Research Article - (2023) Volume 7, Issue 3
Synthesis and Characterization of Nd2O3 Nanoparticles Using Urea as Precipitation Agent
Bahram Khoshnevisan1*,
Maryam Mohammadi1 and
Mohsen Moradian2
1Department of Physics, University of Kashan, Kashan, Iran
2Department of Chemistry, University of Kashan, Kashan, Iran
*Correspondence:
Bahram Khoshnevisan, Department of Physics, University of Kashan, Kashan,
Iran,
Email:
Received: 30-May-2022, Manuscript No. IPNNR-22-13690;
Editor assigned: 01-Jun-2022, Pre QC No. IPNNR-22-13690(PQ);
Reviewed: 15-Jun-2022, QC No. IPNNR-22-13690;
Revised: 20-Sep-2023, Manuscript No. IPNNR-22-13690(R);
Published:
27-Sep-2023, DOI: 10.12769/IPNNR.23.7.24
Abstract
Nano Particles (NPs) of Neodymium oxide were prepared by homogeneous co-precipitation method using Neodymium nitrate and urea as precursors. X-Ray Diffraction (XRD) patterns have been refined by the Rietveld method and the Fourier Transform-Infrared Spectroscopy (FT-IR) showed that the sample had a band gap about 4.59 eV. On the other hand, the Photoluminescence (PL) spectrum indicated that the sample had a low concentration of singly ionized oxygen vacancies. The paramagnetic property of the NPs has been also tested by Vibrating Sample Magnetometer (VSM) measurements.
Keywords
Rare earth elements; Homogeneous precipitation; Urea; Nd2O3 NPs; optical properties;
Rietveld refinement
Introduction
Rare earth elements are strategic resource with widespread
use in industry, agriculture, military industry, environmental
protection and medicine. Their compounds are widely used in
high performance luminescence devices, magnets, catalysts,
display devices, solid state lasers and other basic materials
due to the electronic, optical and chemical properties of their
4f-shell ions [1-3]. Among them, neodymium based materials
have found their place in various applications and products
including magnetic suspensions, photoluminescence, photo
materials, magnetic molecules, soft glass and soft magnets
(hexagonal or garnet crystal structure for hard magnets and
crystalline structure for soft magnets) [4-7]. For instance
Nd2O3 is widely used in the fields of photonics, super
capacitor applications, luminescence and thermo-luminescent materials, protective coatings, thin films, sensors (e.g. CO and
hydrogen gas, etc.) [8-13].
In addition, Nd2O3 Nano Particles (NPs) are widely used for
making glass, capacitor and magnets, so have received special
attention due to their optical properties. Since the size and
morphology of the particles have influence on their
properties and applications, such as ceramic catalyst
applications therefore; scientists are seeking to improve their
sensitizing methods (hydrothermal, combustion solution, solgel,
micro emulsion, etc.) [14-20]. Nd2O3 NPs have also
luminescence applications because their band gap is in the
range of 5.25-5.70 eV and the possibility of obtaining UV,
blue, green and red luminescence (emission range of 200-800
nm) for specific purposes is the most important in the fields of
optics and luminescence materials. The synthesis of
nanocrystalline Nd2O3 through an inductively coupled radiofrequency thermal plasma route could be led to a stable
hexagonal crystal structure and the synthesized nanoparticles
are highly uniform with an average size around 20 nm.
Production of fine NPs from starting nitrate water soluble
salts by homogeneous precipitation using urea is introduced
as a successful and efficient method to synthesize metal oxide
powders. Metal (hydrous) oxides with uniform particle size in
an aqueous medium were prepared and nowadays the
method is a promising issue used this method to synthesize
amorphous and crystalline metal (hydrous) oxide and
according to their study, the decomposition of urea in the
aqueous medium causes the formation of ammonia and
carbon dioxide slowly in the solution. The smooth pH increase
resulting from this degradation at the same time, along with
the releasing of OH- and CO2 ions usually leads to the
precipitation of metal oxide particles with controlled
morphology. In this study, Nd2O3 NPs have been prepared by
homogeneous co precipitation using urea and their structure
and the nanoparticles were characterized in terms of XRD
Rietveld refinement, morphology, size distribution and nature
of their chemical bonds. The XRD and scanning electron
microscopy are used for structural and morphological studies.
