Keywords
Heavy metals; Chelating agents; Metal intoxication
Introduction
The oldest toxins known to mankind probably are heavy metals. For example, lead was used in the preparation of silver in 2000 BC. Hippocrates first described in 370 BC, abdominal colic in a person who extracted the ore. Arsenic was used to decorate Egyptian tombs. Heavy metals are different from other toxins for the reason that neither people nor microorganisms produce it, nor can it be destroyed.
The term "heavy metal" is often used for the group of elements that are associated with environment pollution and have potential toxic effects on wildlife. Despite not having a single scientific definition of heavy metals from the chemical standpoint, this term most often includes elements whose density is greater than 5 or 6 g cm-3 [1]. Due to the high toxicity of metals in this group, beryllium is added to it (1.85 g cm-3) as well as metalloids arsenic and antimony [2]. According to the latest recommendations of the competent international bodies, classification of metals on heavy and light should be based not only on the periodic table of elements and their chemical properties, but also on assessment of the impact of toxic metals in the environment [3]. With the chemical, physiological and environmental aspects in mind, heavy metals constitute a heterogeneous group of elements.
In most of the land, plants and animals, heavy metals are found in low concentrations (of the order of mg kg-3 or less) and are therefore called “trace elements” [4]. Wasteful and toxic trace elements include cadmium, lead, chromium and mercury and metalloid arsenic. In the last decades copper, zinc, iron, manganese, molybdenum, boron, cobalt, nickel, lead, cadmium, chromium, arsenic, mercury and selenium have all been extensively studied, while from the viewpoint of ecology copper, zinc, lead and cadmium are of maximum interest due to the very frequent pollution of water and soil by them and their entering the food chain [5].
Many heavy metals are essential for all organisms, for example, as active centers of an enzyme. In higher concentrations than optimal, however, they are toxic. Due to the importance of this phenomenon and the damage it can inflict on nature and, above all, humans, heavy metal toxicity and mechanisms of toxicity have been a subject of intensive research for many years [6]. In this paper, we have tried to present the most important heavy metals and chelating agents as a treatment modality for intoxication in a comprehensive and transparent way.
Mercury (Hg)
Mercury is the only liquid metal. Natural source of mercury represents the evaporation of the earth's crust (the bottom of the ocean, rivers, land). It exists in the elemental form, and as the Hg (I) and Hg (II) compounds, in the organic or inorganic form. Artificial sources of mercury are of industrial origin: paints, batteries, electrical equipment, cement, explosives, followed by fungicides, amalgams. Methyl mercury and other organic mercury compounds are formed as a product of microbial activity (methane bacteria) in an aqueous medium and are mainly located in fish and fish products, and the average intake is about 3.6 mg/day to 0.6 mg/day from fish. Estimated total daily intake of mercury in the 11 EU countries for adults is 1.1-22 mg. Table 1 Mercury content in foods (μg kg-1).
| Type of food |
Content |
| cereals |
4-22 |
| dairy products |
<1-5 |
| fish |
40-250 |
| vegetables |
1-10 |
| meat: beef |
1-16 |
| beef liver |
2-40 |
| beef kidney |
1-136 |
| other foods |
<1-5 |
Table 1: Mercury content in foods (μg kg-1).
All three forms of mercury are toxic, even though the organic forms are the most common and potentially dangerous. Elemental mercury vapor evaporates and reaches the air, and from there spreads into the water and land. In the cell it is oxidized to divalent inorganic mercury in the presence of the enzyme catalase. Mercury vapors are liposoluble and easily pass through the blood-brain barrier and the placenta.
Inorganic mercury is poorly absorbed in the gastrointestinal tract (7-15%). The highest concentrations of inorganic mercury can be found in the kidney. Methyl mercury is well absorbed in the intestine, about 10 % is distributed to the brain, 5% remains in the blood, and the rest is transferred to other tissues [7].
The mechanisms of toxicity of mercury compounds vary and include the non-specific cell damage and cell death, inhibition of the enzyme activity, the termination of DNA and protein synthesis, autoimmune reaction [8]. The most toxic organic mercury compound is, primarily, methyl mercury, which adversely affects the nervous system in particular parts of the brain which is responsible for the coordination and sensory function. High toxicity can be explained by liposolubility of organic compounds. Methyl mercury is not carcinogenic [9].
Lead (Pb)
The natural environment lead has four stable isotopes found in the following ratio: Pb - 204–1.48%, Pb - 206– 23.6%, Pb - 207–22.6%, Pb - 208–52.3%. The most common lead isotopes in the nature of the end products of three natural radioactive series. With lead-206 ends the series of uranium-238, lead- 207, series of uranium-235 and lead-208 is the last member of a series of thorium-232 is well known and over twenty of radioactive isotopes of lead. Lead is a metal that is widely used in various industries, but also is one of the persistent pollutants that cause significant adverse effects on living organisms and the ecosystem as a whole. The particles of lead are present in a wide range of concentrations in all parts of the environment, air, water and soil.
In nature, most lead appears as Pb2+ ion, a compound of his largely insoluble in water. Lead is a trace element in the Earth's crust whose average content estimated at 15 mg kg-1 [10]. There are a number of lead minerals, and because of their strong affinity for sulfur that is concentrated in rocks and minerals sulfide. In significant quantities of a galena (PbS), anglesite (PbSO4) and solder (PbCO3) [11]. Due to the specific physical and chemical properties of the lead for many years used for different purposes. Besides the combustion of leaded gasoline in internal combustion engines, significant anthropogenic sources of lead mines or smelters, industrial processes, the use of sewage sludge in agriculture.
Lead belongs to the group of very toxic heavy metals [12]. It can enter the body through inhalation aerosol entering the contaminated water and food and through skin contact [13]. Pb absorbed into the bloodstream and soft tissue [14], and the largest part shall be deposited in the bones. The toxic effects of lead are different for adults and children. In adults occur abdominal pain, anemia, renal dysfunction, headache, ataxia, peripheral neuropathy with impaired long neurons, and children are the most important clinical consequences of mental retardation and disorders in speech, consciousness and behavior. Lead is a cumulative poison, so that the chronic effects that occur due to prolonged intake of lead in the body including neurological and gastrointestinal disorders, anemia, kidney damage, interfere with the psycho - physical development of children [15].
