Review Article - (2016) Volume 2, Issue 1
Madhavi latha Samala* and Gowripattapu Sridevi
Department of Pharmaceutics, Aditya Pharmacy College, Aditya nagar, ADB road, Surampalem, India.
*Corresponding Author:
Madhavi LS
Department of Pharmaceutics
Aditya Pharmacy College, Aditya nagar
ADB road, Surampalem, India
Tel: 9963791317
E-Mail: madhavilatha61@gmail.com
Received Date: April 26, 2016; Accepted Date: May 05, 2016; Published Date: May 12, 2016
Citation: Samala ML, Sridevi G. Role of Polymers as Gelling Agents in the Formulation of Emulgels. Polym Sci. 2016, 1:2.
Background: The present review focuses on the critical piece of different polymers as gelling experts in the enumerating of emulgels. The concentrate moreover accomplishes the novel procedure for availability and appraisal parameters of emulgels.
Methods: Late advances for the conscious use of gelling administrators in arrangement of various emulgels are consolidated. A variety of new polymers are evolving these days which are showing multi works basically as thickening agents and emulsifying agents. The new polymers have remarkable property of gellation that is useful in the formulation of stable dispersed systems. Research and overview works of various specialists were altogether considered and joined into the present study.
Findings: Types, uses, method of preparation and a few evaluation tests for emulgels are given in this review. The use of gelling agents either alone or in combination in various emulgel formulations were also represented.
Conclusions: The application of various gelling agent polymers in emulgel preparation and their numerous advantages provides the formulation scientists the wide choice of polymers for use in other pharmaceutical formulations. The favourable physical and rheological properties of emulgels with greater shelf life and better patient compliance than other delivery systems are useful in the formulation of topical dosage forms
Keywords
Emulsified gel, Gelling agent polymers, Carbomers, Hydrophobic drug delivery
Introduction
Gels are new class of semisolid estimation outlines made by ensnarement of significant measure of liquid and hydro-alcoholic liquid in an arrangement of colloid solid particles like inorganic substances or common polymers of trademark or made origin [1,2]. The use of Gels, a novel semisolid measurements shapes has reached out in both cosmetics and pharmaceutical industry nowadays. Even though gels are having many advantages of better patient compliance, less interaction with food and drugs and avoiding first pass effect these exhibit a major drawback of difficulty in delivery of hydrophobic drugs. To thrashing this confinement another procedure in light of emulsion system have risen i.e., Emulgels. Various novel polymers are being building up nowadays which are demonstrating multi works basically as thickeners and emulsifiers. The new gelling agent polymers possess the property of gellation that is useful in the formulation of stable systems by reducing interfacial and surfacial tension thereby increasing the viscosity of the liquid phase [3]. In this way the circuit of gelling administrators into water stage changes over the emulsion into stable emulgel formulation [4,5]. Emulgels are nothing but emulsions that may be of water-in-oil or oil-in-water type which when blended with gelling agents gets jellified. Emulgels acts as excellent medium for hydrophobic or water insoluble drugs [6,7]. The topical movement structure, for instance, emulgel generally used where the other forms of prescription association fails to respect cutaneous disorders for instance, parasitic defilements, psoriasis, skin break out particularly. Since the mid 1980's, emulsion gels have been of creating criticalness in the field of pharmaceutical semisolid formulatios [8,9].
Drug Conveyance Over the Skin
The skin deterrent properties stay in the fringe layer, the stratum corneum. It is 10-15 μm thick grid of dried out, dead keratinocytes embedded in a lipid matrix [10,11]. Two critical layers are there in the skin i.e., epidermis and dermis. Veins are scattered richly underneath the skin in subcutaneous layer. There are three fundamental instruments for pharmaceutical maintenance through the skin: intercellular, transcellular and follicular. The accompanying most essential course of transport is through the pilosebaceous course immersion tends to happen through intercellular cross section, however through transcellular pathway it has been seemed to give a speedier choice course of extremely polar particles. Typically the keratinized corneocytes and non polar lipid horny layer provide major obstruction to permeation of drugs [12]. The pharmaceutical passage for skin can be enhanced by using regular solvents, for instance, propylene glycol, surfactants and DMSO. The immersion enhancers changed the limit properties of the stratum corneum by sorts of part including enhancing dissolvability, distributing the stratum corneum, fluidizing the crystalline structure of the stratum corneum [13]. Creams and gels that are rubbed onto the skin have been used for very much quite a while for effective treatment against pollutions and torment by arrangement. New progressions now allow distinctive meds to be absorbed through the skin. These can be used to treat the impacted regions of the skin and also the whole body by systemic route (Table 1) [14].
