Research Article - (2015) Volume 1, Issue 1
1College of Chemistry and Chemical Engineering, Qinzhou University, Qinzhou, Guangxi 535000, PR China
2State Key Laboratory of Supra molecular Structure and Materials, College of Chemistry, Jilin University, Changchun 130012, PR China
*Corresponding Author:
Junqiu Liu
College of Chemistry and Chemical Engineering
Qinzhou University, Qinzhou
Guangxi 535000, PR China
E-mail: junqiuliu@jlu.edu.cn
Received Date: October 01, 2015; Accepted Date: October 15, 2015; Published Date: December 14, 2015
Citation: Liu J, Yin Y. Temperature Responsive Hydrogels: Construction and Applications. Polym Sci. 2015, 1:1.
Responsive hydrogels have attracted researcher’s special attentions. Up to now, a wide variety of responsive hydrogels are continuously constructed and the responsive mechanisms are deeply investigated. Typically, versatile responsive hydrogels with pH responsive, temperature responsive, light responsive properties are constructed. In this item, temperature responsive hydrogels received much attention and were also extensive investigated. The construction methods, responsive mechanism and related applications of the temperature responsive hydrogels may provide a significant guide in the redesign and application of responsive hydrogels.
Keywords
Benzophenone; PNIPAM microgels; Thermosensitive; Photocrosslinking; Nanofibres
Introduction
As a kind of smart materials, responsive hydrogels exhibit the rapidly responsive ability and sharp volume phase transitions according to the external stimuli from environment. Hydrogels demonstrate large-scale volumetric changes in response to small levels of stimuli. To the best of our knowledge, the most typical external stimuli factors related to responsive behaviors of smart hydrogels are Ph [1-5], temperature [6-8], light [9-11] and multi-factors [12-16]. Additionally, the responsive hydrogels based on enzyme responsive [17,18], calcium responsive [19,20], glucose responsive [21,22], redox responsive [23,24], ionicstrength responsive [25,26], and chemical responsive[27] are also exhibited amazing responsive abilities. Among these insights, temperature responsive hydrogels are particularly important.
This article gives an overview of the fundamentals of temperature responsive hydrogels. Some researches related to the construction methods, responsive mechanisms and applications of responsive hydrogels in the past few years are reviewed.
Construction Method of Temperature Responsive Hydrogels
The temperature responsive hydrogels introduced in this section are mainly prepared based on the scaffolds of supramolecular assembled structure [28-30], hydrogel films [31] and hydrogel sphere [32,33]. Additionally, the temperature responsive hydrogels constructed employing the novel technics are also summarized [34-36] (Figures 1 and 2). Ritter and coworkers pioneered in construction of temperature responsive hydrogel using N-isopropylacrylamide as monomer [37]. As illustrated in Figure 1 based on the host-guest interaction between adamantyl group and cyclodextrin, the thermosensitive hydrogel 1 was prepared. Similarly, based on the host-guest interaction between D3-symmetric tris(spiroborate) cyclophane and [Ir(tpy)2](PF6)3 complex, the assembled chain structure was obtained by Yamaguchi et al.[38]. Moreover, employing the host-guest interaction between cucurbit[8]uril and viologen/ naphthoxy, a thermo-sensitive supramolecular polymer hydrogel 2 was prepared by Scherman and coworkers[39]. Typically, as the dynamic cross-link of CB[8]/viologen/naphthoxy, this hydrogel exhibited thermal reversibility. Additionally, the temperature responsive hydrogels based on hydrophobic interaction of dipeptides [40] and cyclic macromonomer crosslinking interaction[41] were also reported. Using a photo-crosslinkable benzophenone unit, Knoll and coworkers prepared the responsive hydrogel films by photo-cross-link method based on poly(N-isopropylacrylamide)(PNIPAM) copolymer[42]. To fabricate hydrogel nanoparticles with ultrafine nanosized hair, Yang’s group developed a simple self-developed approach consisting of a two-step photoinduced polymerization of crosslinked polyacrylamide soft hydrogel nanoparticles (5-10 nmin diameter) grafted with PNIPAM chains. A fast association process was observed at approximately 33-34°C during the heating process. Additionally, Macdonald and coworkers reported a efficient method for fabricating monodisperse PNIPAM microgels enclosed by a single, continuous, impermeable lipid bilayer[43]. Furthermore, the novel construction protocols and developed technics have largely enhanced the innovation of preparation methods of temperature responsive hydrogel. Kiriy et al.[44] reported the preparation of PNIPAM hydrogel microcapsules by a one pot synthesis method [44]. Then, Hilt and coworkers developed the controlled synthesis method for responsive hydrogel nanostructures via microcontact printing and ATRp [45]. Based on the interface assisted synthesis method, the functional hydrogel particles were prepared by Serpe’s group. This approach might be used to synthesize particles with a variety of different functionalities and compositions.