Cadmium (Cd)
Cadmium is identified Schwarz 1974 [16], and in nature is present at low concentrations, naturally linked to the zinc, lead or copper. High levels of cadmium present in nature are associated with the industry. Soluble cadmium plants quickly absorbed contaminants thus indirectly the soil and water. This metal has a long half-life and persists throughout the years in nature [17,18]. Characterized as one of the 126 largest zagđivača environment by the Agency for Environmental Protection USA. Permanent sources of cadmium contamination related to its application in the industry, as well as anti-corrosion agent, stabilizer in PVC products and the manufacture of tires, paint and pigment in the manufacture of Ni-Cd batteries. The most important source of cadmium contamination for the general population is hrana. Most foods contain traces of Cd, and the maximum amount is giblets, shellfish, mushrooms, some plants -can concentrate Cd (cocoa, rice) [10,19].
Cadmium is introduced into the body in the form of vapor and dust particles, absorbs mainly respiratory route, a small part of the gastrointestinal tract. The largest source of inhalant intoxication with cadmium is smoking. Over 50% of tobacco smoke cadmium is absorbed from the lungs into the systemic circulation during active smoking (19). According to physical-chemical properties of the cadmium has a specific binding affinity for sulfur, as they can easily inhibit the physiological activity of the enzyme with the active -SH groups. One of the foundations of cadmium toxicity and its effect on the enzyme systems of cells, due to replace their metal ions (mainly Zn2+ and Cu2+) in metaloenzymes [20]. Acute cadmium poisoning in laboratory animals resulting in liver damage, while chronic exposure leads to renal failure, anemia, immune disorders and bone lesions. In contrast to acute chronic intoxication leads to the development of some diseases, such as. Chronic obstructive pulmonary disease, renal disease (nephrotoxicity) and bones (arthritis, osteoporosis), anemia, and other growth disorder [21].
Zinc (Zn)
Zinc is one of the essential biogenic elements. It is widespread in the Earth's crust at a concentration of 52 to 80 mg kg-1 [10]. The content of zinc in igneous rocks is fairly uniform, while in sedimentary rocks are concentrated in sediments emerged from shale. Zinc is mostly appears in the Zn2+ oxidation state and has great potential for the formation of different compounds with organic and inorganic groups. The main zinc ores are sphalerite (ZnS) and smithsonite (ZnCO3). Zinc is relatively low toxic metal. Overall, toxicity was limited to acute overdose, for example, in workers who inhaled dust particles with high content of zinc or zinc fumes ("metal fever"). The introduction of high doses of Zn, especially its inorganic compounds, can be harmful to the digestive tract. Ingestion of high concentrations of zinc for several months may cause anemia, damage the pancreas and disorders in the metabolism of high density lipoprotein (HDL) [22].
Copper (Cu)
Copper is the 26th most abundant element in the lithosphere, immediately after zinc. Sometimes it can be found in the elemental form, but is usually in the form of individual primary minerals and complex sulfide. Reserves of copper in the soil consist mainly of secondary minerals (melakonit, hidrotenorit, azurite, etc.). As the average copper content in the lithosphere according to data from the 70 mg kg-1, and in the Earth's crust interval of 24-55 mg kg-1 [23], ie 27-75 mg kg-1 [24]. According to Alloway in (1995) the average value of the copper concentrations in the soils of the world ranges from 20 to 30 mg kg-1.
Copper is an essential element for humans and is composed of several proteins and metal- enzymes. His participation is important in some oxidation-reduction processes and the synthesis of hemoglobin. Although it is used in various industries, in humans have been observed cases of occupational poisoning copper. Except in cases of extreme or contaminating the food with water, the only important example of intoxication with copper is associated with Wilson's disease - a rare disorder of metabolism when there is an increase of absorption by the digestive system of copper and a high accumulation of copper in the liver and brain.
Arsenic (As)
Metalloid arsenic is widespread in the environment, but it is rarely found in the elemental form. It is capable of forming a variety of toxic compounds, and its organic compounds are predominating in the soil. Arsenic occurs primarily in the oxidized state in the form of arsenate As5+ and arsenite As3+ which is toxic [25]. Geochemical behavior of arsenate in the soil is analog to phosphate due to close values of ionic radii of As5+ and phosphorus. As phosphates, arsenates are fixed in the soil and are therefore relatively immobile. Arsenic has been identified in more than 200 minerals, of which the largest number of arsenate, sulphide and sulphate salts. It is most often found in sulphide minerals arsenopyrite (FeAsS) auripigment (As2S3) and realgar (AsS). Due to dissolution of minerals, arsenic is naturally found in groundwater. When using such water people can be exposed to low doses of arsenic. There are regions in the world with unsafe drinking water due to increased arsenic concentrations, which poses a particular risk to human health.
Toxic arsenic effects are derived from trivalent arsenic. It is linked to the SH groups of proteins and enzymes in the mitochondria involved in the Krebs cycle. In this way, arsenic inhibits energy production in mitochondria, which leads to decreased production of ATP in the cells and increased production of peroxides and free radicals which cause oxidative stress, DNA damage, and cell death. Symptoms of arsenic intoxication include fever, vomiting, diarrhea, abdominal pain, in the case of acute poisoning. Symptoms of chronic intoxication are skin changes (hyperkeratosis, hyperpigmentation, dermatitis) and nail changes (characteristic transverse white lines on nails that occur around 6 weeks of the start of arsenic poisoning) [26].
Nickel (Ni)
The nickel content in the Earth's crust is estimated to be 80 mg kg-1 [27]. According to its amount, it is the 23rd most common element on Earth. It is also often associated with carbonates, phosphates and silicates. Volcanic rocks rich in ferro-magnesium minerals (pyroxene, olivine) and sulphides are also rich in nickel. These minerals are nickel-substituted Fe and Mg because of the similarity of their ionic radii. Organic matter has a strong ability to absorb Ni, so that it is quite concentrated in coal and oil.
