Keywords
Outcome; Pulmonary venous obstruction; Risk factors; Surgery; Total anomalous pulmonary venous connection
Abbreviations
ACC: Aortic Cross-Clamp; CPB: Cardiopulmonary Bypass; OP: Operation; PAH: Pulmonary Artery Hypertension; PV: Pulmonary Vein; PVO: Pulmonary Venous Obstruction; TAPVC: Total Anomalous Pulmonary Venous Connection
Introduction
Total anomalous pulmonary venous connection (TAPVC) is a rare congenital heart disease that occurs in only 1% to 3% of children with congenital cardiac anomaly [1,2]. After birth, these patients usually develop progressive pulmonary congestion and heart failure sign due to pulmonary venous return flow obstruction and inadequate cardiac output [3]. Surgical correction is needed after correct diagnosis. Without surgical correction, the prognosis is extremely poor, with a mortality of 80% during infancy [3,4]. In several previous studies, there were many different risk factors for the mortality of TAPVC patients. In light of the variability in risk factors, retrospective evaluation of all patients undergoing repair at National Cheng Kung University Hospital from 1990 to 2015 was done. The purpose of our study was to identify the possible risk factors associated with increased mortality and morbidity.
Materials and Methods
Patients
All patients with a diagnosis of TAPVC were identified from the database of National Cheng Kung University Hospital during 1990 to 2015. Only cases of isolated TAPVC were enrolled. Those with minor associated defects such as atrial septal defect and/or a patent ductus arteriosus were included. However, patients with important concomitant cardiac defects, including single ventricle, atrial isomerism, transposition of the great vessels, and/or hypoplastic left ventricle, were excluded from our study. Only patients who underwent surgical repair and follow-up at our hospital were included. The diagnosis was made by echocardiogram, cardiac catheterization or both. The echocardiographic or catheterization values were extracted from diagnostic reports.
PVO was considered present if Doppler examination measured a pressure gradient along anywhere the vessel of pulmonary venous flow >5 mmHg. Surgical mortality was defined as death within 30 days of surgery or during primary hospitalization. Reintervention was defined as any operation performed secondary to recurrent pulmonary venous obstruction. Demographic and morphological characteristics at initial presentation are shown in Table 1. Data were abstracted by the review of clinical records and diagnostic reports obtained from the time of the initial admission to the last available follow-up. Median follow-up time was 3.38 years (minimum, 1 days; maximum 20 years) from repair, with 15 having >1 year of follow-up, and was available for 18 survivors.
| Variable |
Value |
| Age at surgery, median (minimum, maximum) |
36 d (1 d, 6y1 1m/o) |
| Gender |
12 (35.3%) |
| Female |
22 (64.7%) |
| Male |
|
| Body weight at surgery, mean ± SD (kg) |
4.19 ± 3.12 |
| median (kg) |
3.465 |
| TAPVC type, case number (%) |
| Supracardiac |
14 (41.2%) |
| Coronary sinus |
14 (41.2%) |
| Infracardiac |
5 (14.7%) |
| Mixed |
1 (2.9%) |
| Pulmonary venous obstruction, case number (%) |
| Supracardiac |
10 (71.4%) |
| Coronary sinus |
7 (50%) |
| Infracardiac |
3 (60%) |
| Mixed |
1 (100%) |
Table 1: Demographic and morphological characteristics of patients.
Potential variables associated with mortality were examined. These included age and weight at time of the initial repair, gender, low body weight for age, anatomic subtype of TAPVC, preoperative evidence of obstruction, need for additional surgical procedures, cardiopulmonary bypass (CPB) times, aortic cross-clamp (ACC) times, hypothermia times, preoperation( OP) PVO, pre-OP ventilator FiO2, post-OP ventilator FiO2, pre-operational inotropic use, post-OP epinephrine use, post-OP restenosis, pre-OP blood gas acidosis, postoperational hospitalization days and ICU days.
Statistical analysis
Chi-square and Fisher’s exact tests of association were conducted to differences of categorical variables. Continuous variables for t test that were distributed normally are presented as means (Figure 1). We constructed Kaplan Meier curves for post-operative duration time for pulmonary vein (PV) restenosis (Figure 2), pulmonary vein reintervention (Figure 3) and overall survival rate (Figure 4). Differences across categories were assessed using the log-rank test. Significance for two-sided analysis was set at P<0.05. Statistical analyses were performed using SAS software, version 9.3 (SAS Institute Inc., Cary, NC, USA).
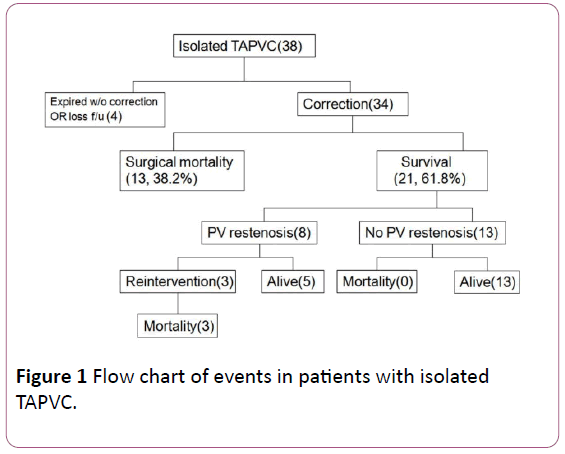
Figure 1: Flow chart of events in patients with isolated TAPVC.
