Research Article - (2015) Volume 1, Issue 1
James J Sheng*
Professor in Department of Petroleum Engineering, Texas Tech University, USA
Received Date: December 03, 2015; Accepted Date: December 08, 2015; Published Date: December 22, 2015
Citation: Sheng JJ. Status of Alkalinesurfactant Flooding. Polym Sci. 2015, 1:1.
This paper provides an update on the status of alkaline-surfactant flooding technology. It covers the following topics: • Phase behavior of the mixture of soap and surfactant • Synergy of an AS system • Technical screening criteria • Laboratory and simulation work • Summary of AS field projects • Alkali and surfactant used • Study of AS application in heavy oil reservoirs • New development in China • Further discussions The data and analysis presented in this paper will provide readers with updated information about alkaline-surfacant flooding. Survey data will provide operators with reference data for their project design and optimization.
Alkaline-surfactant flooding; EOR; Synergy; Surfactant flooding; Alkaline flooding; Chemical Enhanced; Oil recovery
Alkali saponifies the naphthenic acids in crude oil to generate sodium naphthenate (soap) in situ. Some may believe the purpose of adding alkali to surfactant flooding is to generate soap so that the amount of injected surfactant can be reduced. Although generating soap is important, the following mechanisms are probably even more important:
• Reduction in surfactant adsorption because of the high pH from the alkaline injection,
• Improvement of the phase behavior of mixed surfactants of in situ generated soap and injected synthetic surfactant.
The synergies of alkaline-surfactant flooding will be addressed first. Essential experimental work is discussed. Technical screening criteria are updated. Field performance of alkaline-surfactant projects is summarized. Alkaline-surfactant application in heavy oil recovery is presented.
The phase behavior of the mixture of in situ generated soap and an injected synthetic surfactant may be best described with the aid of the activity map of 1.55% Na2O.SiO2 and 0, 0.1, and 0.2% NEODOL 25-3S with a Gulf Coast crude oil at 30.2°C, shown in Figure 1. In the bottom part of Figure 1, the horizontal dash line represents the sodium concentration from 1.55% Na2O.SiO2 solution, which is about 0.24 meq/g. The shaded area shows the type III microemulsion region of in situ generated soap. The mark 0 inside the shaded area means zero synthetic surfactant concentration. This bottom part of the figure shows that the sodium (salt) concentration from the alkaline solution is higher than the type III salinities that are around 0.5 meq/g. In other words, the soap generated microemulsion system is type II, not the optimum type III. Then the interfacial tension between this microemulsion phase and water will not be at minimum. As a result, the oil recovery may not be high. To solve this problem, the alkaline concentration should be reduced. If a lower alkaline concentration is injected, the alkali may be totally consumed by alkaline reactions with rock, oil and water, or the remaining salt concentration may be lower than the optimum type III salinity range. This is the dilemma: we cannot meet the two requirements of optimum type III salinity and alkaline consumption. Generally, the optimum salinity for soap is low [1]. The optimum salinity of a synthetic surfactant is higher than that of soap. If a synthetic surfactant is injected and mixed with soap, the optimum salinity of the mixed surfactant will have an optimum salinity in between. As shown in Figure 1, when 0.1% NEODOL is added, the optimum salinity range of the system is higher than that from 1.55% alkaline solution. To meet this optimum salinity range, sodium chloride needs to be added. Or a higher concentration of alkali can be added so that after alkaline consumption, the remaining salt concentration (ionic strength) can meet the type III salinity requirement. Figure 1 also shows the case when 0.2% NEODOL is injected. What is presented in Figure 1 is quite general for an alkaline-surfactant system. It shows that when a small amount of synthetic surfactant is added in an alkaline solution, the phase behavior of the