eight hours. Amylase levels at forty-eight hours were not significantly different. This is in keeping with previous reports of this model as serum amylase has an early peak and has usually normalised by forty-eight hours [11].
Xigris® given prior to induction of acute pancreatitis was associated with a significant reduction in the degree of pancreatic injury. In contrast Xigris® given after induction of acute pancreatitis was associated with similar patterns of injury to the acute pancreatitis group. These findings can be interpreted as suggesting that the presence of systemic Xigris® ameliorates L-arginine-induced injury whereas administration of Xigris® after induction of injury failed to modulate acute pancreatitis.
An important and original finding of this work is that there was no histological evidence of exacerbation of pancreatic parenchymal haemorrhage in animals treated with Xigris® (in concentrations mirroring those used in the human sepsis treatment setting and in the context of pancreatic inflammatory injury). Equally there was no sign of intra-abdominal haemorrhage at laparotomy and in keeping with this there was no change in haemoglobin measurements in either treatment group. The rise in haematocrit in animals receiving Xigris® after induction of acute pancreatitis may be a chance finding but may also be representative of an effect of Xigris® on microvascular permeability in the presence of endothelial injury and requires further evaluation.
We conclude from these studies that Xigris® given prior to induction of experimental acute pancreatitis results in amelioration of injury. When given after induction of acute pancreatitis, these beneficial effects were not seen in this study. There was no evidence that intervention with Xigris® (in animals with experimental acute pancreatitis) was associated with either intra-abdominal or intrapancreatic haemorrhage. Given the lack of demonstrated benefit from intervention with Xigris® after induction of acute pancreatitis our study is an important and negative caveat to work suggesting that early intervention with this drug modifies the course of experimental acute pancreatitis [5, 6, 14, 15]. The results of this study have to be set in the context of our earlier study in caerulein-induced experimental acute pancreatitis [15]. Although the pancreatic injury was more severe in this earlier study, similarities in findings were that there was no Xigris® induced pancreatic parenchymal haemorrhage and Xigris® also modulated pancreatic injury [15].
The question of whether this agent can modify the complex clinical course of the human disease remains unanswered and we would caution against over-extrapolation to the clinical setting from experimental studies. To take this work forward, further experimental studies may be of benefit. Specifically, evaluation of the effect of Xigris® in another experimental model such as that associated with taurocholate could be useful (although this would involve manipulation of the pancreas) and in particular, in a model with a more representative pulmonary injury [16, 17].
We acknowledge the input of David Schofield of the Pancreatic Laboratory of the Manchester Royal Infirmary, Manchester , United Kingdom
The authors have no potential conflict of interests