It was found that nickel has carcinogenic effects and causes changes in the respiratory tract with occurrences of tumors. In addition to being genotoxic, nickel can cause problems with the reproductive organs and neurological and cardiovascular disorders. Ingestion and inhalation are the major routes of nickel intake into the body, with up to 55% of the nickel particles retained in the lung while inhaling. Nickel absorption through the skin is associated with the occurrence of dermatitis.
Heavy Metals in Plants
Since the mid-18th century, or the beginning of the Industrial Revolution to the present day toxic heavy metals pollution of the biosphere is growing rapidly. It has dramatically increased in the last century due to the development of various industries such as mining, but also because of waste left unprovided for and contamination of agricultural land. Heavy metal pollution therefore increasingly represents a major environmental and health problem for all living beings. In contrast to organic pollutants, heavy metals are persistent environmental pollutants that can not be chemically or biologically degraded. Previously used methods of removal such as dissolution, excavation, encapsulation, covering the contaminated ground with clean land and even their recycling, are often not effective, but also destructive and very expensive.
For these reasons over the last three decades, there's been a special interest in phytoremediation, or the use of plant species of interesting properties as a cheap and efficient way of removing heavy metals from contaminated soil. Plants that naturally live in these fields have developed mechanisms of tolerance to heavy metals, which enable them to reduce entry of heavy metals in the plant tissue and their retention and detoxification in the roots with a minimum transfer to leaves where they would cause damage [28]. Although these features are critical to the survival, growth and reproduction of plants growing in unsuitable habitat conditions polluted by heavy metals, from the aspect of phytoremediation they are not ideal because the metals remain in the root system, making them difficult to remove. However, a promising approach to solving the problem of heavy metals pollution using plants is due to the phenomenon of hyperaccumulating metals in plant species. These species are not only tolerant to heavy metals, but also have the additional ability to accumulate heavy metals from the soil in their tissues [29]. Such plant species we call hyperaccumulative. Hyperaccumulative plants unlike the ones tolerant to heavy metals are actively taking large amounts of one or more different heavy metals, transfering them to the plant organs above the ground (eg. leaves) and are storing up to 100-1000 times higher concentrations of the metal than nonhyperaccumulative species, without symptoms of toxicity [28]. Hyperaccumulative species have three main characteristics that distinguish them from common species: increased rate of entry of heavy metals into the plant, faster and more successful transfer from root to sprout and their markedly increased ability of detoxification and separation in the leaves [28]. The use of plants for metal phytoextraction has since been the subject of many studies [30,31]. In order for a plant to be classified as hyperaccumulative, it must be able to accumulate a certain amount of heavy metals: more than 10 mg of Mn or Zn per gram of plant dry matter (1% of its weight), more than 1 mg of As, Co, Cr, Cu, Ni, Pb, Sb, Se and Tl per gram of plant dry matter (0.1% of its weight) or more than 0.1 mg of Cd per gram of plant dry matter (0.01% dry weight) in the offspring without symptoms of phytotoxicity [28].
Table 2 Contents of metals in plants (mg kg-1 dry weight) are defined for the purpose of categorizing the status of the accumulative or hyperaccumulative plant species [32].
| Element |
Accumulative treshold |
Hyperaccumulative treshold |
Number of hyperaccumulative species |
| As |
- |
1000 |
Not found |
| Cd |
20 |
100 |
1 |
| Co |
20 |
100 |
28 |
| Cr |
50 |
100 |
Unknown |
| Cu |
100 |
1000 |
37 |
| Mn |
2000 |
10000 |
9 |
| Ni |
100 |
1000 |
317 |
| Pb |
100 |
1000 |
14 |
| Se |
10 |
1000 |
20 |
| Zn |
2000 |
10000 |
11 |
Table 2: Contents of metals in plants (mg kg-1 dry weight) are defined for the purpose of categorizing the status of the accumulative or hyper accumulative plant species (Boyd, 2011).
So far it is known about 450 hyperaccumulating species - less than 0.2% of all known plant species - of which the majority hyperaccumulative Ni (75%) [29]. These types of plants belong to many families including members of the Asteraceae, Brassicaceae, Caryophyllaceae, Cyperaceae, Cunouniaceae, Fabaceae, Flacourtiaceae, Lamiaceae, Poaceae, Violaceae and Euphobiaceae. The best known hyperaccumulating plant species is Thlaspi caerulescens, which accumulates large amounts of Zn (39 600 mg kg-1) and Cd (1800 mg kg-1) without visible damage [33]. This small, diploid plant can easily be grown in the laboratory, which is an excellent experimental system for studying the mechanisms of adoption, accumulation and tolerance associated with metal phytoextraction. Hyperaccumulative Zn plant species are less common, and include Arabidopsis halleri and Thlaspi species, as well as Sedum alfredii. Species A. halleri and S. alfredii, together with T. caerulescens and T. praecox are the four best known Zn and Cd hyperaccumulating species. Recently Solanum nigrum (potato) is recognized as the fifth cadmium hyperaccumulator. Species that hyperaccumulate selenium is spread in different families, among which are Fabaceae, Asteraceae, Rubiaceae, Brassicaceae, Chenopodiaceae and Scrophulariaceae. Some angiosperms, such as Brassicaceae Isatis cappadocica and Hesperis persica, or some fern species belonging to the genus Pteris identified as arsenic hyperaccumulative. Arsenic is very well accumulated by the fern Pteris vittata L. (Pteridaceae) and its related species [34]. For cadmium accumulation, Cd/Zn hyperaccumulating plant species Thlaspi caerulescens J. Presl & C. Presl (Brassicaceae) can be used. Berkheya coddii Roessler (Asteraceae) is known as the nickel hyperaccumulator [35,36]. Fern Athyrium yocoscense Christ (Athyriaceae) is known for its outstanding tolerance for lead and it can accumulate over 1 000 mg of lead per gram of dry matter [37]. Nitzschia palea (Kützing) W. Smith hyperaccumulates cadmium by special protein family - frustulins [38].