mixture is optimized. This may be the most important synergy of alkali-surfactant combination. Furthermore, Liu [2] showed that the mixture of soap and a synthetic surfactant has a wider range of salinity in which the IFT is low (Figure 1). From the above example, it can be seen that the optimum salinity of a mixture depends on their concentrations and optimum salinities of soap and surfactant. The soap concentration depends on the crude oil available if we assume enough alkali is available. Then the mixture optimum salinity depends on water-oil ratio or oil saturation. The water-oil ratio is equivalent to the surfactant to soap ratio. Zhang et al. [3] found that the optimum salinity vs. the molar ratio of soap to surfactant falls on the same curve for different water-oil ratios. Based on experimental data, Salager et al. [4] proposed the following logarithmic mixing rule for the optimum salinity of a mixture:
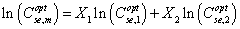 (1)
(1)
Where 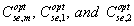 are the optimal effective salinities of the mixture and surfactant components 1 and 2, respectively. The surfactant mole fractions are X1 and X2. Puerto [5] used a linear mixing rule on the optimal salinity to fit their data. In fact, the logarithmic mixing rule has been found to slightly underestimate the optimal salinity, while the liner mixing rule was found to slightly overestimate the optimal salinity. In general, for high-electrolyte concentrations a linear rather logarithmic mixing rule to obtain the mixture optimal salinity is best, whereas at small electrolyte concentrations, a logarithmic mixing rule should be better [6]. Mohammadi [7] found that for the optimum solubilization ratios both logarithmic and linear mixing rules are satisfied:
are the optimal effective salinities of the mixture and surfactant components 1 and 2, respectively. The surfactant mole fractions are X1 and X2. Puerto [5] used a linear mixing rule on the optimal salinity to fit their data. In fact, the logarithmic mixing rule has been found to slightly underestimate the optimal salinity, while the liner mixing rule was found to slightly overestimate the optimal salinity. In general, for high-electrolyte concentrations a linear rather logarithmic mixing rule to obtain the mixture optimal salinity is best, whereas at small electrolyte concentrations, a logarithmic mixing rule should be better [6]. Mohammadi [7] found that for the optimum solubilization ratios both logarithmic and linear mixing rules are satisfied:
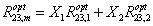 (2)
(2)
or
 (3)
(3)
where 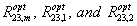 are the optimal solubilization ratio of the mixture and surfactant components 1 and 2, respectively.
are the optimal solubilization ratio of the mixture and surfactant components 1 and 2, respectively.
The synergies are discussed in terms of its effects on phase behavior, IFT, surfactant adsorption, emulsion and wettability.
Effect on phase behavior
Overall, the incremental oil recovery from field alkaline projects is low [8]. From the discussion of phase behavior of an AS system in the preceding section, it can be understand that one of the reasons for the low alkaline performance is that the soap microemulsion is not at an optimum salinity. Adding a synthetic surfactant can lead to the optimum type III microemulsion. Therefore, the most important synergy of an AS system is to optimize AS phase behavior. Liu [9] found that adding nonionic surfactant (BD-185) improved the salinity-resistant capability of anionic surfactant petroleum sulfonate (TH-1) in an alkaline-surfacntat solution. Zhou et al. [10] did a similar experimental study.
Effect on IFT
Liu [11] observed that when 1% sodium carbonate was added to a surfactant system, the salt concentration range for the low IFT region (<10−2 mN/m) is much wider. This synergy makes the AS system more robust in practical applications. Martin [12] attributed such behavior to ionization of the carboxylic acid by alkali. Jackson [13] observed that adding sodium carbonate made a microemulsion to form more quickly. Liu [14] found that such synergy was enhanced when a combination of NaOH and Na2CO3 is used with a surfactant.
