Research Article - (2015) Volume 1, Issue 1
Jingyuan Xu1, Brook Carson2 and Sanghoon Kim1
1National Center for Agricultural Utilization Research, Agricultural Research Service, USDA, 1815 North University Street, Peoria, Illinois, USA
2Archer Daniels Midland Company, James R. R and All Research Center, Decatur, Illinois, USA
Received Date: November 20, 2015; Accepted Date: November 12, 2015; Published Date: December 21, 2015
Citation: Xu J, Carson B, Kim S. The pH Dependence on the Rheology of Wheat Protein Isolate Suspensions. Polym Sci. 2015, 1:1.
Linear and non-linear rheological properties of wheat protein isolate (WPI) suspensions were investigated as a function of concentration and pH. Linear dynamic viscoelastic properties for WPI were strongly dependent on concentration and pH. The higher the concentration, the stronger the viscoelasticity of the WPI would be. In the pH range of 4.0-7.0, higher pH resulted in stronger viscoelasticity. WPI suspensions exhibited viscoelastic fluid behavior at lower concentration and/or lower pH. However, at high concentration and high pH, WPI suspensions showed some transition from viscoelastic fluid into viscoelastic solid, and displayed viscoelastic solid behavior at low frequencies. Concentration and pH ranges for the transition were narrow indicating that the property change for the WPI was in evidence. The non-linear shear viscoelastic properties of WPI were also found to depend on concentration and pH. Viscosities of WPI displayed shear-thinning behavior, and fits by a power law constitutive model. Our results indicate that the WPI structure in suspension changes over a small concentration and pH range, which suggest that WPI could be important for adjusting and controlling dough viscoelastic behavior. The information of this work is useful in the development of more and new applications using wheat protein isolate.
Wheat proteins; Wheat protein isolate; Rheology; Viscoelastic properties
Wheat gluten has been widely used in many existing food applications and new product development. It has long been known that the baking quality of wheat flour depends both on quantity and quality of its gluten proteins [1]. Gluten is adopted as an additive due to its unique viscoelastic attributes and its nutrition advantages etc.., Wheat gluten, as a byproduct of the starch industry, is abundantly available. However, the usage of gluten is limited by its insolubility [2]. Allergenic response for a certain group of people. Wheat protein isolates (WPI), as modified gluten or wheat proteins, are good alternatives of gluten. They can have various properties such as glutenlike vitality, solubility, extensibility, etc. The behavior of WPI is more viscous, which is close to gliadin; while gluten is more viscoelastic [3]. Wheat proteins and wheat protein isolates can provide enormous possibilities in food applications because of their special functional and nutritional properties. Xu reported viscoelastic behaviors of vital wheat gluten as well as wheat gliadin and glutenin in suspensions [4,5]. In addition, we also reported micro- heterogeneity and micro-rheology of gliadin suspensions [6]. We found that gliadin exhibited viscous behavior at lower concentration but viscoelastic behavior at higher concentrations. However, literature reports are rarely found about properties of wheat protein isolates. WPI offers some “relaxed” vitality compared to traditional wheat gluten. It can be used as a protein additive to develop the proper texture in some high protein products such as baked, extruded, and fried food applications [3,7]. However, basic rheological properties of WPI were not conducted, which blocked more widely and further usage of it. In this work, we investigated basic linear and nonlinear rheological properties of WPI in suspension to avoid the problem of dough rheology. The rheological properties of dough can vary over a wide range. It is difficult to establish a baseline for dough rheological parameters. Since many factors, such as water content, mixing procedure, and mixing time, can largely affect dough rheology [4]. Therefore, the viscoelastic properties of suspensions give us more accurate insight of the rheology of WPI.
Materials
Wheat protein isolate PROLITE 200 was developed in Archer Daniels Midland Company (Decatur, IL). PROLITE 200 was produced from wheat gluten by fractionation, filtration and drying. It contained a minimum of 85% of protein (Nx6.25), <5.0% ash, <6.0% fat, and <6.0% moisture. Wheat gliadin sample was obtained from Midwest Grain Products, Inc. (Atchison, Kansas).