| Physiological factors | Physicochemical components |
|---|---|
| Skin thickness and sort of skin | Partition coefficient |
| Lipid content | Molecular weight(<400 dalton) |
| Skin pH | Degree of ionization (unionized prescription gets absorbed well) |
| Thickness of hair follicles and sweat organs | Effect of vehicles |
| Blood Circulation system | |
| Hydration and aggravation of skin |
Table 1: Factors influencing topical assimilation of the drug [15,16].
Emulgel Comprises of Two Fundamental Constituents
1. Emulsion, 2. Gel
? Emulsion [17] are unstable biphasic liquid dosage forms consisting of two immiscible liquids one of which is dispersed as globules in to the other and can be stabilized by the addition of third agent called emulsifying agent. Emulsions can be both of O/W and W/O sort. These are used as vehicles to pass on the medicines (Figure 1).
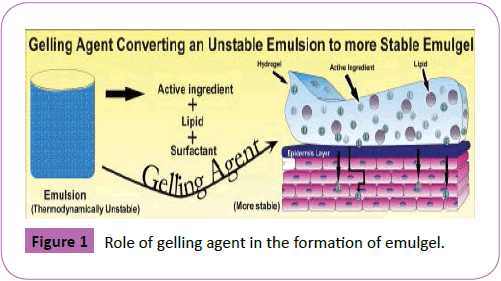
Figure 1: Role of gelling agent in the formation of emulgel.
? Gel [18] involves a physical state which shows the properties widely appealing between those of solids and liquids. Gels involve polymer which swells in the proximity of fluid. Gels are fragile and wet and look like solid material and shows broad bending from solid to liquid state.
Types of Emulgels
? Macro-emulsion gel: The particle size of the globules in these emulgels is more than 400 nm. They are apparently obscure. They can be offset using surface element agents [19,20].
? Nano-emulgel: These are confined by joining of nano-emulsion into gel. Nanoemulsions are thermodynamically enduring clear scatterings of oil and water offset by the proximity of surfactants and co-surfactants. These emulgels have a globule size of under 100 nm [21-23].
? Micro-emulsion based emulgel: These emulgels includes joined properties of microemulsion and gel giving high bioavailability of prescription. The globule size degree from 10 to 100 nm [24].
Advantages of Emulgels
Emulgels have a few points of interest as novel topical conveyance system [25-27]:
1. Emulgels help in the joining of hydrophobic solutions easily into the oil stage and a short time later smooth globules are scattered in liquid stage achieving O/W emulsion and this emulsion can be viably mixed into gel base which gives better reliability and entry of the prescription
2. Better security: Emulgels are more consistent than other transdermal plans.
3. Better stacking limit: Due to gigantic framework emulgels have better stacking limit stood out from other novel philosophies like niosomes and liposomes.
4. Production achievability and less arrangement cost: Preparation of emulgels require basic and few stages which diminish the expense of generation. So these novel measurements structures are conservative.
5. No escalated sonication is required there by item debasement does not happen.
6. Controlled release [28]: Emulgels can be used to postpone the effect of drugs having short half life.
7. Increases contact time and mean living course of action time of the medicine.
8. Emulgels can be used as a piece of helpful purposes.
9. They are less slick and easy to apply growing the patient consistence.
Formulation of Emulgels
Emulgels [29] can be detailed utilizing taking after Excipients.