Effect on surfactant adsorption
The primary mechanism for the adsorption of anionic surfactants on sandstone and carbonate formation material is the ionic attraction between mineral sites and surfactant anion [15]. As an alkali is added, the solution OH- concentration is increased, represented by increased pH. The increased OH- neutralizes some of positively-charged edges of clay minerals, thus reduces the ionic attraction between mineral sites and surfactants. Therefore, surfactant adsorption will be reduced in an AS system [16,17]. For carbonate formations, either hydroxyl ions [18] or sulfate ions Austad et al. [19] may reduce the rock surface charge. However, Liu observed from experimental data that that either hydroxyl ion or sulfate ion did not decrease the surfactant adsorption on the dolomite surfaces. But Zhang observed that carbonate ions reduced surfactant adsorption on dolomite rock surfaces. Their argument was that either hydroxide or sulfate is not a potential determining ion for carbonate surfaces, but carbonate ion .However, if the alkaline concentration is too high, as the concentration is increased, ionic strength is increased. Then flocculation of surfactant micelles may occur. Also, as ionic strength is increased, the counter ions in the diffusion layer may enter the adsorption layer to reduce the electrostatic repulsion between the anionic surfactant and sand surface. Consequently, surfactant adsorption may be increased with alkaline concentration. Also, cationic surfactant adsorption increases with pH [20]. The chemical EOR software, UTCHEM version 9 assumes that surfactant adsorption is affected by pH when pH is lower than some low value or higher than some high pH, and surfactant adsorption is reduced linearly as pH decreases when pH is within the range from the low pH to the high pH.
Effect on emulsion
Soap and surfactant together make emulsions stable owing to the reduced IFT. Emulsions can improve the sweep efficiency, thus oil recovery is improved with an AS system. Recently, one interesting paper was presented by Pei [21]. They compared alkaline performance with AS performance. The alkaline solutions were made of combined NaOH and Na2CO3. The AS solutions were alkaline solutions plus 0.1% anionic surfactant. The crude oil viscosity was 325 cP at 55 °C. The water TDS was 0.5%. They found that the AS solution can provide lower IFT than the alkaline solutions, but the oil recovery factors from the former were lower than the latter. Their micromodel pictures showed that the alkaline solutions could penetrate into the oil, forming droplet flow. Even though the lower IFT AS solutions had higher displacement efficiency, but they had lower sweep efficiency. As a result, the oil recovery factors were lower.
Effect on wettability
Li et al. [22] found that an AS solution changed water-wet sand wettability slightly when S was an anionic surfactant, whereas a water-wet rock changed to oil-wet permanently when S is a cationic surfactant. This indicates that anionic surfactant is more favorable for AS flooding.
No paper has been published to specifically address the technical screening criteria for alkaline-surfactant (AS) flooding. But several screening criteria for alkaline flooding or surfactant flooding alone have been proposed in the literature. Those criteria for separate alkaline flooding and surfactant flooding are listed in Table 1. Detailed discussions are in Sheng [23,24] and are not repeated. Each parameters from field alkaline flooding, surfactant flooding and AS flooding are also listed. The parameter values are generally the medians from their respective field projects. Finally, based on all of the other data, new criteria for AS flooding are proposed in Table 1. Basically, the new criteria must satisfy the criteria for both alkaline flooding and surfactant flooding. Note that this new screening criteria for AS flooding based on every parameter's satisfaction for both alkaline flooding and surfactant flooding at the same time. The screening criteria developed with this manner do not consider the AS synergy. The AS combination is expected to be more powerful than any individual. For instant combination of nonionic and anionic surfactant has better salinity tolerance than any individual as stated earlier in this paper. And the combination of nonionic and anionic surfactant has better thermal stability than any individual. The screening criteria considering the AS synergy are currently being developed and will be presented in a future publication.