Methods
Sample preparation
WPI powders were suspended with three concentrations (25%, 30%, and 35%; wt.%) in buffers of 50 mM sodium phosphate and 3M urea at pH of 4.0, 5.5, and 7.0 at 25°C through mixing by agitation. Suspended samples were stored at 4°C and used within 2 days of preparation to prevent degradation. At least two samples were made at each concentration and pH for measurements.
SDS-PAGE
Sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) was carried out with a 12.5% polyacrylamide gel as described by Laemmli [8]. Samples were analyzed as reduced with dithiothreitol (DTT). Procedures for SDS-PAGE are: 1) making a gel and assembling the gel apparatus, 2) mixing protein samples with sample buffer containing SDS and heat the mixture at high temperature, 3) loading samples and running the electrophoresis, 4) fixing and staining the separated proteins.
Rheological measurements
Rheological properties for three concentrations (25%, 30%, and 35%; wt.%) and three pHs (pH 4.0, pH 5.5 and pH 7.0) of WPI suspensions were measured with a strain-controlled fluids Rheometric ARES rheometer (TA Instruments, New Castle, DE), using a 50-mm diameter cone and plate geometry. A humidity chamber was used to cover the apparatus to prevent sample evaporation. Temperature was controlled at 25 ± 0.1°C using a circulation system. Prior to dynamic rheological parameter measurements, a strain-sweep experiment was conducted to ensure that measurements were in the linear viscoelastic range. An applied shear strain in the linear range was adopted for the other viscoelastic property measurements for the same material; fresh samples were used for each experiment. Linear viscoelasticity means that measured parameters are independent of shear strains. Below 12% of strain, all measured materials in this study were in the linear range. Small-amplitude oscillatory shear experiments (10% of shear strain) were conducted over a frequency (δ) range of 0.1-500 rad/s, yielding the shear storage G’ and loss G” moduli. The storage modulus (G’) represents the non-dissipative component of mechanical properties, and is characteristic of elastic properties of the material. Elastic or “rubber-like” behavior is suggested if the G’ spectrum is independent of frequency and greater than the loss modulus over a certain range of frequency. The loss modulus (G”) characterizes the dissipative part of the mechanical properties and represents viscous flow of the material. The phase shift is defined by δ=tan- 1(G”/G’), which indicates whether a material is solid (δ=0°), liquid (δ=90°), or in between (0°<δ<90°). Stress relaxation experimentswere also performed in the linear viscoelastic range. These experiments measured the stress relaxation with the time after the material is subject to a step increase in strain. Non-linear rheological measurements were conducted as steady shear in the range of shear rate of 0.1s-1-500s-1. Each measurement was repeated at least twice with fresh samples. The relative errors were all within the range of ±12%.
The SDS PAGE of WPI and wheat gliadin is compared in Figure 1. It is to notice that WPI contained more high-molecular-weight proteins between 45,000 to 100,000 than gliadin did. Linear rheological properties of dynamic frequency sweep measurements were conducted on all 9 kinds of samples at 25°C. Both storage (G’) and loss (G”) moduli of the WPI suspensions were strongly dependent on concentration. Figure 2A displays the results of three concentrations of WPI suspensions at pH7.0, 25°C. For 25% (wt.%) WPI suspension, G” was higher than G’ over the measured frequency range; the moduli were strongly frequency dependent, and no plateau was observed (Figure 2A). These are typical viscoelastic fluid or liquid behaviors [9]. At 25%, the storage moduli (G’) of WPI were in the range of 0.3-72.5 Pa, the loss moduli (G”) were in the range of 0.7-214.2 Pa, and phase shifts were in the range of 58.9-71.3 degrees (Figures 2A and 2B). All these results suggested that WPI exhibit viscoelastic fluid or liquid properties on this condition. From 25% to 35%, values of the moduli increased with increasing concentration. At 30% (wt.%), G” were still higher than G’ for the measured frequencies, but increased compared to 25% WPI, and the phase shift (δ) significantly dropped (Figure 2B). G’ and G” for 30% WPI were in the range 1.2-157.5 Pa and 1.5-425.9 Pa, respectively. The phase shifts decreased to 50.3 -69.7 degrees (Figure 2B). At 30%, even though WPI suspensions were somewhat viscous, the molecular chain-chain interactions must increase. At higher concentration of 35% (wt.