Arabidopsis halleri and Thlaspi careulescens species are model planta in which hyperaccumulating mechanisms are explored at the physiological and molecular level. However, some properties of hiperacumulating plant species limit their use in phytoremediation: accumulation of only one specific metal, adaptation to specific environmental conditions, making them geographically limited, impossibility of growth as conventional crops, slow growth, shallow penetration of the root system into the ground, small biomass and often very short life cycle, hiperacumulating plant species not foundfor some metals. Table 3 Examples of suitable plant species for metal remediation [39].
| Plant species |
Metal |
| Alyssum wulfenianum |
Ni |
| Azolla pinnata, lemna minor |
Cu,Cr |
| Brassica juncea |
Cu, Ni |
| Arabiodopsis halleri |
Cd |
| Pteris vittata |
Cu, Ni, Zn |
| Psychotria douarrei |
Ni |
| Pelargonium sp. |
Cd |
| Thlaspi caerulescens |
Zn, Cd i Ni |
| Amanita muscaria |
Hg |
| Arabis gemmifera |
Cd i Zn |
| Pistia stratiotes |
Ag, Cd, Cr, Cu, Hg, Ni, Pb i Zn |
| Piptathertan miliacetall |
Pb |
| Astragulus bisulcatus, Brassica Juncea |
Se |
| Sedum alfredii |
Cd |
| Helianthus annuus |
Pb |
| Helianthus indicus |
Pb |
| Sesbania drummondi |
Pb |
| Lemna gibba |
As |
| Pteris vittata |
As |
| Sedum alfredii |
Pb/Zn |
| Thlaspi caerulescens |
Zn, Pb, Zn i Cd |
| Chengiopanax sciadophylloides |
Mn |
| Tamarix smyrnensis |
Cd |
| Potentilla griffithii |
Cd/Zn |
| Brassica napus |
Cd |
| Arabidopsis thaliana |
Zn i Cd |
| Crotalaria juncea |
Ni i Cr |
| Cynodon dactylon |
Ni i Cr |
| Rorippa globosa |
Cd |
Table 3: Examples of suitable plant species for metal remediation (Sarma, 2011).
Chelation in Metal Intoxication
Chelation therapy is a medical procedure used primarily to reduce the toxic effects of metal ions. Its application covers a wide range of disorders, from acute metal poisoning overload to genetic material metal intoxication. The use of chelating agents is, however, compromised by a number of serious side effects, which mainly are due to disorders of homeostasis of essential metals and their transport and the formation of the depot complex in certain organs. For this reason, chelation therapy is limited to certain critical cases and its application should be be put into clinical context, ie. Assess the risk-benefit ratio for the patient prior to use.
Chelation therapy is an important concept and method for changing the concentration of metals in the body. Chelation as a word is derived from the Greek word chele which means "crab claws", which vividly represents a way of bonding by firm grasping. The term chelation was first used by Sir Gilbert T. Morgan and H. D. K. Drew in 1920 [40]. They suggested this term for groups that function as a means of connection and are linked to the central atom so that it can form a heterocycle. Chelating agents such as inorganic or organic compounds are capable of binding to the metal ion in the complex ring structures which are called chelates. Chelating agents have ligand-binding atoms that are associated with metal-covalent or coordinate covalent bonds. Depending on how the connection is formed, we say that the chelating agent is mono- , bi-, tri- or polydentate. Table 4 Selected chelating agents (highlighted in red are the atoms that bind to the metal).
| Chemical name |
Chemical structure |
Metal |
| D-penicillamine |
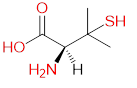 |
Cu, As, Zn |
| Ethylenediaminetetraacetic acid (EDTA) |
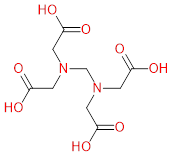 |
Pb, Cd, Zn |
2,3-disulfanylprop-1-ol
(BAL) |
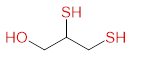 |
As, Au, Hg, Pb |
| 2,3-disulfanyl-1-propanesulfonic acid (DMPS) |
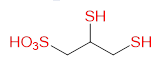 |
Hg, Pb, As |
Table 4: Selected chelating agents (highlighted in red are the atoms that bind to the metal).
Although the concept of chelation is based on simple co-ordination chemistry, chelator evolution and the search for the ideal one that specifically and fully removes the toxic metal from the tissue, with no side effects, is a process that includes access to drug design. Ideal universal chelator should be well soluble in water, able to form non-toxic complexes with a range of heavy metals, that can be easily removed from the body and it should pass through the cell membrane. There should be a possibility for the formulation of the compound in the oral, intravenous or intramuscular formulation depending on the type of intoxication. It is also necessary to show a greater affinity for the toxic metal than for the essential metals from the organism. For now there is no such unique chelating agent, although several preparations have been registered in America [41-44].
Mercury chelation
Mercury poisoning was earlier treated by chelators BAL and D-panicilaminom. Now, however, BAL usage is considered to be contraindicated in many cases of poisoning because it increases the level of the deposit of the inorganic and organic mercury in the brain [45,46]. DMSA (meso-2,3-dimercaptosuccinic acid) is an effective antidote in cases of systemic poisoning of Mercury(II) chloride or Methyl-mercury in experimental animals [47,48], while in these cases penicillamine was not sufficiently effective. The latter study showd that DMSA has significantly reduced levels of mercury in the brain of experimental animals after oral administration and improved the clinical signs of nerve toxicity of methyl-mercury [49]. Both agents were shown to reduce the mortality of animals against a lethal dose of Mercury (II) chloride [50].