1. Vehicles [30]: The vehicle is a basic association between medicine power and accommodating suitability. Two components are of essential importance in selecting the dermatological vehicle. They are: Solubilizing the medicine in the vehicle and boosting advancement of pharmaceutical from vehicle to stratum corneum.
Vehicles are of two types [31].
a. Aqueous solvents: These structure watery period of the emulsion. Water and alcohols are generally utilized.
b. Oils: These structure slick period of the emulsion. Comprehensively used oils are non-biodegradable mineral and castor oils, fish liver oils, changed oils of vegetable starting, for instance, arachis, cotton seed and maize oils.
2. Emulsifiers: Emulsifying agents are used to lift the emulsification and to offset the arranging. Some of them are polyethylene, glycol 40 stearate, navigate 80, tween 80, stearic acid [32,33].
3. Gelling agents [34]: These are one of the thickening authorities used to manufacture the consistency of the formulation. Gelling administrators encounter an abnormal state of cross-interfacing or alliance when hydrated and scattered in the disseminating medium, or when separated in the scrambling medium. This cross-interfacing or relationship of the scattered stage will change the thickness of the scrambling medium. The improvement of the diffusing medium is constrained by the scattered stage, and the consistency is extended (Figure 2).
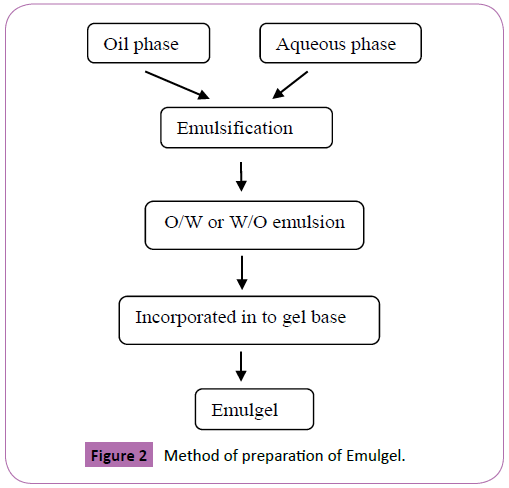
Figure 2: Method of preparation of Emulgel.
Types of gelling agents: There are a variety of polymers acting as gelling agents [35,36].
a. Natural polymers-Proteins like gelatin, casein, collagen, egg whites, polysaccharides like guar gum, acacia, tragacanth, bug bean gum, pectin, starch, xanthan gum, dextran, succinoglucon etc (Tables 2 and 3).
| Polymer Name | Viscosity | Properties |
|---|---|---|
| Carbopol® 910 | 3,000-7,000 | • Effective in low fixations. • Will give a low consistency formulation. |
| Carbopol® 934 | 30,500-39,400 | • Effective in thick details, for example, emulsions, suspensions, sustained release formulations, transdermals, and topicals. • Forms clear gels with water. |
| Carbopol® 934P | 29,400-39,400 | • Same properties as 934, however expected for pharmaceutical plans. • "P" = exceptionally purified product |
| Carbopol® 940 | 40,000-60,000 | • Effective in thick formulations. • Very great clarity in water or hydroalcoholic topical gels. • Forms clear gels with hydroalcoholic frameworks. |
| Carbopol® 941 | 4,000-11,000 | • Produces low consistency gels. • Very great clarity. |
Table 2: Different grades of Carbomers.
