Research Article - (2021) Volume 7, Issue 11
Received: 10-Dec-2021 Published: 31-Dec-2021
Objective: The oral and esophageal squamous cell carcinoma are very common in India and their highest incidence has been reported in north-eastern states. Of the several factors that are implicated for these cancers, the habit of chewing raw Areca Nut (AN) with or without tobacco appears to be highly relevant. These cancers have a poor prognosis when the diagnosis is delayed, but curative treatment is possible if the diagnosis is early. The present objective is to validate the screening of Securin upregulation, the frequency of Precocious Anaphase (PAS) and Sister Chromatid Exchanges (SCEs) in non-target cells like human Peripheral Blood Lymphocytes (PBLs) as early indicative parameters of AN with and without tobacco users.
Design: Peripheral blood was collected from 220 donors of which 124 were noncancerous and 96 were cancerous. All the cytogenetical end-points were evaluated from lymphocytes culture whereas the expression of Securin gene was assessed by immunohistochemistry and/or by qRT-PCR.
Results: A significant increase in the frequency of PAS, aneuploid cells and SCEs was observed in all AN/tobacco users compared to non-users. The frequency of aneuploidy cells was more in cancerous people than non-cancerous ones. The level of Securin was increased significantly in all AN/tobacco users than non-users. Such upregulation of Securin was also observed in the tumour tissues.
Conclusion: Upregulation of Securin and elevated frequency of PAS and SCEs in the PBL of both cancerous and non-cancerous people with AN/tobacco consumption habit indicate that these end-points can give relevant information on the genotoxic effect and could become potential intermediate biomarkers for risk assessment.
Sister chromatid exchanges; Precocious anaphase; Securin gene; Esophageal squamous cell carcinoma; Oral squamous cell carcinoma
Both Oral and Esophageal Squamous Cell Carcinoma (OSCC and ESCC) are considered to be a major health problem in the Indian subcontinent and both rank among the top four types of cancer in the country [1]. Many risk factors have been implicated for the causation of these cancers; however, tobacco and Areca Nut (AN) consumption in various forms, alcohol, hot beverages and, poor nutrition remain the predominant predisposing factors. Epidemiological studies have shown that around 40% of the population in India, have used Betel Quid (BQ) in which AN is one of the constituents. The AN is consumed either alone or with a slaked lime wrapped in a betel-leaf known as BQ. OSCC is the most common malignancy in India and strong indications for a causal association between AN or quid chewing habits and oral mucosal diseases such as leukoplakia, oral submucous fibrosis and, oral cancer are established [2]. The north-east Indian variety of AN is raw and unprocessed and consists of higher alkaloids, polyphenol and, tannins compared to the dried one and is locally known as ‘kwai’ [3]. Here people swallow the whole betel-quid after chewing and such a habit has been associated with the development of not only oral but also esophageal and gastric cancers [4,5]. It is known that both tobacco and betel quid may act synergistically as a carcinogen [6]. The IARC review suggested that AN is carcinogenic in humans and that it is linked to cancers not only of the oral cavity but also of the pharynx, esophagus, liver, biliary tracts and the uterus [7].
In India and particularly in rural areas, these cancers are diagnosed at later stages due to the lack of people’s awareness and inadequate health care services. The earlier the diagnosis of these cancers and their treatment, the better the outcome and disease-free survival. Therefore, in order to diagnose early, several studies were performed by selecting certain end-points relevant to carcinogenic processes that could serve as biomarkers [1,8]. In this context, we had performed a study with chewers of raw AN in North-eastern India and observed that the frequency of chromosomal aberrations, Sister Chromatid Exchanges (SCEs) and the expression of p53 were increased significantly whereas the level of endogenous glutathione was decreased in the blood lymphocytes of heavy chewers than non-chewers [4]. From this study, it was stated that these parameters can be used as potential screening markers for the identification of genotoxic effects during earlier days of raw AN exposure. Subsequently, we have extended our study in both mouse model as well as in human samples and the results indicate that raw AN and lime together induce oral, esophagous and stomach cancers and highlighted the occurrence of precocious anaphase (premature separation of sister-chromatids) and higher expression of p53 and Securin as potential screening markers for identification of mitotic checkpoint defects during early days of raw AN exposure [9,10]. On the basis of these studies, only three relevant parameters such as, Securin expression and induction of Premature Anaphase Separation (PAS) and SCEs are used in this study which can be evaluated promptly within a lesser period in the Peripheral Blood Lymphocytes (PBL).