Fourier transform infrared spectroscopy is used to investigate
the nature of existing bonds. The optical response of the
nanoparticles is investigated through the electronic transition
of Nd3+ ions in its crystalline structure via DRS technique. PL
was used for studying presence of defect states and
corresponding activation energies in the samples and to
investigate the samples magnetic properties VSM technique
was employed, respectively.
Materials and Methods
Sample Preparation
To make the neodymium oxide NPs, according to the
literature with some modification, first 0.05 gr of Nd
(NO3)3.6H2O and 0.5 gr of urea was dissolved in 40 ml of
deionized water, and then the solution was solicited for 30
minutes. The mixtures were heated to 90°C and maintained
for 3 to 4 hours for sedimentation. This sediment was
calculated for 3 hours in the furnace at 800°C to obtain
neodymium oxide NPs. The chemical process would be along
the following relation:
2Nd (NO3)3+5CH4N2O→Nd2O3+5CO2+8N2+10H2O
Characterization
XRD has been employed for structural studies of the
synthesized NPs (XRD, philips expert pro, with Cu kα radiation
with λ=1.5405 Å), and the result was analyzed by Rietveld
refinement technique via GSAS (General Structure Analyze
System) software. The magnetic properties were studied by
VSM with the maximum field up to 10 kOe at room
temperature. The morphology and microstructure of the
sample were observed with a Field Emission Scanning
Electron Microscope (FESEM) (TESCAN BRNO-Mira3 LMU),
and Energy Dispersive X-ray (EDX) analysis was used for testing elemental composition (with accelerating voltage of
25.0 kV). IR spectrum was recorded with a KBr pellet on a
Perkin Elmer 781 Spectrophotometer. The Photoluminescence
(PL) studies have been carried out using a Perkin Elmer LS-55
luminescence spectrophotometer equipped with Xe lamp and
also The UV-Vis spectra of the Nd2O3 sample was obtained by
a Diffuse Reflection Spectroscopy (DRS) method (Shimadzu,
UV-1800, Japan).
Results and Discussion
The phase and crystal structure of the prepared Nd2O3 NPs by
the homogenous urea co precipitation method was examined
by rietveld refinements of the XRD pattern which is a unique
and comprehensive method for the determination of crystal
structures, which it makes a modeled (calculated) diffraction
pattern via the crystallographic features of the sample and
proceed for an interactive curve fitting with the observed
diffraction pattern. The refined profile is shown in Figure 1,
and some of the refined structural parameters like atomic site
occupancy and the relative position of the atoms in the unit
cell (x,y,z) are listed in Table 1.
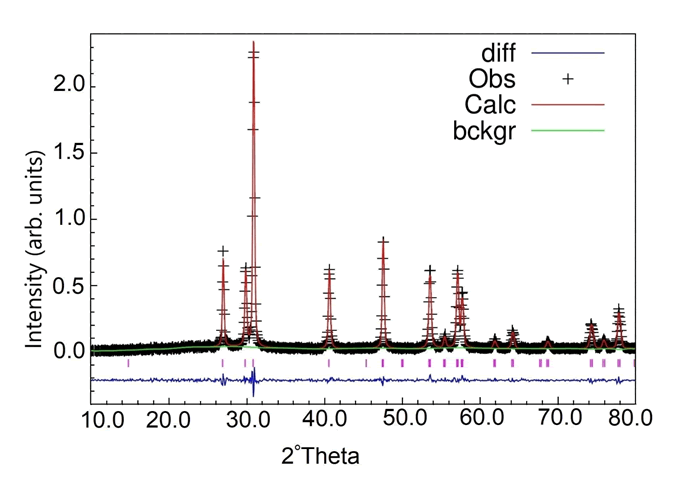
Figure 1: Rietveld refinement of the Nd2O3 NPs. Comparison
between the diffraction data (Obs) and the modeled data
(Calc) is shown by the difference cure (diff) which is almost
negligible and horizontal. The little tags in beneath of each
peak show the exact angle for the peaks.