| S.No | Drug | Type | Polymer | Purpose | Reference |
|---|---|---|---|---|---|
| 1 | Chlorphenisn | Emulgel | Carbopol 934,HPMC | Effect of gelling agent on release | [6] |
| 2 | Nimesulide | Gel | HPMC,Carbopol940, Natural polymer | Effect of gelling agent on release | [7] |
| 3 | Ketoconazole | Emulgel | Carbopol- 934,940 | Comparative study of polymer and drug release | [4] |
| 4 | Fluconazole | Liposomal gel | Carbopol-934 | Increase permeation and deposition | [8] |
| 6 | Miconazole | Emulgel | Carbopol- 940,934 | Controlled delivery | [12] |
| 7 | Mefanamic acid | Emulgel | Carbobol 934, HPMCK4M | Release study and Pharmacologic action | [13] |
| 8 | Aceclofenac | Gel | Carbopol, HPMC,Sod. CMC | Carbopol gel show superior release | [15] |
| 9 | Clotrimazole | Jojoba oil based emulgel | Carbopol 934 P, HPMC | Effect of different concentrations of polymers | |
| 10 | Clotrimazole | Emulgel | Carbopol 934 | Study of rheological property | |
| 11 | Piroxicam | Emulgel | Carbopol- 940,934 | Comparative study of drug release | |
| 12 | Ibuprofen | Gel | Chitosan | Study of topical and systemiceffect | [16] |
Table 3: Use of gelling agents as polymers in various gel formulations [1].
b. Semisynthetic polymers-Cellulose subordinates like carboxymethyl cellulose, ethylcellulose, hydroxyethyl cellulose, hydroxypropyl cellulose, magnesium aluminum silicate (Veegum®), methylcellulose, sodium alginate etc
c. Synthetic polymers-Carbopols® (now known as carbomers), poloxamers (Pluronics®), polyvinyl alcohol.
? Cellulose polymers
a. Methylcellulose (MC) – used at 1500 cps. It makes more thin gels however with high resistance for drugs. It is great with water, alcohol (70%) and propylene glycol (half). It is exacerbated with 1/3 bubbling heated water, then when the remaining water scattered is incorporated as cold water or ice chips.
b. Hydroxypropyl cellulose – Is a not too bad gelling administrator if 15% or a more noteworthy measure of a characteristic dissolvable is relied upon to separate the dynamic solution.
c. Hydroxypropylmethyl cellulose – Is a better than average gelling administrator for time?released definitions.
d. Carboxy methyl cellulose – It is used as a piece of centralizations of 4 to 6% of medium thickness to convey gel. It is conflicting with alcohol. Glycerine can be added now and again to abstain from drying.
? Carbomers: Carbomer is a non particular name for a gathering of polymers known as Carbopol®. Carbopols® were at first used as a part of the mid 1950s. As a social event, they are dry powders, which have high mass densities and structure acidic watery courses of action (pH around 3.0). They thicken at higher pHs (around 5 or 6). They will similarly swell in liquid game plan of that pH to as much as 1000 times their extraordinary volume. Their answers range in thickness from 0 to 80,000 centipoise (cps).
? Poloxamers (Pluronics®): Poloxamers are copolymers of polyoxyethylene and polyoxypropylene. They will shape thermoreversible gels in center reaching out from 15% to half. This infers they are liquids at cool (cooler) temperature; however are gels at room or body temperature. Poloxamer copolymers are white, waxy granules that casing clear liquids when scattered in cold water or cooled to 0-10°C overnight.
Pluronic® F-127 is habitually united with a lecithin and isopropyl palmitate answer for make what is known as a "PLO gel."
4. Permeation enhancers [37-40]: These specialists cross into and cooperate with skin constituents and instigate a transitory and reversible increment in skin penetrability by upsetting and fluidizing the lipid boundary of the skin. Enhancers can expand the medication diffusivity through skin proteins. E.g. Oleic corrosive, lecithin, isoporopyl myristate, urea, linoleic corrosive, menthol, chinopodium oil, eucalyptus oil, dimethyl sulfoxide and so forth, are usually utilized as permeation enhancers.
Permeation enhancers act by one or a greater amount of the three mechanisms [41]:
a. Disrupting exceedingly requested structure of lipids of stratum corneum.
b. Interacting with intercellular protein.
c. Improvement of apportioning of medication from the dissolvable into the stratum corneum.
5. Preservtives: Preservatives like propyl and methyl paraben, Benzalkonium chloride, benzyl liquor, benzoic corrosive and so on., are usually utilized.
6. Antioxidants: Butylated hydroxyl anisole, Butylated hydroxyl toluene, Ascorbyl palmitate and so forth, are utilized as cell reinforcements to shield the formulation from experiencing oxidation.