SCE is a well-established and sensitive parameter for detecting DNA damage in the form of double-strand breaks induced by genotoxic agents [11,12]. PAS, also known as premature sister chromatid separation, is a condition when anaphase separation begins before the alignment of all the chromosomes in the metaphase plate [13]. In fact, partial loss of the spindle assembly checkpoint function can induce PAS and Chromosomal Instability (CIN) in mammalian cell [14]. Moreover, the frequency of SCE is also widely used indicator of CIN in response to genotoxic and environmental agents [15]. Mammalian securin is also known as Pituitary Tumor Transforming Gene 1 (PTTG1), is an oncogene and has been implicated in the development and progression of several malignancies [16,17]. Upregulation of securin and subsequent dysregulation of chromosome segregation leading to CIN has been observed in several human cancers [18,19]. It has already been proposed that the Securin level may be considered a molecular marker that can be a potential therapeutic target for many cancers [20]. Therefore, analysis of expression status of Securin and induction of SCEs and PAS in PBL can give relevant information on the genotoxic effect of AN with or without tobacco and their role in acquiring CIN in the early stages of AN-induced carcinogenesis.
Selection of study participants
The samples were collected from Nazareth hospital and North Eastern Indira Gandhi Regional Institute of Health & Medical Sciences, Shillong, India. Peripheral blood was collected from 220 donors in heparinized vials, under aseptic condition. The demography of these subjects is mentioned in the Table 1. Of 220 donors, 124 were from the noncancerous individuals and 96 were cancerous. The noncancerous samples were further classified as only AN-chewers (n=44), AN chewer with tobacco users (n=36) and without any habit (n=44). A total of 96 cancerous samples, 56 samples were from oral cancer patients and 40 samples from esophageal cancer patients. All these AN-chewers are considered heavy chewers since they have been consuming around 58-72 g of nut with an average 25 chews per day. The age ranged from 38 to 72 years (mean ± SD; 60.04 ± 8.62) for cancer samples whereas the age varied from 36 to 71 years (mean ± SD; 51.1 ± 11.32) for the noncancerous samples and in case of people with no habit, the age varied from 29 to 68 years (mean ± SD; 48.18 ± 13.46). The non-cancerous control samples were collected from people either accompanying the cancer patients or attending pathology department in the above-mentioned hospitals.
Lymphocyte culture procedure and preparations of metaphases
Heparinised peripheral blood (1.5 ml) was added to 8.0 ml Rosewell Park Memorial Institute 1640 medium (Biological Industries Ltd, Israel) supplemented with 10% heat inactivated foetal calf serum (Biological Industries Ltd, Israel). Lymphocytes were stimulated with Phytohae-magglutinine (PHA). Additional 5-bromodeoxyuridine (5 μg/ml) was added to each culture at the time of initiation. All cultures were incubated at 37°C and fixed at 72 h and 88 h for the analysis of PAS, aneuploidy and SCEs frequencies. Colcemid was added at a concentration of 0.02 μg/ml during the last 3 h in all the cultures. Conventional cell harvesting procedure follows: the cells were treated with 0.075 M KCl pre warmed to 37°C for 18 min and fixed in chilled acetic acid and methanol (1:3). The slides were prepared by air drying method from 88 h fixed cultures and were stained in 5% Giemsa for 4-5 min, air-dried and mounted on synthetic medium for scoring the frequency of PAS and aneuploidy. For scoring of SCEs, slides were prepared from 72 h fixed cultures and differential staining procedure was followed.