According to the reference (JCPDS Card No.074-2139)
the structure was hexagonal and the difference curve
indicated a significant confirmation between the calculated
and the collected XRD pattern. The lattice parameter values
were obtained a=b=3.83Å; c=6.00Å and the cell
volume was calculated 77.68A3. The NPs were well
crystallized with very high purity. By using Scherrer's
equation:
D=kλ/βcosѲ
(Where k=0.89 is a constant, Ѳ is Bragg's angle and β is full
width at half maximum (FWHM) of the peaks), and with
consideration of refinement results in several directions, the
average size of the NPs was ~ 30 nm.
| a |
Oxidation state |
x |
y |
Z |
occupancy |
| Nd1 |
+3 |
0.333301 |
0.666700 |
0.240802 |
1 |
| Nd2 |
+3 |
0.666600 |
0.333299 |
0.747891 |
1 |
| O1 |
-2 |
0.000000 |
0.000000 |
0.034332 |
1 |
| O2 |
-2 |
0.333301 |
0.666700 |
0.633250 |
1 |
| O3 |
-2 |
0.666600 |
0.333299 |
0.346650 |
1 |
Table 1: Rietveld refinement results for Nd2O3
Figure 2 Shows the IR spectrum of the fabricated Nd2O3 NPs,
and the absorption peak at 409.58 cm-1 indicated Nd-O bond
vibrations.
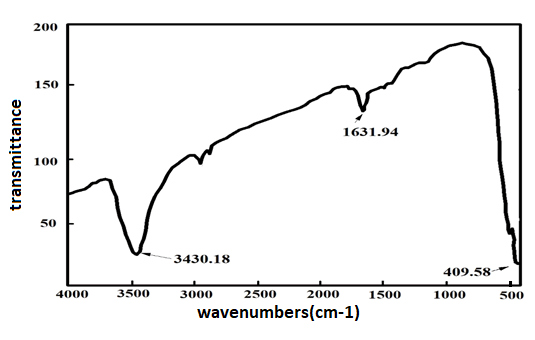
Figure 2: FTIR spectra of Nd2O3 NPs.
As the NPs are prepared in an aqueous medium and since the
organic material used in this study is urea, based on the
literature, urea reacts with water above 70°C and forms
ammonium, hydroxide, and carbonate ions (carbon dioxide),
which causes the pH rise up to 9 during the aging period
about 1 to 2 hours at 85°C, hence the absorption band at
1631 cm-1 and 3430 cm-1 are related to the stretching
vibration of C=O and the bending vibrations of OH,
respectively. Also the weak bands over the range 2500 to
3000 cm-1 suggest intermolecular H-bonds of water with
carbonate ions. Since urea is water soluble and is slowly
hydrolyzed only by heating above 70°C, result in a uniform
release of OH- ions and ammonia in the reaction system
according to the equation below:
(H2N2)2CO+3H2O→CO2+2NH4 ++2OH−
Therefore Nd (OH)3 is precipitated by Nd (NO3)3 in the
presence of ammonia, which upon further heat treatment
transformed into neodymium oxide.
Figure 3 shows the morphology (ellipsoidal) of the Nd2O3 NPs
as formed after heat treated at 800°C for 3 hours and its
fractional NPs size range is also shown inset. The NPs average
size is about 60 to 80 nm and according to the FESEM of
nanoparticles in two different scales of 1 μm (for general view
of the agglomerated sample) and 200 nm (to see the shape of
the individual NPs clearly). They were mostly ellipsoidal,
agglomerated and consisting of several interconnected
crystallites.

Figure 3: FESEM of the prepared Nd2O3 calcinated at 800°C (a)
1 μm; (b) 200 nm and the inset shows size distribution of the
NPs.
The elemental composition is shown by the EDX analysis and
there is somehow discrepancy between the reported atomic
ratio and its chemical anticipation but it must be recalled that
the EDX measurements for light elements like Oxygen would
not be very exact, at all (Figure 4).
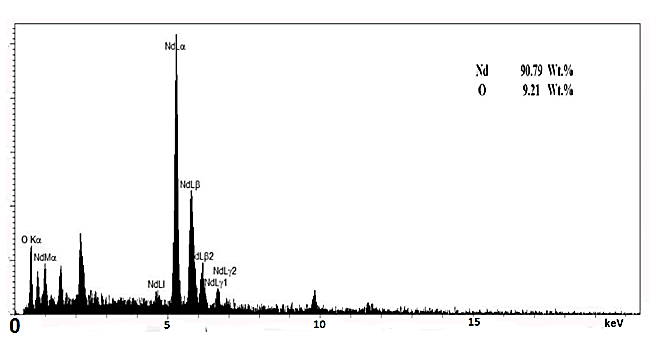
Figure 4: EDX result of Nd2O3 nanoparticles for elemental
composition of materials
DRS technique was employed to investigate the optical
property of the Nd2O3 NPs.The optical energy band gaps Eg of
the nano-and microcrystalline materials are calculated by
using the Tauc relation.