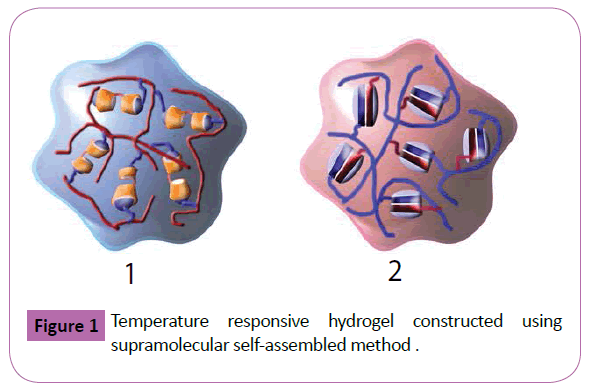
Figure 1: Temperature responsive hydrogel constructed using supramolecular self-assembled method .
![]()
Figure 2: The silicon-containing molecules used to preparing crosslinkable composite hydrogels.
Construction of Composite Hydrogels
As the functional materials are anchored into the composite hydrogels, the properties of composited hydrogel such as structural stability, mechanical strength and response behaviors are significantly improved. Usually, the functional materials including silicon-containing molecules [46], iron oxide [47], starch [48], liposome [49], alginate [50] are employed. As shown in Figure 2, to fabricate a thermo sensitive hybrid hydrogel thin film, Du’s group reported a facile method from a cross-linkable hydrid copolymer using silicon-containing monomer [46]. After hydrolysis of the methoxysilyl groups and subsequent condensation of the silanol groups, the hybrid hydrogel was formed. Using siliconcontaining molecule bearing benzophenone groups as material, Brom prepared a new temperature responsive hydrogel by photo crosslinking using UV irradiation [51]. This composited was very stable and exhibited excellent responsive properties. Additionally, combining a polyhedral oligomer silsesquioxane possessing eight epoxide groups and commercial PNIPAM, Lin et al. [52] constructed the thermo responsive hydrogel nanofibre [52]. These nanofibres showed excellent hydrogel characteristics with fast swelling and deswelling responses triggered by temperature changes (Figure 2). To obtain thermo responsive composite microcapsules that potential be used in remotely delivery system, Štepánek and coworkers synthesized the responsive microcapsules consisting of PNIPAM hydrogel and hydrophilic citrate-stabilized super-paramagnetic iron oxide nanoparticle [47]. And they described a novel method of synthesizing composite PNIPAM/alginate particles that changed their optical properties in response to a temperature change but kept both their original shape and size [50]. Moreover, Wang and coworkers presented a novel approach for fabricating the thermo responsive PNIPAM hydrogels based on starchbased Nano spheres using derivable allyl groups as Nano scale crosslinkers [48]. The obtained hydrogels possessed an extremely high mechanical strength and a remarkably rapid shrinking time. Additionally, Hong et al. demonstrated a microfluidic method to direct the assembly of liposome-PNIPAM hybrid nanoparticles [49]. Based on a radical-induced graft polymerization technique, Park’s group prepared a new series of thermo responsive graft copolymers, which exhibited thermo responsive sol-to-gel transitions depending on chain length of the grafted PNIPAM.