Differential staining
This was carried out following the method of Goto, et al. [21]. Slides were incubated for 10 min with Hoechst 33258 (50 μg/ mL) in the dark at room temperature, rinsed in distilled water, incubated in 2XSSC (NaCl-Na-Citrate, pH 6.8) under sunlight for 30-/40 min depending on the intensity of sunlight. After rinsing in distilled water, slides were stained in 2% Giemsa for 4 min, airdried and mounted on synthetic medium.
Immunohistochemistry analysis
Ten oral and nine oesophageal cancer patient samples were used for this study. Tumour and adjacent normal tissues were dehydrated, paraffin embedded and sectioned with a microtome (Leica). Slides were deparaffinized, rehydrated and treated with 3% H2O2 in PBS. Antigen retrieval was done with 0.01% sodiumcitrate buffer followed by blocking in PBST containing 0.1% BSA and 10% FBS. After blocking for endogenous peroxidase activity, the sections were incubated with anti-Securin (DCS-280; ab3305; Abcam, USA) primary antibody for Immunohisto-Chemistry (IHC) analysis. IHC analysis was performed with a Strept-Avidin Biotin Kit (Dako). Slides were washed and incubated with appropriate biotinylated secondary antibody for 1 h at room temperature. Following washing, slides were treated with streptavidin- HRP (1:1000) and subsequently washed (PBS containing 0.1% Tween-20) and color was developed with DAB+H2O2. Slides were counterstained with haematoxylin, washed and mounted in DPX (Sigma-Aldrich, USA). The staining intensity of the images was evaluated by two independent investigators.
Quantitative real-time PCR
qRT-PCR analysis was carried out in blood lymphocytes of six non-chewers, 10 AN-chewers and 10 AN-chewers with tobacco. Human lymphocytes were separated out from heparinised whole blood on a Ficoll-hypaque density gradient by the method of Boyum [22]. Freshly collected lymphocytes were washed into ice-cold PBS. Total RNA was isolated with Trizol from tumor as well as normal tissue samples collected from each patient and then purified using the RNeasy Mini Kit (Qiagen) according to the manufacturer’s protocol.
Synthesis of cDNA was performed with 1 μg of total RNA from each sample using Quantiscript Reverse Transcriptase, Quantiscript RTbuffer and RT Primenr-mix of QuantiTect Reverse Transcription kit (Qiagen GmbH, Hilden, Germany) according to the manufacturer’s protocol. Quantitative real-time PCR was performed using in 96-well optical reaction plates (Applied Biosystems, Darmstadt, Germany) using a StepOne Plus amplification and detection system (Applied Biosystems). The real-time RT-qPCR reactions were prepared using SYBR® Select Master Mix (Life Technologies), and the following conditions were used: 95°C for 5 min, 40 cycles of 95°C for 30 s, 60°C for 30 s and 72°C for 30 s. The primers of target genes used for this analysis were Securin, and GAPDH was used as the reference gene. The primer sequences are listed below. The gene copy numbers of Securin were calculated by using a standard curve that was constructed using the OE33 cell line. The 2−ΔΔCT method was used as a relative quantification strategy for qPCR data analysis.
PTTG1 F 5′‐ACCCGTGTGGTTGCTAAGG‐3′ Product size: 90 bp
PTTG1 R 5′‐ACGTGGTGTTGAAACTTGAGAT‐3′
GAPDH F 5’–ACAGTCCATGCCATCACTGCC–3’ Product size: 265bp
GAPDH R 5’–GCCTGCTTCACCACCTTCTTG–3’
Scoring and statistical analysis
For studying the frequency of SCEs, PAS and aneuploidy, the slides were coded in random. Figure 1A shows a second-cycle metaphase plate with SCEs and Figure 1B shows a Premature Anaphase Separation (PAS). At least 100 well-spread metaphase plates were selected for scoring the frequency of PAS and aneuploidy (except a few cases where the number is below 100 but above 90). We performed chromosome counts on metaphase spread. Values are expressed as mean ± SEMs.