%), molecule-molecule interactions of WPI increased more. Storage moduli (G’) were higher than loss moduli (G”) at lower frequencies. In addition, there was a short plateau of moduli at lower frequencies (Figure 2A). G’ and G” ranged from 15.5-594.6 Pa and 8.3-1103.3 Pa respectively, and their phase shifts were the smallest among the three concentrations of WPI suspensions, 28.2-61.7 degrees (Figure 2). The phase shifts for three concentrations of WPI suspensions exhibited clear difference from 10-1 to 102 rad/s. Higher frequencies made differences smaller (Figure 2B). At 25%, phase shifts were the highest and not very frequency dependent among three concentrations of WPI, which indicated that the material was pretty liquid-like. At 30% and 35%, phase shifts were frequency dependent. At lower frequencies, phase shifts were smaller. Because chain-chain interactions became stronger at higher concentrations (30% and 35%), the material would resist slow external forces (low frequencies) by re-arrange their chain-chain network. While at high frequencies, the network had no time to re-arrange their chain-chain formation so the material was not strong enough to resist quick external forces (high frequencies). The results indicated that at 30% or more, WPI suspensions deviated further and further from viscoelastic fluid behavior, and exhibited viscoelastic solid properties at low frequencies. Stress relaxation measurements, G(t), also showed clear differences among the three concentrations of WPI suspensions (Figure 3). 25% WPI suspension was fully relaxed within one second after it was subjected to a step increase of strain. However, 35% WPI suspension had not been completely relaxed for 100 seconds, and exhibited a plateau modulus. The behaviors for 30% WPI suspension were in between (Figure 3). These results implied that the structure of WPI altered when the concentration changed. At higher concentrations, the molecule-molecule or chain-chain interactions became significantly stronger than those at lower concentrations. At pH5.5 and pH4.0, all measured WPI suspensions exhibited the same trend of concentration dependence as at pH7.0. Both storage moduli (G’) and loss moduli (G”) increased with the concentration increased (data not shown). Linear viscoelastic properties for WPI also depended on the pH of the suspensions (Figure 4). From above, we already know that at pH7.0, 25°C and low frequencies, 35% WPI suspension exhibited somewhat viscoelastic solid behavior (G’ were higher than G”) and there was a short plateau of moduli (Figures 2A and 4A). But at pH4.0, 35% WPI suspension showed viscoelastic fluid properties since G” was greater than G’ over the measured frequency range. G’ and G” were in the range of 3.3-357.9 Pa and 3.5-830.9 Pa, respectively for 35% suspension at pH4.0 compared to the range of 15.5-594.6 Pa and 8.3-1103.3 Pa, respectively for 35% suspension at pH7.0 (Figure 4A). The phase shifts varied from 46.4 -66.7 degrees for 35% suspension at pH4.0 to the range of 28.2 -61.7 degrees for 35% suspension at pH7.0 (Figure 4C). The behaviors for 35% WPI suspension at pH5.5 were in between (Figures 4B and 4C). G’ and G” were in the range 6.4 -482.9 Pa and 4.8-920.5 Pa, respectively. It exhibited a little viscoelastic solid behavior, G’were slightly higher than G” at low frequencies, and there was a shorter plateau of moduli than that at pH7.0. Figure 4C displayed clear change of the phase shift (δ) for the 35% WPI suspension at different pHs. In the range of pH4.0-pH 7.0, higher the pH, the lower the phase shift and the more solid-like the material (Figure 4B). Stress relaxation measurements further supported the above conclusion (Figure 5). At pH4.0, the material relaxed the fastest; while at pH7.0, the material relaxed the slowest. At pH5.5, the material relaxed in between (Figure 5). These results suggested that the chain-chain interactions of WPI suspensions should be stronger at higher pH level between pH4.0 to pH7.0. The 25% and 30% WPI suspensions showed the same trend of pH dependence in the range of pH4.0-pH7.0: the higher the pH, the higher values of moduli and the stronger viscoelasticity of the material (data not shown).The pH dependence properties above are coincident with the results of the dough behavior and baking quality of WPI obtained by Carson [7]. Carson reported that WPI dough made at different pHs exhibited different viscoelastic properties: the lower the pHs, the weaker the viscoelasticity’s of the dough. Apparently, altering pH level could change the molecular structure and/or chain-chain interactions of the WPI. Therefore, the viscoelasticity of WPI can be controlled and adjusted by altering pH levels, which should be important and useful in food applications using WPI. To better understand the chewing and processing behavior, the non-linear steady shear viscoelastic properties of WPI suspensions were studied in a range of 0.1 s-1 to 500 s-1. Most food processing and chewing behaviors occur in the shear rate range of 1 s-1 to 100 s-1 stated by Holdsworth [10]. The non-linear shear properties were dependent on concentration (Figure 6) and pH (Figure 7). WPI suspensions exhibited shear-thinning behavior over the entire measured shear rates at all measured concentrations and pHs. Shear viscosities were higher at higher WPI concentrations, as expected (Figure 6). The highest viscosities for 25% WPI suspensions were at pH 7.0 and the lowest at pH 4.0. Viscosities were in between at pH5. 