7. Humectants: Glycerin, propylene glycol and so on are utilized as humectants.
Preparation of Emulgels
The methodology for arranging of emulgels incorporate three stages [42,43]:
? Step 1: Formulation of O/W or W/O kind of emulsion: Oil time of the emulsion was set up by dissolving emulsifier e.g. cross 20 in oil vehicle like liquid paraffin while the watery stage is set up by dissolving hydrophilic emulsifier like tween 20 in refined water. Added substances like methyl paraben and priopyl paraben are separated in humectant like propylene glycol. The medicine was separated in watery dissolvable like ethanol. Both the plans of solution and added substances are mixed with watery stage with consistent blending. Both the smooth and liquid stages were freely warmed to 70°C then the smooth stage was added to watery stage with constant blending. This mix was cooled to room temperature to shape an emulsion.
? Step 2: Formulation of gel base: The gel stage is set up by dissolving the polymer in the separated water with enduring mixing at moderate pace using mechanical shaker and the pH was adjusted to 6-6.5 using triethanolamine.
? Step 3: Incorporation of emulsion into gel base with steady blending: the gel stage is mixed into the emulsion stage in the extent of 1:1 to procure emulgel.
Evaluation Parameters
The following parameters are generally evaluated for the prepared emulgel formulations [44-48]
1. Physical appearance: For choosing the physical appearance the organized Emulgel arrangements are to be apparently examined for their shading, homogeneity, consistency and pH. The 1% liquid courses of action of the organized emulgels are ordinarily taken for measuring the pH by using a pH meter.
2. Rheological Studies: The consistency of the organized emulgel arrangements is generally chosen using a cone and plate viscometer with shaft 52 or 7 which is connected with a thermostatically controlled streaming water shower kept up at 25°C. The arrangement whose thickness was to be determined was taken into a holder secured with thermostatic coat. In the blink of an eye the Spindle was allowed to move uninhibitedly into the emulgel definition and the examining demonstrated was noted.
3. Spreadability [49]: Spreadability is controlled by the gadget prescribed by Mutimer et al. By this technique, spreading coefficient can be measured on the reason of "Slip" and "Drag" characteristics of emulgels. The gadget involves a wooden square, which is given by a pulley toward one side. An excess of emulgel (around 2 gm) under study must be determined to the ground slide which is starting now changed to the wooden piece. The emulgel is then sandwiched between this ground slide and another glass slide having the same estimations as that of modified ground slide. The second glass slide is generally outfitted with a catch. A known weight (500 mg, 1 gm or up to 1 Kg) is determined to the most noteworthy purpose of the two slides for 5 minutes to expel air and this give a uniform film of the emulgel between the slides. If any wealth of the emulgel is accessible at the edges of the slides it is to be scrapped off. Measured measure of weight was placed in the compartment associated with the pulley with the help of catch. The time (in seconds) required by the top slide to cover a division of 5 or 7.5 cm must be noted. Less time taken shows better spreadability. Spreading coefficient can be figured by using the formula,
S=(M.L)/T
Where, S: Spreadability or spreading coefficient,
M: Weight fixing to upper slide,
L: Length of glass slides
T: Time taken for the complete partition of the slides from each other.
4. Extrudability study [50]: It is a standard observational test done to gage the force required to remove the gel from the tube. The method is associated with choose the shear associated in the area of the rheogram contrasting with a shear rate surpassing the yield regard and demonstrating ensuing connection stream. In the examination works the system grasped for surveying emulgel enumerating for extrudability is all things considered based upon the sum in rate of emulgel i.e., removed from lacquered aluminum collapsible tube on use of weight in grams required to oust no under 0.5 cm piece of emulgel in 10 seconds. More sum is removed from the tube better is the extrudability. The estimation of extrudability of emulgel definition must be done in triplicate and the typical qualities are to be presented. The extrudability is then figured by taking after equation:
Extrudability=Applied weight to expel emulgel from tube (in gm)/Area (in cm2)
5. Globule size and its transport in emulgel: A gadget called Malvern zeta sizer is used to choose globule size and course. The system incorporates dissolving a 1.0 gm test of emulgel game plan in refined water and inciting vigorouly to get homogeneous disseminating. The resulting diffusing of test is to be mixed into the photocell of zeta sizer.