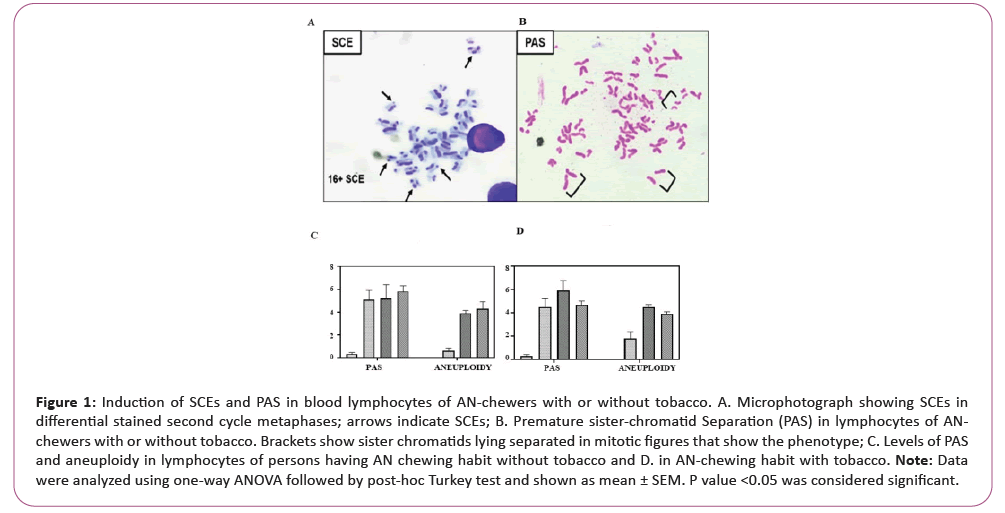
Figure 1: Induction of SCEs and PAS in blood lymphocytes of AN-chewers with or without tobacco. A. Microphotograph showing SCEs in
differential stained second cycle metaphases; arrows indicate SCEs; B. Premature sister-chromatid Separation (PAS) in lymphocytes of ANchewers
with or without tobacco. Brackets show sister chromatids lying separated in mitotic figures that show the phenotype; C. Levels of PAS
and aneuploidy in lymphocytes of persons having AN chewing habit without tobacco and D. in AN-chewing habit with tobacco.
Note: Data
were analyzed using one-way ANOVA followed by post-hoc Turkey test and shown as mean ± SEM. P value <0.05 was considered significant.
Metaphase (M) cells with differentially stained sister chromatids from each sample were studied for evidence of SCEs. The data of observed SCE-increase per metaphase were obtained. A miniumum of 25 metaphase cells with differentially stained sister chromatids from each sample were studied for evidence of SCEs.
The scoring of immunohistochemical stains in each specimen was determined using a histological (H) score [23]. Briefly, in the regions of the histologic sections, the chromogenic immunolabelling was systematically categorised into four groups: 0 (no labelling), 1+ (weak labelling), 2+ (moderate labelling) and 3+ (strong staining; observable with X10 objective). Semi-quantitative staining analysis was done by H-score by counting 500 cells from eight different fields in the slides considering low, moderate and higher intensity of protein expression. The percentage of positive cells with a given intensity for each sample was determined independently by a pathologist and a trained reader. A single manual H-score based on a scale of 0-300 was generated for each labelled section by taking the sum of the percentage of cells labelling 1+, double the percentage of cells labelling 2+ and triple the percentage of cells labelling 3+(H-Score=((%3+) × 3)+((%2+) × 2)+(%1+)). Student’s t-test was performed for comparing the Securin expression in normal and cancerous samples. Statistical significance was considered when P value is <0.05. The statistical significance in the frequency of SCE and PAS and expression of Securin from the blood lymphocytes was determined by one-way ANOVA. The Turkey test was used for post hoc analysis. Statistical analysis was performed with GraphPad Prism software 5.1. The values were considered statistically significant if the P value was 0.05 or less.