Table 1: Summary of screening criteria for alkaline, surfactant and alkaline-surfactant flooding.
| Source | k (mD) | Tr (°C) | Formation water salinity, (TDS, ppm) | Divalent (ppm) | Lithology | Clay | µo (cP) | So (frac.) | Aquifer | Gas cap | API gravity | Acid number | Depth, ft |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AS projects | 54.5 | 72.2 | Sandstone | Low | 2.1 | 0.53 | 34.5 | 3900 | |||||
| Alkaline projects (24) | 240 | 45.2 | 24,313 | 145 | Majority sandstone | Low | 17 | 0.52 | Generally none | Generally none | 22.3 | 0.82 | 2650 |
| Proposed for alkaline (24) |
>10 | <93.3 | <50,000 | <100 | Sandstone | Low | <150 | >0.35 | Weak | Weak | NC | Organic acid | NC |
| Surfactant projects (23) |
152 | 25.3 | 39,750 | Majority sandstone | Low | 5.8 | 0.4 | Generally none | Generally none | 36.5 | NC | 1800 | |
| Proposed for surfactant (23) |
> 10 | < 93.3 | < 50,000 | < 100 | Sandstone | Low | <35 | >0.3 | Weak | Weak | NC | NC | NC |
| Proposed for AS | > 10 | < 93.3 | < 50,000 | < 100 | Sandstone | Low | <35 | >0.35 | Weak | Weak | NC | Organic acid | NC |
In the table, μo is the oil viscosity, So is the oil saturation before ASP, Tr is the reservoir temperature, NC means not critical.
For alkaline-surfactant flooding, one important experiment is to measure alkaline consumption. This is because we need to consider alkaline consumption requirement and optimum salinity requirement for the AS system when we design an AS injection scheme. Like any combination of chemical injection processes, compatibility tests must be done to ensure alkali and surfactant are compatible. We need to run salinity screen tests for surfactants and other fundamental experiments as described by Sheng. The salinity scan tests for surfactants are done at a fixed water-oil ratio (generally one). As discussed earlier, different water-oil ratios will generate different amount of soap. Soap phase behavior like optimum salinity is different from the surfactants. Then different water-oil ratios will require different optimum salinity requirement. After surfactant scan is passed, we need to repeat the salinity scan tests at different water-oil ratios to ensure the mixture of soap and surfactant will perform at desirable or acceptable conditions. Such experiment is called oil scan. The oil scan test results are generally presented in the called activity map like (Figure 1). A detailed description of these tests are described in Sheng [25]. At the end, coreflood tests are needed to see whether a selected AS formula will produce significant incremental oil recovery. Simulation of surfactant flooding using UTCHEM is relatively easier. The main challenge to simulate AS flooding is to simulate alkaline flooding which is very tedious in UTCHEM. A detailed description of alkaline flooding is provided in Sheng [25,26] described how to simulate AS phase behavior, and simulation results were analyzed. More sensitivity results of AS parameters are presented in Sheng. The status of general chemical EOR simulation and issues are discussed in Sheng [27].
Only 13 field projects were found, all in USA onshore except Angsi in Malaysia offshore (Table 2). All field projects were carried out in sandstone reservoirs. For the Angsi project, only single chemical tracer test was carried out. For AS projects, alkali and surfactant must be injected in the same slug. Sometimes, a preflush of alkaline slug was injected, for example, in the West Ranch project [28]. The objective was to improve the water-wetness of the rock and condition the reservoir, so that the subsequent surfactant loss was minimized. After a main AS slug is injected, sometimes polymer post-flush is followed before waterflooding, so that the subsequent water will not finger into the AS slug. Unfortunately, project performance data like incremental oil recovery are not much available in the literature. The amount (average) of alkalis injected from the 4 projects with data available was 25 PV×concentration (product of injection pore volume in % and injection concentration in %). This account is higher than that used in alkaline flooding projects alone which was 17, but lower than the amount used in the surveyed ASP projects which was 43.16 by Sheng [27]. The average of surfactants was 59 (product of PV in % and concentration in %). This amount is close to that from the surveyed surfactant flooding projects alone which was 52 by Sheng [23], and much higher than that used in the surveyed ASP projects which was 9.44 by [27]. The amount of polymer in the postflush from two surveyed projects was 1.36 (PV in % × concentration in %). This amount is close to that from the postflush of surveyed ASP projects which was 1.94 [27].