6. Swelling Index [51]: To choose the swelling record of orchestrated emulgel, 1 gm of gel is handled porous aluminum foil and a short time later set freely in a 50 ml holder containing 10 ml of 0.1 N NaOH. The samples are removed from measuring utensil at different time intervals and put on dry spot for a long time. After some time it is to be reweighed. Swelling file is given by the recipe:
Swelling Index (SW) %=[(Wt – Wo)/Wo] × 100.
Where, (SW) %=Equilibrium percent swelling,
Wo=Original weight of emulgel at zero time
Wt=Weight of swollen emulgel after time t.
7. Ex-vivo Bioadhesive quality estimation of topical emulgel (MICE SHAVEN SKIN) [52]: A balanced strategy is to be used for the estimation of bioadhesive quality. The mechanical get together contains two arm adjustment. Fresh skin is cut into pieces and washed with 0.1 N NaOH. Two bits of skin are joined to the two glass slide autonomously from that one glass slide is settled on the wooden piece and other piece is tied with the arm on right hand side. Weight is proceeded with the left hand side compartment. The benefit and left skillet are balanced by including extra weight the left hand compartment. The adjustment is to be kept in this position for 5 min. 1 g of emulgel was decisively weighed and put between these two slides containing exposed new mice skin pieces, and extra weight from the left skillet was cleared to sandwich the two bits of glass and some weight is associated with remove the closeness of air. The equality was kept in this position for 5 min. Weight is incorporated step by step at 200 mg/min to the other side hand dish until the two glass slides got isolated from each other. The weight (gram power) required to separate the emulgel from the glass surface gives the measure of bioadhesive quality. The bioadhesive quality is figured by using the mathematical statement:
Bioadhesive Strength=Weight required (in gms)/Area (cm2)
8. Drug content determination [53]: Drug obsession in the organized emulgel is measured by using UVspectrophotometer. Separate known measure of emulgel in dissolvable (methanol) by Sonication method. Reasonable weakenings are to be made to choose the absorbance of each in UV/VIS spectrophotometer.
9. In Vitro release study [54]: The in vitro drug release studies were done using a changed Franz scattering (FD) cell. The emulgel itemizing was associated on dialysis film which was supported amidst supplier and receptor compartment of the FD cell. Phosphate pad of reasonable pH can be used as a breaking down media. The receptor chamber was stacked with the deterioration media. The temperature of the cell was kept up at 37°C (taking after body temperature) by coursing water coat. This whole social affair is proceeded with an appealing stirrer and the plan was blended reliably using an alluring spot. A similar clear set was keep running in the meantime as a control. Tests (as a general rule 5 ml) are pulled back at appropriate time breaks and supplanted with proportionate measures of fresh crumbling media. Tests were destitute down spectrophotometrically at reasonable wavelength after true blue weakenings and the consolidated % drug release is determined as a part of time. The differentiation between the readings of prescription release and control was used as the honest to goodness scrutinizing as a part of each case.
Drug release kinetic study: To break down the component of medication discharge from the topical gel, the discharge information ought to be fitted to taking after mathematical statements
Zero – order equation: Q=K0 t
Where Q is the amount of drug released at time t, and K0 is the zero – order release rate constant
First- order rate equation: ln (100 – Q)=ln 100 – K1 t
Where Q is the percent of drug released at time t, and K1 is the first order release rate constant
Higuchi's mathematical statement: Q=K2√t
Where Q is the percent of drug release at time t, and K2 is the diffusion rate consistent.