In the study Bluma et al examined a group of 26 factory workers who ingested significant amounts of mercury vapors. They were hospitalized more than two weeks after mercury exposure. A 14-days DMSA treatment of one group and penicillamine in the other groups of patients was initiated. DMSA has increased mercury urinary excretion five times as compared to the untreated group, while penicillamine excretion increased it two times [9]. In Europe, the mercury intoxication is usually treated by DMPS (2,3-disulfanyl-1-propansulfonic acid). In comparison with D-penicillamine and N-acetyl-D, L-penicillamine, DMPS is much more efficient in removing mercury from the bloodstream [51].
Lead chelation
DMSA was registered in 1991 in the United States as an antidote for oral use in children with lead poisoning. Indication of the concentration of lead in the blood is >450 g L-1 [52]. Recommendations for treatment of lead intoxication with DMSA are based on numerous case studies and cohort studies in animal models. DMSA is more efficient than penicillamine in the reduction of lead levels in the tissue and urinary excretion in mice and rats. Parenteral therapy in combination with CaEDTA and BAL is no better than therapy with DMSA only. In the case of acute toxicity in rats, oral administration of DMSA injection is more effective than EDTA in reducing the lead level in the brain, blood, liver and kidneys. EDTA even slightly increases lead accumulation in these organs, which is caused by a reaction of inflammation and increased permeability of the blood-brain barrier [53,54].
Several small cohort studies of chelation therapy in human lead poisoning were published. Five workers at the foundry were treated with oral doses of DMSA for one week. This treatment reduced the blood lead levels by 50% [55]. In another study, nine workers received DMSA therapy during five days. Lead levels in the blood are reduced, as in the previous study by about 50% (35- 81%), and urinary excretion was significantly increased. Three weeks after initiation of therapy clinical signs of lead poisoning have disappeared or been greatly reduced [56].
In 2002, Gordon and colleagues published an interesting study of three cases in which they followed three workers who renovated a house using lead-based wall paint. All three showed symptoms of fatigue, arthralgias, malaise, abdominal cramps and nausea. After diagnosis, chelating therapy was administred and lead levels monitored [57]. Table 5 Lead poisoning of workers - a case study (Gordon, 2002).
| Day |
Worker 1 |
Worker 2 |
Worker 3 |
| |
[Pb] |
Therapy |
[Pb] |
Therapy |
[Pb] |
Therapy |
| 1 |
4.18 |
NaCaEDTA, 40 mg kg-1 i.v. every 12 h during 48 h, followed by DMSA 30 mg kg-1 p.o. 5 days, than 20 mg
kg-1 14 more days |
4.04 |
NaCaEDTA, 40 mg kg-1 i.v. every 12 h during 48 h, followed by DMSA 30 mg kg-1 for 14 days. After 14 days, patient has developped DMSA alergy |
4.09 |
19 days DMSA, without NaCaEDTA |
| 7 |
|
4.33 |
|
| 8 |
4.13 |
|
|
| 9 |
2.86 |
2.17 |
|
| 11 |
|
0.72 |
|
| 36 |
2.13 |
|
2.47 |
[Pb] – measured lead concentrations in plasma (mmol L-1);
DMSA - succimer, 2,3-dimercaptosuccinic acid
Table 5: Lead poisoning of workers - a case study (Gordon, 2002).
Cadmium chelation
The therapeutic effect of several chelating agents, including BAL and its soluble glycosides has unsuccessfully been tested in acute cadmium intoxication [58]. In the year 2005, deferasirox has become the first oral formulation approved by the FDA (Food and Drug Administration) for the treatment of iron intoxication, and next year it was approved in Europe for the same indication [59].
The aim of Fatemi and assistants’ study from 2011 was comparing the chelating potential of deferasirox and deferiprone (L1) in the cadmium poisoning cases in experimental animals. Testing was conducted using acute experimental rat model. The animals were divided into 4 groups. The first group was the control group and the second group received deferasirox, the third one deferiprone and the fourth group was given a combination of the two chelators. In each group there were five rats. The study results showed that in the control group the level of cadmium ranged 0.225 ± 0.021 mg kg-1 body weights of rats. Levels in the second, third and fourth groups were: 0.089 ± 0.015; 0.110 ± 0.016 and 0.067 ± 0.017 mg kg-1, respectively. The conclusion is that the combination of L1 with deferasirox can eliminate cadmium from the body and signs of toxicity. This combination is a promising treatment modality for cadmium poisoning, not only in rats, but also in humans [60].
Zinc chelation
The most important extracellular zinc chelator has so far been CaEDTA; however, it may prevent the influx of zinc in the cell and indirectly cause the death of neurons, though it does not cross the blood-brain barrier itself [61]. As a result, two new chelators have been developed and tested. These compounds are 1-hydroxypyridine-2-thione (pyrithione), with lower affinity for zinc and N, N, N’, N’-tetrakis-(2-pyridylmethyl) ethylenediaminepentaethylen (TPEN), having a high affinity for zinc. TPEN itself is toxic in high concentrations [62]. Both agents do not interfere with the permeability of the cell membrane.
Copper chelation
D-penicillamine, as well as Trien and DMSA injections were tested in the cases of mice copper salts intoxication and they have proven to be effective agents, and are used as antidotes [63]. These chelators reduce hemolysis caused by the presence of copper in the human red blood cells in vitro [64]. Studies in several species have shown an increased urinary excretion of copper after giving Trien or D-penicillamine. Oral administration of DMSA increased urine copper excretion in rats previously given intravenous copper salts injection [65].
Only a few cases of acute copper intoxication have been observed in the developed world. In these cases, D-penicillamine was orally administered with success [66]. Wilson's disease, a hereditary chronic copper hyperaccumulating and therefore poisoning has been the subject of the study conducted by Walsh and associates. They have shown that in this indication D-penicillamine can successfully be applied [67], however, its use is limited due to serious side effects [68], among which the most important is an immune response that leads to the development of systemic lupus erythematosus or kidney disease caused by deposition of immune complexes. When D-penicillamine intolerance is present, triethylenetetramine (Trien) is indicated [69]. Since Trien can cause side effects, a third chelating agent for copper toxicosis is introduced. This agent is tetrathiomolybdate [70,71] and it inhibits the copper absorption in the stomach [72].