Table 2: Summary of alkaline-surfactant projects worldwide
| Project | Start Date | Tr, °C | Injectors | Producers | Porosity | Perm | FW-TDS | FW-divalents | OilVisc-live | TAN | References |
| Angsi | 119.0 | 2 | 25,000 | 2200 | 0.30 | 0.478 | Ibrahim[49], Zainal[50], Manap[51] | ||||
| Big Muddy 1 | 6-Nov-1974 | 47.8 | 4 | 1 | 0.194 | 56 | 4 | O&GJ, Farrel[52], Borah and Gregory[53], Farrel[52] | |||
| Big Muddy 2 | 14-Aug-1973 | 4 | 1 | Gilliland and Conley[54] | |||||||
| Big Sinking | 21-Nov-2003 | 47 | Miller [55] | ||||||||
| Doe | 1-Dec-1991 | 121.1 | 1 | 1 | 0.14 | 20 | 2 | O&GJ | |||
| Doris | 1-Jun-1993 | 31.7 | 4 | 10 | 0.15 | 10 | 1 | O&GJ | |||
| Driscoll | 1-Dec-1995 | 68.3 | 1 | 1 | 0.16 | 60 | 15.6 | O&GJ | |||
| Ittner Field Unit | 74.0 | 0.20 | 2.04 | 0.05 | Martin et al. | ||||||
| Pownall Ranch | 1-Feb-1996 | 82.2 | 5 | 11 | 0.16 | 70 | 8 | Kon et al.[56], Pope[57], Pownall Ranch[58] | |||
| West Ranch | 1-Jun-1974 | 76.1 | 1 | 0.31 | 950 | 0.7 | Murphy et al. [59] | ||||
| White Castle | 63.8 | 1 | 2 | 0.31 | 2.8 | 1.5 | Falls et al.[60], Shahinand Thigpen [61] | ||||
| Wichita Co. Regular | 1-Oct-1975 | 31.7 | 10 | 20 | 0.22 | 53 | 160000 | 2.2 | Talasjand Strange[62], Hammershaimb et al.[63], Delamaide et al.[64] | ||
| West Harrisburg Unit | Martin et al.[65]. |
In the AS field projects, these alkalis were used: sodium hydroxide, sodium carbonate, sodium metasilicate, sodium tripolyphosphate and potassium hydroxide. Dong et al. [29] found that sodium hydroxide and sodium carbonate is a better combination to maximize oil recovery efficiency in their channeled sand packs. The surfactants used were sodium sulfonate, petroleum sulfonates like ORS-162HF and Exxon 914-22, Internal Olefin Sulfonates, and NEODOL25-12. Surfactants used in laboratory research include sodium alkane sulfonate (Bio-Terge PAS-8S by Stepan), sodium C14-16 olefin sulfonate by Stepan, secondary alcohol ethoxylate (Tergitol 15-S-20 by Dow Chemicals), tridecyl alcohol with 30 ethoxy groups (TDA-30EO by Sasol), sodium olefin sulfonate (Petrostep C1 by TIORCO), and sulfo betaine (S13-B by TIORCO).