10. Microbiological measure: Ditch plate framework can be used. It is a strategy used for the appraisal of bacteriostatic or fungistatic development of a compound. It is generally associated for semisolid definitions. Officially masterminded Sabouraud's agar dried plates are used. Three grams of the orchestrated emulgel is set in a trench cut in the plate. Normally organized society circles are streaked over the agar at a right edge from the trench to the edge of the plate. Subsequent to bring forth for 18 to 24 hours at 25°C, the parasitic improvement is viewed. In the blink of an eye the rate impediment is measured as takes after.
% inhibition=L2/L1 × 100
Where; L1=complete length of the streaked culture, and
L2=length of inhibition
11. Skin Irritation Test (Patch Test) [55]: The emulgel is connected on the appropriately shaven skin of rodent and its antagonistic impact like change in shading, change in skin morphology ought to be looked up to 24 hours. The aggregate arrangement of 8 rats can be utilized for the study. In the event that no bothering happens it shows that the test is passed. On the off chance that the skin disturbance manifestation happens in more than 2 rats the test ought to be rehashed.
12. Accelerated stability studies of Emulgel: Stability studies are performed by guidelines. The organized emulgels were full in aluminum collapsible tubes (5 g) and subjected to strength learns at 5°C, 25°C/60% RH, 30°C/65% RH, and 40°C/75% RH and 60 ± 2° for a period of 3 months. Tests were pulled back at 15-day time between times and surveyed for physical appearance, pH, rheological properties and pharmaceutical substance.
13. Marketed Formulations: Several marketed formulations [1,56] of emulgels are already available. And many more new formulations are being emerging in the market and are gaining acceptability at an extravagant rate owing to yheir better absorption, efficient release of drug and many advantages. Some of the marketed emulgels are mentioned below (Table 4).
| S.No | Drug | Marketed product | Manufacturer |
|---|---|---|---|
| 1. | Diclofenac-diethyl- ammonium | Voltarenemulgel | Novartis pharma |
| 2. | Miconazole nitrate, Hydrocortisone | Miconaz-H-emulgel | Medical union pharmaceuticals |
| 3. | Clindamycin,Adapalene | Excex gel | Zee Laboratories |
| 4. | Benzoyl peroxide | Pernox gel | Cosme Remedies Ltd. |
| 5. | Metronidazole, Clindamycin | Lupigyl gel | LupinPharma |
| 6. | Clindamycin phosphate, Allantion | Clinagel | StiefelPharma |
| 7. | Clobetasol propionate | Topinate gel | SystopicPharma |
| 8. | Kojic acid, DipalmitateArbuti | Kojivit gel | Micro Gratia Pharma |
| 9. | Aceclofenac | Acent gel | Intra Labs India Pvt. Ltd. |
| 10. | Azithromycin | Avindo gel | CosmePharma Lab. |
| 11. | Clotrimazole, Beclomethasone | Cloben gel | Indoco Remedies |
| 12. | Nadifloxacin | Nadicin cream | Psycho remedies |
| 13. | Tezarotene | Zorotene gel | Elder Pharmaceuticals |
| 14. | Hibiscus, liqourice and natural extracts | Levorag® emulgel | THD Ltd. |
| 15. | Diclofenac diethyl amine | Diclobaremulgel | Barakatpharma |
| 16. | Diclofenac sodium | Pennsaid | Nuvopharma |
Table 4: Marketed formulations of Emulgels.
14. Future prospects: Many polymers are coming into light step by step. These novel polymers are playing an imperative and fabulous part in the definition of different novel medication conveyance frameworks like emulgels. In the late years the usage of gelling pros is being developed record of their gigantic inclinations and flexibility in their use. Emulgel is the late framework for the movement of hydrophobic solutions and obviously it is a better than average technique for medicine transport of blend of both hydrophilic and hydrophobic meds. Emulsion based gel gives an appropriate medium to movement of such hydrophobic prescriptions where such solutions can be combined into its smooth stage and passed on to skin. In the coming years the topical prescription movement will be used extensively to give better patient consistence. Since emulgel is helpful in enhancing Spreadibility, grasp, consistency and ejection, it will wind up being a surely understood movement structure for topical application in future. In future various polymers both of trademark and built beginning stage will come into nearness for their wide application in pharmaceuticals.