The study population consisted of 220 participants, 124 were non-cancerous and 96 were cancerous. Within non-cancerous, 44 individuals had no habit and 80 individuals had the habit of chewing raw AN with and without tobacco as shown in Table 1. In total 154 samples were used for the analysis of the frequency of SCEs, PAS and aneuploidy.
| Characteristics | Total number | Gender | Age (yr) | Age (yr) | |
|---|---|---|---|---|---|
| Male | Female | Range | Mean ± SD | ||
| Non-chewers (non-cancerous) | 44 | 24 | 20 | 29-68 | 48.2 ± 13.5 |
| AN-chewers (non-cancerous) | 44 | 16 | 28 | 36-71 | 51.1 ± 11.3 |
| AN-chewers (with OSCC) | 16 | 4 | 12 | 38-70 | 58.1 ± 13.9 |
| AN-chewers (with ESCC) | 16 | 12 | 4 | 47-64 | 57.8 ± 07.6 |
| AN-chewers with tobacco (non-cancerous) | 36 | 16 | 20 | 36-71 | 50.6 ± 09.6 |
| AN-chewers with tobacco (with OSCC) | 40 | 28 | 12 | 50-70 | 60.4 ± 07.8 |
| AN-chewers with tobacco (with ESCC) | 24 | 16 | 8 | 51-72 | 62.3 ± 08.2 |
| Total | 220 | 116 | 104 | ||
Table 1: Demography of non-cancerous and cancerous group.
Studies on metaphase spreads
To determine whether continuous exposure to RAN+lime with and without tobacco has any effect on chromosomes, we studied on metaphase spreads in human PBL. It was possible to distinguish unequivocally the number of divisions in the presence of BrdU and for scoring the frequency of SCEs, the second replication cycle cells were selected. The frequency of SCEs in AN-chewers with or without tobacco was significantly more than non-chewers. It is also noted that the frequency of SCEs in cancerous samples was almost at the same level of non-cancerous AN-chewers with or without tobacco. In fact, the number of cells having 0 to 5 SCEs are significantly higher in non-chewers than AN chewers irrespective of cancer status. No association was noted between the frequency of SCEs and the age of the individual. As shown in Table 2.
| Subjects (no. of samples) | SCEs/M ± SEM | Percentage of cells having SCEs | |
|---|---|---|---|
| 0-5 | >6 | ||
| Non chewers (30) | 4.8 ± 0.3 | 62.7 | 37.3 |
| AN chewers (30) | 8.4 ± 0.5* | 34.1 | 65.9 |
| AN chewers (OSCC) (12) | 8.1 ± 0.4* | 19.3 | 80.7 |
| AN chewers (ESCC) (12) | 8.3 ± 0.6* | 20.2 | 79.8 |
| AN chewers+tobacco (30) | 7.9 ± 0.6* | 39.8 | 60.2 |
| AN chewers+tobacco (OSCC) (20) | 9.2 ± 0.3* | 20.8 | 79.2 |
| AN chewers+tobacco (ESCC) (20) | 8.8 ± 0.5* | 22.2 | 77.8 |
Note: *Data were analyzed using one-way ANOVA followed by post-hoc Turkey test and shown as mean ± SEM. P value <0.05 was considered significant.
Table 2: Frequency and distribution of SCEs in peripheral blood lymphocytes in areca nut chewers with and without tobacco in non-cancerous and cancerous individuals.