5, but closer to those at pH 4.0 (Figure 7). The shear-thinning rheological behaviors can be characterized by a power law constitutive equation [11]. The power law equation can be written as δ= K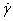 n-1 (1). Where δ is the shear viscosity, K is the front factor,
n-1 (1). Where δ is the shear viscosity, K is the front factor, 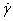 is the shear rate, and n is the power law exponent. Equation (1) gave very well fit to shear-thinning viscosity data of WPI suspensions (Figures 6 and 7). The resulting fit parameters are summarized in Tables 1 and 2. The higher concentrations or the higher pH levels, the lower power law exponents (Tables 1 and 2). The exponent for 25% WPI suspension at pH4.0 was 0.90, which was a non-Newtonian fluid but close to a Newtonian fluid (n=1 for a Newtonian fluid). However, with increasing pH or concentration, power law exponent decreased, indicating more non-Newtonian like behaviors. All other measured concentrations of WPI suspensions exhibited the same trend of pH dependence in the range of pH 4.0-pH 7.0: the higher the pH, the higher the viscosity of the material (data for the other concentrations not shown). Understanding the mechanical/rheological properties of wheat proteins is very important for elucidating the mechanism of baking, and to guide future research on wheat proteins for food and non-food applications. Dough rheology depends on many factors, such as water content, mixing procedure, mixing time, etc. This may lead to wheat protein doughs of widely varying viscoelastic properties, which complicates the investigation into the molecular factors affecting rheological properties of wheat proteins. To better understand basic rheology of wheat protein isolate without these complications of dough systems, we studied the basic viscoelastic properties in suspensions. WPI suspensions used in this work have no hydrogen bond involved due to the urea in the buffer. So the protein chain-chain interactions and disulfide bond interaction/cross-linking mainly contribute to the viscoelastic properties [4]. We found that both linear and nonlinear viscoelastic properties of WPI suspensions were dependent on concentrations and pHs. These concentration and pH dependence behavior should be related with structure alteration. This structure change may be due to increased chain-chain and molecular interactions, entanglements, or cross-linking etc. It is difficult to directly observe these structure differences. The molecular interpretation of rheological properties is not as well understood as other modern biochemical and molecular biology techniques. The curve shapes of moduli do not show the evidence of rubber-like behavior of cross-linking. In addition, our relaxation experiments exhibit different extent of relaxation after WPI is subject to a step increase in strain, which also suggests that there is no cross-linking. If a material has a network that is chemically cross-linked, there should not be any relaxation at all after the material is subject to a step increase in strain. So, there must be more physical chain-chain interactions than chemical interactions in the network. A more reasonable explanation for the change in structure is that more entanglements occur at higher concentrations and pHs for WPI. At different pH levels, protein molecules’ hydrophilic and hydrophobic status should be different so that the network and molecules chain-chain interactions behaved differently. This might be the reason why WPI properties are pH dependent. The concentration dependence rheological properties for WPI suspensions are similar to those for wheat gliadin suspensions [5]. However, WPI has slightly stronger viscoelasticity than gliadin because WPI contains some more proteins with higher molecular weight compare to gliadin (Figure 1). The pH dependence behavior of WPI is unique and no report regarding this behavior was found for wheat gliadin, glutenin and gluten. The current work combined with further biochemical/ biophysical structure work could help us to gain more clear insight into wheat protein isolate behavior. As an additive in many food applications, WPI can be used to manipulate, control and adjust food baking quality and behavior by altering concentration and pH according the information obtained in this work.