(αhν)2=A(hν-Eg)
Where α is the optical absorption coefficient near the
fundamental absorption edge and the band gap is obtained
from the intersection of the tangential slope of the high
energy range of the DRS spectrum and the horizontal axis
(Figure 5).
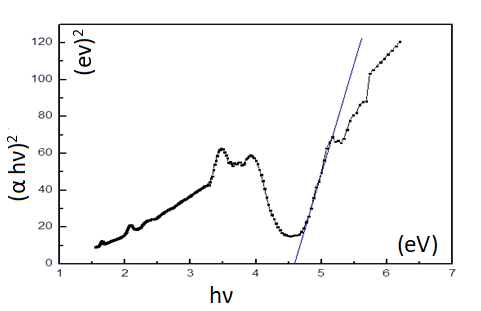
Figure 5: Finding the band gap of the Nd2O3 NPs.
The energy band gap of these nanoparticles is about 4.59 eV
and indicated that the Nd2O3 sample might be employed as a
photo catalyst under ultraviolet light. The complementary
study of the emission property of nanoparticles has been
done by PL spectroscopy and the spectrum was scanned in
the wide wavelength range of 200 nm ~ 800 nm. The
excitation wavelengths were 300, 340, 380 nm. The weak
peaks were observed at ~ 368-398 nm (UV) shown in Figure 6 related to the excitation wavelengths of 300 nm which these
emissions in UV region was attributed to singly ionized oxygen
vacancies in the Nd2O3 NPs (by the radioactive recombination
of photo generated holes and electron occupying the oxygen
vacancies. It is worth noting that the relatively low intensity of
the PL spectrum in this range(compared to the literature)
means that the sample had a low concentration of singly
ionized oxygen vacancies via this fabrication method,
moreover, the emission spectra also exhibit a series of
emission peaks with maxima at 418–481 nm (blue). The PL
peak 340 nm excitation shows series of emission peaks with
maxima at 440-484 nm (blue) and 519 nm (green), and also
the 380 nm excitation wavelength shows a maximum peak at
540 nm (green) (Figure 7). In general, The UV, blue and green
emissions exhibited the presence of surface defects and
recombination of electrons by shallow defect levels exciting
holes in the valence band. The blue and green emissions are
presumably due to surface defects such as Scotty and Frankel
in the lattice. In addition, they indicated that the fabricated
Nd2O3 NPs are a promising candidate for optical and
biomedical applications because the blue and the green
emission bands were result from (4I9/2→2P3/2, 2K15/2) and
(4I9/2→4G5/2, 4G7/2) transitions, respectively.
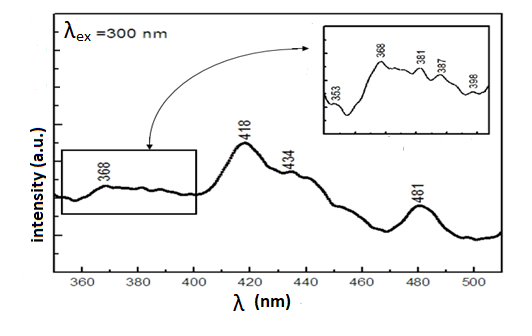
Figure 6: PL spectra of the Nd2O3 NPs in the range of 340 ~
520 nm.
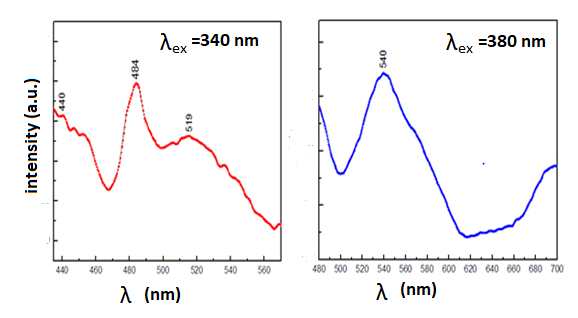
Figure 7: PL spectra of the Nd2O3 NPs in the range of 430~
580 nm and 480-700 nm.