We observed 4.8-5.8% mitotic figures with prematurely separated sister chromatids (precocious anaphase) in 88 h fixed lymphocytes of AN-chewers with or without cancer. Such mitotic figures were not seen in the blood lymphocytes collected from non-chewers. Chromosome counts revealed a stable karyotype (2n=46) in non-chewers samples without showing any aneuploid cells. The frequency of aneuploid cells, ranging from 1.5 to 9.5% was observed in several individuals having RAN-chewing habit with and without tobacco. However, the frequency of aneuploidy cells was higher in cancerous samples than noncancerous samples. Inter-individual variations in the frequencies of PAS and SCEs were noted, however no association between the frequency of both the parameters and age of the individual was observed. As shown in Figures 1C and 1D.
Analysis of over-expression of Securin in blood lymphocytes in AN-chewers
qRT-PCR analysis is illustrated as shown in Figure 2A from blood lymphocytes of six non-chewers, 10 only AN-chewers and 10 AN-chewers with tobacco. The levels of Securin were increased significantly in non-cancerous AN-chewers with and without tobacco with respect to non-chewers.
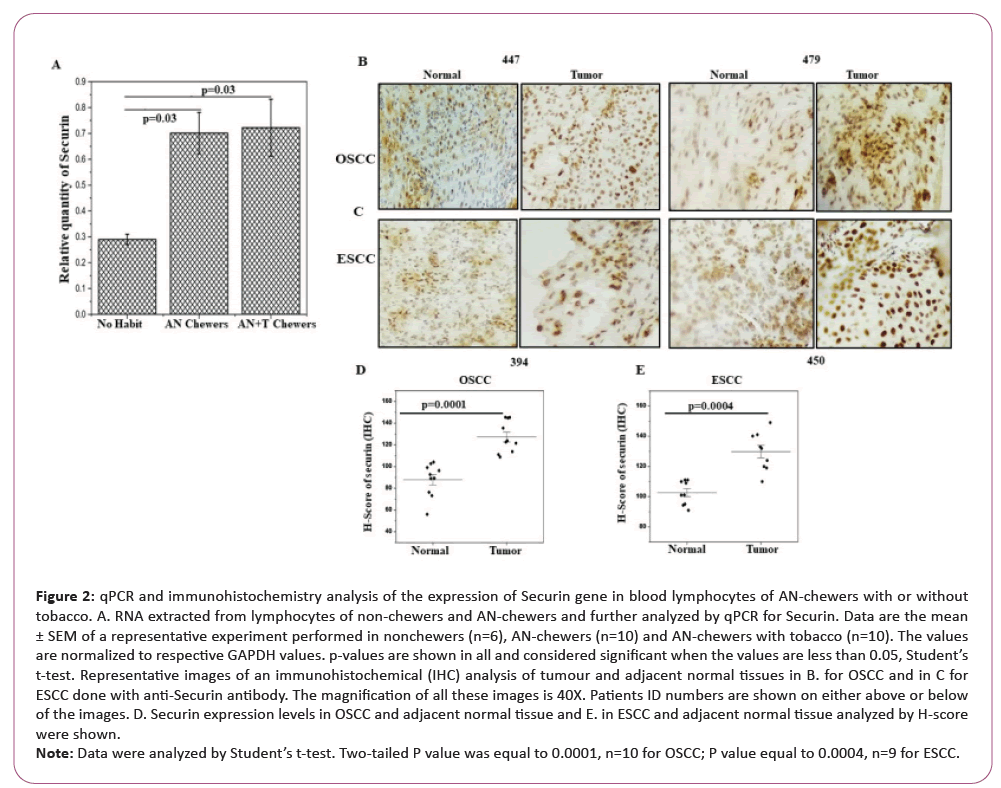
Figure 2: qPCR and immunohistochemistry analysis of the expression of Securin gene in blood lymphocytes of AN-chewers with or without
tobacco. A. RNA extracted from lymphocytes of non-chewers and AN-chewers and further analyzed by qPCR for Securin. Data are the mean
± SEM of a representative experiment performed in nonchewers (n=6), AN-chewers (n=10) and AN-chewers with tobacco (n=10). The values
are normalized to respective GAPDH values. p-values are shown in all and considered significant when the values are less than 0.05, Student’s
t-test. Representative images of an immunohistochemical (IHC) analysis of tumour and adjacent normal tissues in B. for OSCC and in C for
ESCC done with anti-Securin antibody. The magnification of all these images is 40X. Patients ID numbers are shown on either above or below
of the images. D. Securin expression levels in OSCC and adjacent normal tissue and E. in ESCC and adjacent normal tissue analyzed by H-score
were shown.