is the shear rate, and n is the power law exponent. Equation (1) gave very well fit to shear-thinning viscosity data of WPI suspensions (Figures 6 and 7). The resulting fit parameters are summarized in Tables 1 and 2. The higher concentrations or the higher pH levels, the lower power law exponents (Tables 1 and 2). The exponent for 25% WPI suspension at pH4.0 was 0.90, which was a non-Newtonian fluid but close to a Newtonian fluid (n=1 for a Newtonian fluid). However, with increasing pH or concentration, power law exponent decreased, indicating more non-Newtonian like behaviors. All other measured concentrations of WPI suspensions exhibited the same trend of pH dependence in the range of pH 4.0-pH 7.0: the higher the pH, the higher the viscosity of the material (data for the other concentrations not shown). Understanding the mechanical/rheological properties of wheat proteins is very important for elucidating the mechanism of baking, and to guide future research on wheat proteins for food and non-food applications. Dough rheology depends on many factors, such as water content, mixing procedure, mixing time, etc. This may lead to wheat protein doughs of widely varying viscoelastic properties, which complicates the investigation into the molecular factors affecting rheological properties of wheat proteins. To better understand basic rheology of wheat protein isolate without these complications of dough systems, we studied the basic viscoelastic properties in suspensions. WPI suspensions used in this work have no hydrogen bond involved due to the urea in the buffer. So the protein chain-chain interactions and disulfide bond interaction/cross-linking mainly contribute to the viscoelastic properties [4]. We found that both linear and nonlinear viscoelastic properties of WPI suspensions were dependent on concentrations and pHs. These concentration and pH dependence behavior should be related with structure alteration. This structure change may be due to increased chain-chain and molecular interactions, entanglements, or cross-linking etc. It is difficult to directly observe these structure differences. The molecular interpretation of rheological properties is not as well understood as other modern biochemical and molecular biology techniques. The curve shapes of moduli do not show the evidence of rubber-like behavior of cross-linking. In addition, our relaxation experiments exhibit different extent of relaxation after WPI is subject to a step increase in strain, which also suggests that there is no cross-linking. If a material has a network that is chemically cross-linked, there should not be any relaxation at all after the material is subject to a step increase in strain. So, there must be more physical chain-chain interactions than chemical interactions in the network. A more reasonable explanation for the change in structure is that more entanglements occur at higher concentrations and pHs for WPI. At different pH levels, protein molecules’ hydrophilic and hydrophobic status should be different so that the network and molecules chain-chain interactions behaved differently. This might be the reason why WPI properties are pH dependent. The concentration dependence rheological properties for WPI suspensions are similar to those for wheat gliadin suspensions [5]. However, WPI has slightly stronger viscoelasticity than gliadin because WPI contains some more proteins with higher molecular weight compare to gliadin (Figure 1). The pH dependence behavior of WPI is unique and no report regarding this behavior was found for wheat gliadin, glutenin and gluten. The current work combined with further biochemical/ biophysical structure work could help us to gain more clear insight into wheat protein isolate behavior. As an additive in many food applications, WPI can be used to manipulate, control and adjust food baking quality and behavior by altering concentration and pH according the information obtained in this work.
Table 1: Power law model fitted parameters for different concentrations of WPI suspensions at 25°C, pH7.0.
| Concentration of WPI | K (Pa-sn) | n | R2 |
| 25% (wt.%) | 1.08 | 0.80 | 0.99 |
| 30% (wt.%) | 7.60 | 0.70 | 0.98 |
| 35% (wt.%) | 37.5 | 0.50 | 0.85 |
Table 2:Power law model fitted parameters for 25% (wt.%) WPI suspensions at 25°C, pH4.0, pH5.5 and pH7.0
| pH | K (Pa-sn) | n | R2 |
| 4.0 | 0.46 | 0.90 | 0.95 |
| 5.5 | 0.58 | 0.85 | 0.96 |
| 7.0 | 1.08 | 0.80 | 0.99 |
The technical assistance of Sheila Maroney is acknowledged with gratitude. This work was financially supported by US Department of Agriculture, Agricultural Research Service.