One of the important alkaline flooding mechanisms is emulsion flooding Johnson et al. [30]. As the interfacial tension at the emulsion interface becomes smaller, the stability of emulsion is increased. Adding surfactants in alkaline solution helps to reduce the interfacial tension. Also, heavy oils generally have a high content of acid components. Thus relatively high amount of surfactant (called soap) can be generated by the alkali-crude oil reaction. Emulsions could be either water-in-oil (W/O) type, oilin- water (O/W) type or the mixture. O/W emulsification converts viscous heavy oil into a low viscosity water-continuous emulsion. This mechanism is advocated by some authors, e.g., McAuliffe [31], Jennings et al. [32], Liu et al. [33]. O/W emulsions were only observed in a few coreflood cases, probably the oil drops coalesce into an oil bank Liu et al [34]. Bryan and Kantzas [35],observed that the mechanism of emulsification and entrainment of O/W emulsions is not efficient in recovering oil; the dominant mechanism is emulsification and entrapment. W/O emulsions are more viscous than the constituent heavy oil phase resulting a favorable mobility ratio, as supported by Wang et al. [36]. It is more likely that W/O emulsions are formed in heavy oil because of high viscosity of oil and in oil-wet conditions Bryan [37]. Both types of emulsions improve the mobility ratio by altering the effective viscosity of either the oil or the injected phase. The results from Bryan and Kantzas [35,38] have suggested that the fluid re-distribution and diversion in the porous medium is more important than the change in fluid properties to improve oil recovery. Bryan and Kantzas [39] concluded that both types of emulsions have the same oil recovery mechanism which is blocking off water channels and thus improving sweep efficiency. To make this water-blocking mechanism work effectively, the injection rate or pressure gradient should be kept low. Kumar et al. [40] studies the emulsion behavior of oil-brine-surfactants and evaluated the selected AS solution in sand packs in thin steel tubes. Relatively hydrophilic surfactants were added to form oilin- water emulsions at a low concentration in the presence of sodium carbonate. The viscosity of oil-in-water (O/W) emulsions was low compared to the oil viscosity. The incremental oil recovery factor over waterflooding was 25-40% OOIP. The oil viscosity was about 10,000 cP. Subsequently, Kumar and Mohanty [41] conducted AS flooding in a water-wet 5-spot 2D sand pack. The main mechanism was that the O/W emulsion phase fingered into the heavy oil phase. They advocated O/W emulsions.
Several laboratory studies were also conducted in China to explore AS applications in heavy oil reservoirs [34,42-45]. Xong et al. [42] found that the air permeabilities from 50 core measurements were reduced after AS flooding, indicating AS caused formation damage.
It has been observed that during an alkaline-surfactant-polymer flooding, the injection pressure becomes too high, as a result, the injectivity is decreased; and the chemical cost is high. To make more effective use of polymer benefit to improve sweep efficiency and alkali-surfactant benefit to improve displacement efficiency, alternating injection of alkaline-surfactant and polymer was proposed in China by Li [46]. Here is a field test in Daqing. The test area was 2.02 km2, and the pore volume (PV) was 5,284,690 m3. The formation net pay was 12.2 m, and the permeability was 624 m. There were 28 injectors and 40 producers. The following were the main events:
1. August 2006, waterflood,
2. November 8, 2008 – May 25, 2009, 0.1037 PV pre-flush polymer,
3. May 27, 2009 – June 1, 2013, 5 cycles of AS alternating P (polymer) injection,
4. June 2, 2013, polymer postflush,
5. By November 30, 2013, total 0.772 PV injections.
A laboratory study showed that the chemical cost of AS alternating P was reduced by 22.57% compared with ASP to reach a similar oil recovery. The following have been observed from the field test compared with ASP zones.
• More than 50% of wellhead samples showed stable ultra-low IFT.
• 4% higher injectivity
• 4% oil recovery higher
• Improved injection profile
For more research work, see Jiang et al. [47], Han [48].
Apparently, for chemical EOR methods, many individual chemical methods have been tested or implemented in field. See the published survey papers for alkaline flooding by Sheng [24], surfactant flooding by Sheng [8], especially polymer flooding. For the research, alkaline-surfactant-polymer flooding was focused in the past years Sheng [27]. Not many projects were based on any combinations of two chemical methods like AS. For the AS flooding, most important mechanism may be the emulsion mechanisms to improve sweep efficiency, based on the most important synergy of improved phase behavior of an AS system. Recent research effort has been made to explore the AS applications in heavy oil reservoirs.