For the investigation of the magnetic properties of the
sample, we have employed VSM measurement (Figure 8). The
coercively magnitude of these NPs was about 23.29 Oe. In
addition, the magnetization is linear with the applied field and
does not saturate even at a maximum applied field of 10 kOe
which reflected the paramagnetic nature of these NPs. This
paramagnetic property can be attributed to the relatively
unbalanced spins of the 3d electrons of the Nd3+ ion magnetic
moments.
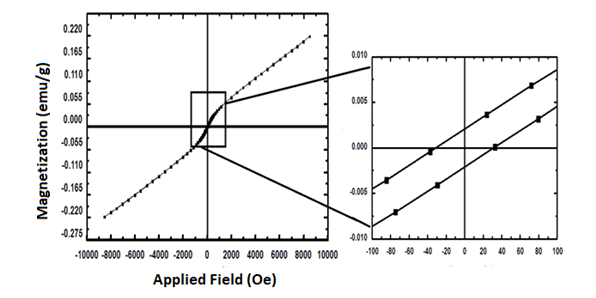
Figure 8: VSM of Nd2O3 NPs at room temperature.
Conclusion
In this study, the synthesis of Nd2O3 NPs by using the urea co precipitation method was investigated, which was confirmed by Rietveld refinement of the XRD pattern with the lattice parameters of a=b=3.83 Å; c=6.00 and volume cell 77.68 Å3 and space group of p3 m1. SEM image shows that the NPs were agglomerated and the average size of nanoparticles was in the range of 60 nm-80 nm. The energy gap of the NPs was obtained using the DRS analysis ( ~ 4.59eV). The PL spectrum of these NP investigated with excitation wavelengths of 300, 340 and 380 nm. At the excitation of 300 nm, the NPs showed the weak emission peaks at ~ 368 nm-398 nm in the UV region which means this sample has a low concentration of singly ionized oxygen vacancies. The 340 nm excitation wavelength showed series of emission peaks with the maximum at 440 nm-484 nm (blue) and 519 nm (green), and also at the 380 nm excitation wavelength showed a peak at 540 nm (green). By the VSM, it was found that the sample is paramagnetic that arising from the unpaired spins of the 3d electrons of the neodymium ions.
Conflicts of Interests
The authors indicate that there is no potential competing
interest.
References
- Qu X, Dai J, Tian J, Huang X, Liu Z, et al. (2009) Syntheses of Nd2O3 nanowires through sol–gel process assisted with porous Anodic Aluminum Oxide (AAO) template. J Alloys Compd. 469:332-335.
[Crossref][Google Scholar][Indexed]
- Qian L, Gui Y, Guo S, Gong Q, Qian X (2009) Controlled synthesis of light rare-earth hydroxide nanorods via a simple solution route. J Phys Chem Solids. 70: 688-693
[Crossref][Google Scholar][Indexed]
- Bazzi R, Flores-Gonzalez MA, Louis C, Lebbou K, Dujardin C, Brenier A (2003) Synthesis and luminescent properties of sub 5 nm lanthanide oxides nanoparticles. J Lumin. 102:445-450.
[Google Scholar][Indexed]
- Vera Serna P, Garcia Campos C, Sanchez de Jesus F, Bolarin Miro AM, Loran J, et al. (2016) Mechanosynthesis, Crystal Structure and Magnetic Characterization of Neodymium Orthoferrite. Mater Res. 19:389
[Crossref][Google Scholar][Indexed]
- Li W, Qu J, Wei X, Liu F, Yuan CL, et al. (2018) Structural characteristics and microwave dielectric properties of a new Sm2O3-Nd2O3-MgO-CeO2 ceramic system. Mater Chem Phy. 207:44-49.