Note: Data were analyzed by Student’s t-test. Two-tailed P value was equal to 0.0001, n=10 for OSCC; P value equal to 0.0004, n=9 for ESCC.
Immunohistochemical staining for Securin in cancer samples
We studied Securin expression by immunostaining in a panel of patient samples of OSCC (n=10) and ESCC (n=9) who had the habit of chewing raw AN. Both the oral and oesophageal tumour sections showed significantly higher expression of Securin than did their adjacent normal counterparts as shown in Figures 2B and 2C. The mean H-score of Securin increased from 88 to 130 for oral cancer samples and 100 to 130 for oesophageal cancer samples as shown in Figures 2D and 2E. All these cancer patients are ANchewers with or without tobacco.
Early detection is associated with improved survival rates for both OSCC and ESCC. Therefore, it is important to diagnose these cancers before the symptoms develop, and thus the treatment can dramatically improve prognosis. It is believed that understanding the pathogenesis of these cancers will reveal biomarkers that can be vital to guiding early diagnostic strategies. Therefore, the objective of this study was to assess whether sister chromatid exchanges, precocious anaphases and expression of Securin can be used as an early biomarker to estimate the risk of cancer following RAN+lime exposure. Since, noninvasive, lowcost screening tools for these cancers are currently not employed and therefore, it is hoped that the present study will fulfil this demand and the results can be achieved within a short period.
Earlier studies demonstrated higher DNA damage and p53 expression, greater delay in cell kinetics and lower GSH-level in Peripheral Blood Lymphocytes (PBLs) of heavy AN-chewers than non-chewers in the state Meghalaya and therefore it was suggested that besides cytogenetical parameters, the level of endogenous GSH and p53 protein could act as an effective biomarker for RAN chewers [4]. Subsequently, it was shown that RAN+lime exposure for 240 days or more induces stomach cancer in mice with securin upregulation [9]. Further, it was demonstrated that the level of Securin is elevated in non-target cells like peripheral blood lymphocytes which in turn induced chromosomal instability in humans having the habits of chewing AN+lime with or without tobacco [10]. From these results, it was suggested that perturbations in components of the chromosome segregation machinery could be involved in the initial process of carcinogenesis which can be detected in the non-target cells like PBL in humans or BMC in mice [9,10].
The present study, therefore, was undertaken to further investigate if precocious anaphase could indeed serve as a biomarker for the identification of mitotic checkpoint defects in non-target cells like PBLs in AN-chewers besides Securin expression and SCEs. It is an established fact that Securin is involved in chromatid separation and its over-expression has been shown to induce aneuploidy, a hallmark of cancer [24,25]. Therefore, in the present study, the expression of Securin was monitored not only during the early days of exposure in non-target cells but also in the tumour cells. Here, a significant increase in the expression of Securin and PAS were observed in the PBL of both cancerous and noncancerous individuals with a habit of using AN with and without tobacco as it was also observed earlier [10]. Such upregulation of Securin induces aneuploidy, arising from chromatid mis-segregation in human cells [26]. In this study, PBL cells fixed at 88 h displayed a higher frequency of precocious anaphase and aneuploid cells and that most of these cells had completed 3 or 4 divisions after PHA stimulation. Earlier study showed that 88 h fixation showed higher frequency of PAS and aneuploidy than 56 h fixed cells [10]. This indicates that cell division is required to acquire the abnormal karyotype. Similar phenomenon has been observed not only in human cancer cell lines but also in a vast array of malignancies, including oral and esophageal squamous cell carcinoma [27,28]. Moreover, elevated levels of Securin are correlated with higher tumor grade, invasiveness and tumor vascularity [29]. Thus, it has been proposed that the Securin level may be considered a molecular marker that can be a potential therapeutic target for many cancers [20].