Arsenic chelation
Current recommended treatment of arsenic poisoning is the use of DMPS or DMSA. They are more soluble in water than the BAL and may be administered orally [73]. All known chelating agents may have unwanted effects and should be used carefully. In cases of arsenic poisoning, chelating is recommended due to the fact that the benefit of therapy exceeds the risk of side effects. Even when there are unconfirmed suspicions of arsenic ingestion, chelation therapy is indicated for the prevention of acute renal failure [74]. A case study exsists where a 22-month old baby who ingested 0.65 g of sodium arsenate is monitored. On admission to hospital concentration of 5 mg/dm3 of arsenic in blood were recorded and after treatment it was lowered to 0.1 mg L-1 [75].
Nickel chelation
In the treatment of acute human nickel poisoning resulting from nickel carbonyl inhalation, sodium diethyldithiocarbamate (Dithiocarb) proved to be extremely good and specific antidote. Tetraethylthiuram (Antabuse) was less effective, while D-penicillamine and dimercaprol (BAL) have a limited therapeutic significance. In the treatment of eczema and dermatitis in humans caused by nickel, curing is achieved by a combination of diet with reduced concentration of nickel and the oral administration of disulfiram or Dithiocarb [76].
In experimental models, dithiocarb showed an inhibitory effect on the occurrence of rhabdomyolysis in rats. Rhabdomyolysis occurs as a result of nickel sulphide intramuscular implantation. Due to these results, it is assumed that the chelator may be used in the therapy of human tumors resulting from the implantation of nickel containing prosthesis. Kaaber and colleagues conducted a study back in 1979 in which they examined nickel potential for causing dermatitis and treatment options of the resulting dermatitis. In Table 6 are given nickel concentrations measured in the sera of (g L-1), and urinary excretion of nickel (mg/24 h) in three patients after treatment with chelating agents such TETD (tetraethylthiuramdisulfide) [77].
Patient
number |
Dermatitis duration |
Before treatment |
1 week after treatment |
2 weeks after treatment |
| |
|
serum |
urine |
serum |
urine |
serum |
urine |
| 1 |
9 |
0.2 |
1 |
4.5 |
34 |
2.4 |
26 |
| 2 |
4 |
0.4 |
1.6 |
3.8 |
54 |
1.4 |
43 |
| 3 |
4 |
0.1 |
1.1 |
4.7 |
31 |
2.3 |
16 |
Table 6: Patients’ clinical data.
Conclusion
In the end we can say that every day we come into contact with heavy metals, due to their omnipresence: heavy metals are present in drinking water, food, air, products for personal and domestic hygiene. It is important to know that they can not be avoided completely, but once settled in our body, can cause serious health problems and ailments. It is therefore not surprising that a large number of doctors, specialists, nutritionists and phytotherapists regularly study this field of science, constantly trying to contribute to the prevention and treatment of this serious health and environmental problems. Chelation therapy is effective for reducing toxic effects of metal ions. Having in mind that the use of chelating agents is limited by its adverse effects, it is necessary to choose the most suitable chelating agent as a medicament, and to avoid disruption of metal homeostasis.
Conflict of Interest
None declared.
Acknowledgements
These results are part of projects No. 45017 and 41018 and were realized through the partial financial support of the Republic of Serbia Ministry of Education and Science.
References
- Thornton I (1995) Metals in the global environment-facts and misconceptions, ICME, Ottawa.
- Vukmirović Z (1997) Teški metali u vazduhu. U: Kastori R. (urednik): Teški metali u životnoj sredini. Naučni institut za ratarstvo i povrtarstvo, Novi Sad.
- Duffus JH (2002) “Heavy metals” a meaningless term? Pure Appl Chem 74: 793-807.
- Phipps DA (1981) Chemistry and biochemistry of trace metals in biological systems. In: Lepp NW, editors. Effect of heavy metal pollution on plants: effects of trace metals on plant function, Vol. I London and New Jersey: Applied Science Publishers, Pp: 1-54.
- He ZL, Zdenko Yang XE, Stoffella PJ (2005) Trace elements in agro ecosystems and impacts on the environment. J Trace Elem Med Biol 19: 125-140.
- Küpper H, Kroneck PMH (2005) Heavy metal uptake by plants and cyanobacteria. Met Ions Biol Syst 44: 97-142.
- Greaf JW (1994) Heavy metal poisoning. In: Isselbacher KJ, Braunwald E, et al, editors. Harrison’s principles of internal medicine. New York: Mc Graw-Hill Inc, 13th Edn, Pp: 2461-2466.
- Nordberg GF, Fowler BA, Nordberg M, Friberg L (2007) Handbook on the toxicology of metals. Academic Press, New York.
- Bluhm RE, Bobbitt RG, Welch LW, Wood AJ, Bonfiglio JF, et al. (1992) Elemental mercury vapour toxicity, treatment, and prognosis after acute, intensive exposure in chloralkali plant workers. Part I: History, neuropsychological findings and chelator effects. Human Exp Toxicol 11: 201-210.
- Kabata-Pendias A, Pendias H (1984) Trace elements in soils and plants. Baton Rouge, USA: CRC Press, Pp: 1-403.
- Fergusson JE (1990) The heavy elements, chemistry, environmental impact and health effects. Pergamon Press, Oxford.
- Vapa M (1993) Teški metali i zdravlje ljudi i životinja. U: Teški metali i pesticidi u zemljištima Vojvodine. Poljoprivredni fakultet, Institut za ratarstvo i povrtarstvo, Novi Sad.
- Moore MR (1989) The percutaneous absorption of lead-203 in humans from cosmetic preparations containing lead acetate, as assessed by whole-body counting and other techniques. Food Cosmet Toxicol 18: 399-409.
- Rabinowitz MB, Wetherill GW, Kopple JD (1976) Kinetic analysis of lead metabolism in healthy humans. J Clin Invest 58: 260-270.