[Crossref][Google Scholar][Indexed]
- Bissengaliyeva MR, Gogol DB, Bespyatov MA, Taimassova ST, Bekturganov NS (2019) Thermodynamic and magnetic properties of compounds in the system MeO–Nd2O3–Mo(W)O3 (Me=Mg, Ca, Sr). Mater Res. Express. 6:106109
[Crossref][Google Scholar][Indexed]
- Jan AM, Sahar MR, Sulhadi S, El-Mallawany R (2019) Thermal, structural and magnetic properties of TeO2-MgO-Na2O-Nd2O3 glass system with NiO nanoparticles. J Non Cryst Solids. 522:119566
[Crossref][Google Scholar][Indexed]
- Que W, Kam CH, Zhou Y, Lam YL, Chan YC (2001) Yellow to violet up conversion in neodymium oxide nanocrystal/titania/ormosil composite sol–gel thin films derived at low temperature. J Appl Phys. 90:4865-4867.
[Crossref][Google Scholar][Indexed]
- Kubra KT, Sharif R, Patil B, Javaid A, Shahzadi S, et al. (2020) Hydrothermal synthesis of neodymium oxide nanoparticles and its nanocomposites with manganese oxide as electrode materials for supercapacitor application. J Alloys Comp. 815:152104.
[Crossref][Google Scholar][Indexed]
- Umesh B, Eraiah B, Nagabhushana H, Sharma SC, Sunitha DV, et al. (2013) Structural characterization, thermoluminescence and EPR studies of Nd2O3:Co2+ nanophosphors. Mat Res Bull. 48:180.
[Crossref][Googlescholar][Indexed]
- Chevalier S, Bonnet G, Larpin JP. Metal organic chemical vapor deposition of Cr2O3 and Nd2O3 coatings. Oxide growth kinetics and characterization. 2000. Appl Surf Sci. 167:125-133.
[Googlescholar][Indexed]
- Sarma R, Saikia D, Saikia P, Saikia PK, Baishya B (2010) Pentacene based thin film transistors with high-k dielectric Nd2O3 as a gate insulator. Braz J Phys. 40:357-360.
[Crossref][Googlescholar][Indexed]
- Michel CR, Martinez-Preciado AH, Contreras NL (2013) Gas sensing properties of Nd₂O₃ nanostructured microspheres. Sensor Actuat B Chem. 184:8.
[Crossref][Googlescholar][Indexed]
- McKittrick J, Shea LE, Bacalski CF, Bosze EJ (1999) The influence of processing parameters on luminescent oxides produced by combustion synthesis. Displays. 19:169-172.
[Crossref][Googlescholar][Indexed]
- Fu YP, Wen SB, Hsu CS (2008) Preparation and characterization of Y3Al5O12:Ce and Y2O3:Eu phosphors powders by combustion process. J Alloys Compd. 458:318-322.
[Crossref][Googlescholar][Indexed]
- Jia G, Liu K, Zheng Y, Song Y, Yang M, et al. (2009) Highly Uniform Gd(OH)3 and Gd2O3:Eu3+ Nanotubes: Facile Synthesis and Luminescence Properties. J Phys Chem. 113: 6050-6055.
[Crossref][Googlescholar][Indexed]
- Kepinski L, Zawadzki M, Mista W (2004) Hydrothermal synthesis of precursors of neodymium oxide nanoparticles. Solid State Sci. 6:1327-1336.
[Crossref][Googlescholar][Indexed]
- Balboul BA, Myhoub AY (2010) The characterization of the formation course of neodymium oxide from different precursors: A study of thermal decomposition and combustion processes. J Anal Appl Pyrol. 89:95-101.
[Crossref][Googlescholar][Indexed]
- Sreethawong T, Chavadej S, Ngamsinlapasathian S, Yoshikawa S (2008) Sol–gel synthesis of Mesoporous assembly of Nd2O3 Nano crystals with the aid of structure directing surfactant. Solid State Sci. 10:20-25.
[Crossref][Googlescholar][Indexed]
- Jian-Hua R, Tong-Gang Z, Jian-Hua L, Juan K, Jia-Xin H, et al. (2008). Synthesis and photoluminescence properties of Nd2O3 nanoparticles modified by sodium bis (2-ethylhexyl) sulfosuccinate. Chin Phys B. 17:4669.
[Crossref][Googlescholar][Indexed]
Citation: Khoshnevisan B, Mohammadi M, Moradian M (2022) Synthesis and Characterization of Nd2O3 Nanoparticles Using Urea as Precipitation Agent. J Nanosci Nanotechnol Res. 6:44.
Copyright: © 2022 Khoshnevisan B, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.