Hydrogel Temperature Responsive Mechanism
Herein, the temperature responsive mechanisms of hydrogels including switching kinetics [52], permeability [53],revival, mechanical and surface properties [54] were investigated. Various methods including atomic force microscopy [54] visualizing method [55] and theory research [53] were employed. Typically, Müller- Busch Baum and coworkers investigated the switching kinetics of thin thermo responsive hydrogel films [52]. They proved that the collapse transition proceeds could be seen as three steps. The first step was the shrinkage of the initially swollen film by a release of water. In the second step, the thickness remained constant with water molecules embedded in the film. In the third step, water was reabsorbed from the vapor atmosphere, thereby giving rise to a relaxation process. Furthermore, to investigate the swelling, permeability, and mobility of tracer molecules in thermo responsive hydrogel films, Raccis and coworkers employed fluorescence correlation spectroscopy to carry out the related researches [53].
Moreover, Knoll’s group studied the thermo responsive behavior of hydrogels with different thicknesses in MilliQ water by the surface Plasmon diffraction technique [56]. Furthermore, employing atomic force spectroscopy as analysis method, Jonas deeply investigated the temperature-induced structural and adhesive changes. And they also achieved the simultaneous measurement of mechanical and surface properties in thermo responsive hydrogel films using atomic force spectroscopy [54]. Additionally, near-infrared multispectral imaging microscope was used to measure images and spectra of individual hydrogel directly and simultaneously [57]. It was possible to determine lower critical solution temperature (LCST) values based on monitoring either change in the spectra or the volume of the hydrogel particle. Moreover, Wu et al. investigated precise chain collapse and revival thermodynamic mechanism of PNIPAM using two-dimensional correlation infrared spectroscopy and a newly developed perturbation correlation moving window technique. It indicated that in the heating process, PNIPAM hydrogel occurred to collapse along the backbone before water molecules were expelled outside the network. In the sequential cooling process, PNIPAM hydrogel had water molecules diffusing into the network first before the chain revival along the backbone occurred. Commonly, responsive hydrogels based on supra molecular assembled structures are endowed with property of reversible transformation. To reveal the transition mechanisms between self-assembled structures (micelle [58], vesicle [59]) and hydrogels, several investigated were carried out. Additionally, Frank [60] and Zhu [61] devoted themselves in exploring the interaction mechanisms between hydrogels and the surrounding hydrogel matrix.
Application of Temperature Responsive Hydrogels
The applications of the composite hydrogel with temperature responsive significantly focused on the preparation of bio-related materials [62]. Disease-associated blood biomarkers exist in exceedingly low concentrations within complex mixtures of highabundance proteins such as albumin is prepared biomarker based on the smart hydrogel by introducing an affinity bait molecule into N-isopropyl acrylamide [63].The hydrogel performed three independent functions within minutes:(a) molecular size sieving, (b) affinity capture of all solution-phase target molecules, (c) complete protection of harvested proteins from enzymatic degradation. Akaike et al. [64] regulated the cellular morphology using temperature responsive hydrogel for integrin-mediated mechanical force stimulation [63]. During gel swelling, changes of cell shape were clearly observed. And it proved that cells formed filo podia-like structures in response to mechanical cues, suggesting that mechanical forces facilitated actin polymerization at the peripheral region. Bhattacharyya’s group reported the synthesis of reactive Fe and Fe/Pd nanoparticles in a temperature responsive hydrogel [64]. These nanoparticles were stabilized in the hydrogel network and the chlorination reactivity in water was enhanced and controllable in the temperature range of 30–34°C. Additionally, the methylcellulose based thermally reversible hydrogel system for tissue engineering applications was developed by Devireddy and coworkers [65].