- Hanninen H, Mantere P, Hernberg S, Seppalainen A, Kock B (1979) Subjective symptoms in low-level exposure to lead. Neurotoxicology 1: 333-348.
- Schwarz Y (1974) Trace element metabolism in animals. University Park Press, Baltimore.
- Goyer RA, Clasen CD (1995) Metal toxicology, Academic Press, San Diego.
- Shah K, Dubey RS (1995) Effect of cadmium on RNA level as well as activity and molecular forms of ribonuclease in growing rice seedlings. Plant Physiol Biochem 33: 577-584.
- Satarug S, Baker JR, Urbenjapol S, Haswell-Elkins M, Reilly PE, et al. (2003) A global perspective on cadmium pollution and toxicity in non-occupationally exposed population. Toxicol Lett 137: 65-83.
- Seiler HG, Sigel H (1988) Handbook of toxicity of inorganic compounds. Marcel Dekker, New York.
- ATSDR, Agency for Toxic Substances and Disease Registry (1999) Toxicological profile for cadmium, Atlanta, GA.
- Finkelman RB (2005) Sources and health effects of metals and trace elements in our environment: an overview. In: Moore TA, Black A, Centeno JA, Harding JS, Trumm DA, editors. Metal Contaminants in New Zealand. Christchurch, New Zealand: University of Canterbury Press, Pp: 25-46.
- Alloway BJ (1995) Soil processes and the behaviour of metals. In: Alloway BJ, editor. Heavy Metals in Soils. Dordrecht, the Netherlands: Springer Science & Business Media,
Pp: 11-37.
- Kabata-Pendias A, Mukherjee AB (2007) Trace Elements from Soil to Human. Springer, New York.
- Kisić I (2012) Sanacija onečišćenog tla. Agronomski fakultet Sveučilišta u Zagrebu, Zagreb.
- Hughes MF (2002) Arsenic toxicity and potential mechanisms of action. Toxicol Lett 133: 1-16.
- Adriano DC (2001) Trace elements in terrestrial environments: biogeochemistry, bioavailability and risks of metals. 2nd Edn. Springer-Verlag, New York, Pp: 1-796.
- Rascio N, Navari-Izzo F (2011) Heavy metal hyper accumulating plants: how and why do they do it? and what makes them so interesting? Plant Sci180: 169-181.
- Verbruggen N, Hermans C, Schat H (2009) Molecular mechanisms of metal hyper accumulation in plants. New Phytol181: 759-776.
- Baker AJM, McGrath SP, Reeves RD, Smith JAC (2000) Metal hyper accumulator plants: a review of the ecology and physiology of a biochemical resource for phytoremediation of metal-polluted soils. In: Terry N, Banuelos G, Vangronsveld J, editors. Phytoremediation of contaminated soil and water. Boca Raton, FL, USA: Lewis Publishers, Pp: 85-107.
- McGrath SP, Sidoli CMD, Baker AJM, Reeves RD (1993) The potential for the use of metal-accumulating plants for the in situ decontamination of metal-polluted soils. In: Eijsackers HJP, Hamers T, editors. Integrated soil and sediment research: a basis for proper protection. Dordrecht, the Netherlands: Kluwer Academic Publishers, Pp: 673-677.
- Boyd RS (2011) The defense hypothesis of elemental hyper accumulation: status, challenges and new directions. Plant Soil 293: 153-176.
- Bhargava A, Carmona FF, Bhargava M, Srivastava S (2012) Approaches for enhanced phytoextraction of heavy metals, Review. J Environ Manage 105: 103-120.
- Meharg AA (2003) Variation in arsenic accumulation-hyper accumulation in ferns and their allies. New Phytol 157: 25-31.
- Anderson TR, Howes AW, Slater K, Dutton MF (1996) Studies on the nickel hyperaccumulator, Berkheya coddii. In: Jaffre T, Reeves RD, Becquer T, editors. The ecology of ultramafic and metalliferous areas. Proceedings of the second international Conference on Serpentine Ecology, Noumea, New Caledonia, July 31-Aug. 5, 1995. OSTROM, New Caledonia, Pp: 261-266.
- Robinson BH, Brooks RR, Clothier BE (1999) Soil amendments affecting nickel and cobalt uptake by Berkheya coddii - potential use for phytomining and phytoremediation. Ann Bot 84: 689-694.
- Kamachi H, Komori I, Tamura H, Sawa Y, Karahara I, et al. (2005) Lead tolerance and accumulation in the gametophytes of the fern Athyrium yokoscense. J Plant Res 118: 137-145.
- Santos J, Almeida SFP, Figueira E (2013) Cadmium chelation by frustulins: a novel metal tolerance mechanism in Nitzschia palea (Kützing) W. Smith. Ecotoxicology 22: 166-173.
- Sarma H (2011) Metal hyper accumulation in plants: a review focusing on phytoremediation technology. J Environ Sci Technol 4: 118-138.
- Morgan GT, Drew HDK (1920) Researches on residual affinity and co-ordination. Part II. Acetylacetones of selenium and tellurium. J Chem Soc 117: 1456-1465.
- Bamonti F, Fulgenzi A, Novembrino C, Ferrero ME (2011) Metal chelation therapy in rheumathoid arthritis: a case report. Successful management of rheumatoid arthritis by metal chelation therapy. Biometals 24: 1093-1108.
- Brent J (2013) Commentary on the abuse of metal chelation therapy in patients with autism spectrum disorders. J Med Toxicol 9: 370-372.
- Budimir A (2011) Metal ions, Alzheimer’s disease and chelation therapy. Acta Pharm 61: 1-14.
- Ernst E (1997) Chelation therapy for peripheral arterial occlusive disease: a systematic review. Circulation 96: 1031-1033.
- Berlin M, Ullberg S (1963) Increased uptake of mercury in mouse brain caused by 2,3-dimercaptopropanol. Nature 197: 84-85.