SCE is an error-free recombination mechanism that allows the physical quantification of exchanged genetic material between homologous sister chromatids [30]. In fact, Chromosomal Aberrations (CAs), Micronuclei (MN), and SCEs in peripheral lymphocytes have widely been used as biomarkers of genotoxic exposure and effects in humans. It was indicated earlier, that the frequency of SCEs does not appear to be associated with the cancer risk due to technical variation in SCE level [31,32]. However, exposure to various genotoxic carcinogens increase the frequency of SCEs and therefore SCEs have been used to monitor populations currently exposed to carcinogens [33-35]. In addition, it was suggested that human exposure to carcinogens will alter cytogenetic responses of cultured lymphocytes to subsequent mutagenic challenges such as mitomycin induced SCEs. Therefore, SCEs as a ‘mutagenic challenge marker’ may be an indicator for detection of previous exposure [36]. Moreover, the frequency of SCE is widely used as an indicator of CIN in response to genotoxic and environmental agents [15]. Thus, due to the readily quantifiable nature of SCEs with high sensitivity for revealing mutagen-DNA interactions and considering a convenient tool in experimental studies, we used SCE as a parameter in this study along with PAS and Securin expression. The present data indicate that like Securin and PAS, the frequency of SCEs was also increased significantly in every AN-chewers with or without tobacco. Such an increase in the frequency of SCE is an indicator of elevated recombination activity and genome instability, which is a hallmark of cancer [37]. Thus, analysis of SCE levels and PAS along with Securin expression in PBL can give relevant information on the genotoxic effect of AN and their role in acquiring CIN in the early stages of AN-induced carcinogenesis. In fact, all these parameters and their level of induction in PBL are thought to represent surrogate endpoints for more specific chromosome alterations in target tissues of carcinogenesis.
Therefore, the aim of this study was to investigate the impact of these three parameters as early diagnostic screening biomarkers for the people who have the habit of consuming tobacco, areca nut and alcohol for several years. We observed an elevation in the level of Securin and the frequency of PAS and SCEs in the non-target tissue of AN-chewers and therefore, these end-points could become potential intermediate biomarkers for the risk assessment.
This study was approved by the Institutional Ethics Committee for Human Samples/Participants (IECHSP/2016/31) in North-Eastern Hill University, Shillong, India. The tumor tissues as well as blood samples were obtained from the cancer patients after having their consent for participation and were individually interviewed before taking the biopsy. Informed consent was obtained from all the individuals studied before sample collection. Every biopsy was kept in RNA later soon after its collection and all experiments were performed in accordance with relevant guidelines and regulations. Biopsy and resection samples were reviewed by the pathologists in the hospital to confirm the diagnoses and also select representative blocks for immune histochemical analyses. All the donors had no viral diseases or antibiotic therapy during the last 6 months.
This work was supported by grants from the Department of Biotechnology, India VNOCI Project (102/IFD/SAN/4990/2017- 2018) to AC.
All the authors declare that they have no competing interests.
Citation: Chatterjee A, Singh C, Banerjee A, Boruah N, Swargiary P, et al. (2021) The Emerging Potential of Securin Upregulation, Premature Anaphase Separation: Sister Chromatid Exchanges as Intermediate End-points to Monitor Oral and Esophageal Carcinogenesis. Biomark J Vol. 7 No.11: 109.
Copyright: This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.