Moreover, Wells et al prepared the temperature responsive microcapsules with variable optical signatures based on the incorporation of quantum dots into a highly biocompatible hydrogel [66]. The resulting microcapsules displayed temperature responsive behavior, as the optical signature displayed a linear dependence on temperature over the range of 20- 45°C. Inci anchored the boradiazaindacene dye molecules to a polyethylene glycol based hydrogel with different concentrations using a picosecond time-resolved spectroscopic technique [67]. Based on this hydrogel, they investigated the dynamics of the fluorescence mechanism of boradiazaindacene dye molecules. Hilt et al. presented a demonstration of remote controlled flow regulation in a microfluidic device using a hydrogel Nano composite valve [68]. The swelling and collapse of the resultant hydrogel Nano composite was remotely controlled by application of an alternating magnetic field. Moreover, Xu and coworkers prepared the grapheme oxide/poly (acrylic acid-co-acrylamide) super-absorbent hydrogel nanocomposites67. Interestingly, the hydrogel Nano composites exhibited significant improvement of swelling capacity in neutral medium although they just contained 0.10 wt% of grapheme oxide. And they could also retain relatively higher swelling capacities to a certain degree at acidic and basic solutions. Additionally prepared novel hydrogel pore-filled composite membranes with tunable and temperature responsive size-selectivity based on PNIPAM hydrogel (Figure 3). Liu and coworkers introduced the active sites of glutathione peroxidase into temperature responsive PNIPAM hydrogels scaffolds [69]. Similarly to the catalytic behavior of glutathione peroxidase with the ability of scavenging of reactive oxygen species and protecting cells from oxidative damage, the functional hydrogels exhibited the typical glutathione peroxidase catalytic behavior. More importantly, the catalytic activity of this micro gel enzyme model displayed an obvious temperature responsive characteristic. Typically, the catalytic activity of the micro gel could be turned on and off reversibly by changing the temperature (Figure 3) Suzuki and coworkers firstly applied PNIPAM hydrogel as a particulate steric stabilizer in the dispersion polymerization of styrene [70]. It proved that some of the resultant polystyrene particles were covered with hydrogel particles. Yu et al. synthesized a thermo and pH responsive hydrogel using N-isopropyl acrylamide and potassium 4-(acryloyloxy) pyridine-2, 6-dicarboxylate as monomers [71]. This hydrogel functioned as scaffold for supported-palladium catalyst of copper-free Sonogashira reaction in aqueous media. Hu and coworkers prepared the monodisperse spheres composed of PNIPAM and used them as building blocks for a thin hydrogel film. This film not only absorbed a large amount of water like conventional hydrogel but also exhibited iridescent colors due to long range ordered micro gel arrays. Zhang et al. developed thermo sensitive macro porous hydrogel showing selectivity for the lysozyme based on metal coordinate interaction [72]. In the recognition process of the protein, the interaction of the imprinted thermo sensitive hydrogel to protein can be switched between the coordinate effect and the electrostatic effect by adding or not adding Cu ions. Moreover, based on the special recognition property of designed hydrogels, the absorption of bovine serum albumin [73] and reusable nickel (II) ions [74] were achieved using thermo sensitive hydrogels. Additionally, thermo sensitive hydrogels used to activate adaptive liquid micro lense were also developed.
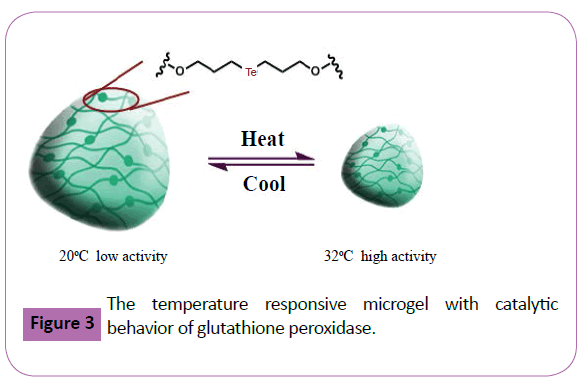
Figure 3: The temperature responsive microgel with catalytic behavior of glutathione peroxidase.
Conclusions and Outlook
With special three-dimensional polymer network cross linked by chemical bonds or physical nodes, hydrogels exhibited the unique property of swollen behavior. Commonly, responsive hydrogels can function as the amazing scaffolds for the fabrication of smart devices to investigate the physiological processes in living system and have attracted researcher’s special attentions. Herein, both the theory researches and exploration of construction methods of temperature responsive hydrogels are summarized. Moreover, the applications of temperature responsive hydrogels are also summarized, which may provide a significant guide in the redesign and application of responsive hydrogels. Usually, the mainly applications of hydrogels are focused on the regenerative medicine, drug delivery system and nano sensors. To expand the application fields of responsive hydrogels and to construct more amazing smart materials based on responsive hydrogels, there are still more efforts that needs to be done.
Acknowledgments
Financial support from the Natural Science Foundation of China (No: no. 21234004, 21420102007, 21574056 and 21221063), the Natural Science Foundation of Guangxi Province (No. 2013GXNSFBA019043), the Natural Science Foundation of Education Bureau of Guangxi Province (No. 2013YB254)