- Aaseth J (1973) The effect of mercaptodextran on distribution and toxicity of mercuryin mice. Acta Pharmacol Toxicol 32: 430-441.
- Magos L (1976) Effects of dimercaptosuccinic acid on excretion and distribution ofmercury in rats and mice treated with mercuric-chloride and methylmercurychloride. Br J Pharmacol 56: 479-484.
- Aaseth J, Friedheim EAH (1978) Treatment of methyl mercury-poisoning in mice with 2,3-dimercaptosuccinic acid and other complexing thiols. Acta Pharmacol Toxicol 42: 248-252.
- Aaseth J, Alexander J, Raknerud N (1982) Treatment of mercuric chloride poisoningwith dimercaptosuccinic acid and diuretics: preliminary studies. J Toxicol Clin Toxicol 19: 173-186.
- Nielsen JB, Andersen O (1991) Effect of 4 thiol-containing chelators on disposition oforally-administered mercuric-chloride. Human Exp Toxicol 10: 423-430.
- Ashton CE, Illa KK, Mant T (1992) 2,3-dimercaptopropane-1-sulphonate (DMPS) in the treatment of heavy metal poisoning, an effective and potentially lifesaving treatment. EAPCCCT, XV Congress, Istanbul, Turkey.
- AAPCD (1995) American Academy of Pediatrics Committee on Drugs. Treatment guidelines for lead exposure in children. Pediatrics 96: 155-160.
- Flora GJ, Seth PK, Prakash AO, Mathur R (1995) Therapeutic efficacy of combined meso-2,3-dimercaptosuccinic acid and calcium disodium edetate treatment during acute lead intoxication in rats. Human Exp Toxicol 14: 410-413.
- Abbott NJ (2000) Inflammatory mediators and modulation of blood–brain barrier permeability. Cell Mol Neurobiol 20: 131-147.
- Friedheim E, Graziano JH, Popovac D, Dragovic D, Kaul B (1978) Treatment of leadpoisoning by 2,3-dimercaptosuccinic acid. Lancet 2: 1234-1236.
- Fournier L, Thomas G, Garnier R, Buisine A, Houze P, et al. (1988) 2,3-Dimercaptosuccinic acid treatment of heavy metal poisoning in humans. Med Toxicol Adverse Drug Exp 3: 499-504.
- Gordon JN, Taylor A, Bennett PN (2002) Lead poisoning: case studies. J Clin Pharmacol 53: 451-458.
- Dalhamn T, Friberg L (1955) Dimercaprol (2, 3-dimercaptopropanol) in chronic cadmium poisoning. Acta Pharmacol Toxicol 11: 68-71.
- Yang LPH, Keam SJ, Keating GM (2007) Deferasirox: a review of its use in the management of transfusional chronic iron overload. Drugs 67: 2211-2230.
- Fatemi SJ, Saljooghi AS, Balooch FD, Iranmanesh M, Golbafan MR (2011) Chelation of cadmium by combining deferasirox and deferiprone in rats. Toxicol Ind Helath 27: 371–377.
- Chen CJ, Liao SL (2003) Neurotrophic and neurotoxic effects of zinc on neonatal cortical neurons. Neurochem Int 42: 471-479.
- Lu C, Xiao-Kun L, Ye S, Cherian MG (2005) Essentiality, toxicology and chelation therapy of zinc and copper. Curr Med Chem 12: 2753-2763.
- Jones MM, Weaver AD, Basinger MA (1981) Characteristics of chelate antidotes for acute Cu(II) intoxication. J Inorg Nucl Chem 43: 2175-2181.
- Aaseth J, Benov L, Ribarov S (1990) Mercaptodextran-a new copper chelator and scavenger of oxygen radicals. Zhongguo Yao Li Xue Bao 11: 363-367.
- Aaseth J (2012) Experimental copper chelation: clinical implications. Erudite J Drugs 1: 1-3.
- Jantsch W, Kulig K, Rumack B (1984-1985) Massive copper sulfate ingestion resulting inhepatotoxicity. J Toxicol Clin Toxicol 22: 585-588.
- Walshe JM (1956) Penicillamine a new oral therapy for Wilson’s disease. Am J Med 21: 487-495.
- Walshe JM (1968) Toxic reactions to penicillamine in patients with Wilson’s disease. Postgrad Med J, Pp: 6-8.
- Walshe JM (1982) Treatment of Wilsons-disease with trientine (triethylenetetramine) dihydrochloride. Lancet 1: 643-647.
- Ding X, Xie H, Kang YJ (2011) The significance of copper chelators in clinical andexperimental application. J Nutr Biochem 22: 301-310.
- Walshe J (1986) Tetrathiomolybdate (MoS4) as an anticopper agent in man. In: Schein-berg I, Walshe J, editors. Orphan diseases and orphan drugs. Manchester: manchester University Press, Pp: 76-85.
- Brewer GJ (1995) Practical recommendations and new therapies for Wilson’s disease. Drugs 50: 240-249.
- Mazumder DG, De BK, Santra A, Ghosh N, Das S, et al. (2001) Randomized placebo-controlled trial of 2,3-dimercapto-1-propanesulfonate (DMPS) in therapy of chronic arsenicosis due to drinking arsenic-contaminated water. Clin Toxicol 39: 665-674.
- Murunga E, Zawada E (2007) Environmental and occupational causes of toxic injury to the kidneys and urinary tract. In: Rom W, Markowitz S, editors. Environmental and occupational medicine, 4th Edn. Hagerstown, MD: Lippincott Williams & Wilkins.
- Cullen NM, Wolf LR, St Clair D (1995) Pediatric arsenic ingestion. Am J Emerg Med 13: 432-435.
- Sunderman FW (1981) Chelation therapy in nickel poisoning. Ann Clin Lab Sci 11: 1-8.
- Kaaber K, Menne T, Tjel JC, Veien N. Antabuse (1979) Antabuse treatment of nickel dermatitis. Chelation - a new principle in the treatment of nickel dermatitis. Contact dermatitis 5: 221-228.