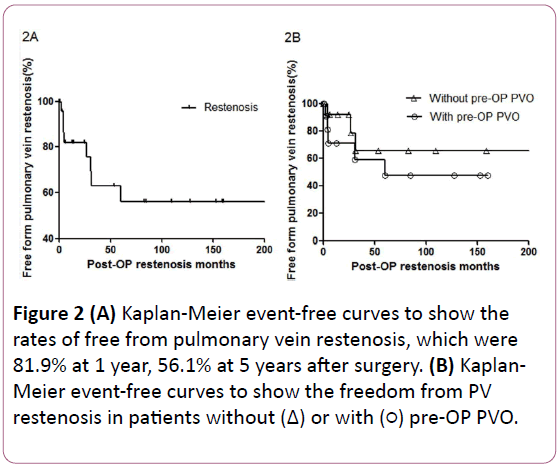
Figure 2:(A) Kaplan-Meier event-free curves to show the rates of free from pulmonary vein restenosis, which were 81.9% at 1 year, 56.1% at 5 years after surgery. (B) Kaplan- Meier event-free curves to show the freedom from PV restenosis in patients without (Δ) or with pre-OP PVO.
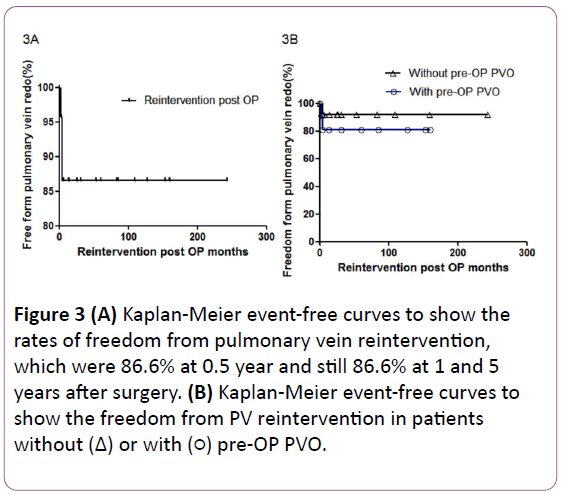
Figure 3:(A) Kaplan-Meier event-free curves to show the rates of freedom from pulmonary vein reintervention, which were 86.6% at 0.5 year and still 86.6% at 1 and 5 years after surgery. (B) Kaplan-Meier event-free curves to show the freedom from PV reintervention in patients without (Δ) or with pre-OP PVO.
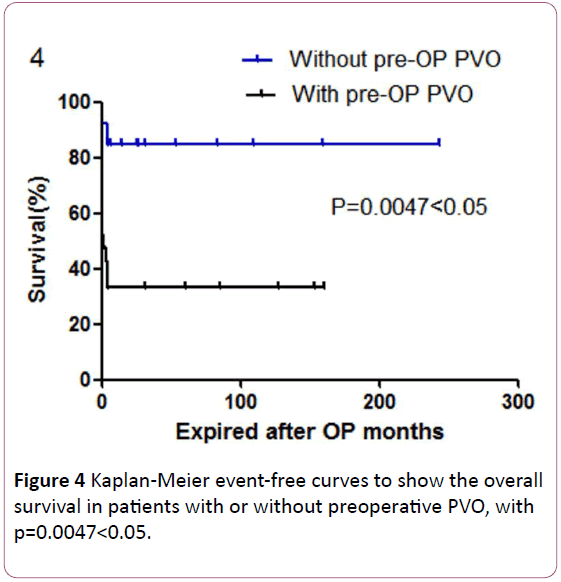
Figure 4: Kaplan-Meier event-free curves to show the overall survival in patients with or without preoperative PVO, with p=0.0047<0.05.
Results
A total of 44 patients were diagnosed with TAPVC. Excluding those associated with other major cardiac anomalies and no operation or follow-up in our hospital, 34 patients with isolated TAPVC were enrolled into our study. Of these 34 patients, 22 were male (64.7%); 12 were female (35.3%). Median age at time of surgical repair was 36 days (range, 1 to 2520 days) with median weight 3.465 kg (range, 1.4 to 20.7 kg). As shown in Table 1, 14 patients was supracardiac type (14/34, 41.2%), 14 patients presented with coronary sinus type (14/34, 41.2%), 5 patients presented with infracardiac type (5/34, 14.7%), and 1 patient presented with mixed TAPVC (1/34, 2.9%).
The mixed type was a combination of supracardiac and infracardiac types. At the time of presentation, PVO was present in 61.8% (21/34) of patients. Excluding mixed type (due to only one case), the supracardiac type subgroup had highest rate of PVO (10/14, 71.4%), the infracardiac type had second highest rate of PVO (3/5, 60%) and lowest rate in the coronary sinus type (7/14, 50%). Among the 10 patients with PVO in supracardiac type TAPVC, eight had obstruction of the ascending vertical vein and two had obstruction of the left innominate vein. Of the 3 patients with PVO in infracardiac type TAPVC, two had obstruction of the vertical vein and one had obstruction in the intrahepatic vein. Among the seven patients with PVO in coronary sinus type, three had obstructions in ASD; four had obstructions in the vessel connecting the common pulmonary vein to the coronary sinus.
Figure 1 shows the clinical results of the patient seen at our hospital. Of the 38 patients, 4 patients were excluded due to no operation in our hospital or loss to follow-up. Among the remaining 34 patients, 13 patients had surgical mortality after the surgical repair or during the first hospitalization. The overall surgical mortality was 38.2%. There were three patients (14.3% of surgical survivors) who required surgical reintervention for pulmonary venous obstruction after primary repair. However, these patients had surgical reintervention for PVO but all expired after surgery. The survival rate after first surgery was 61.8%. The overall survival rate during follow-up in our study was 52.9%.
Preoperative pulmonary venous obstruction was noted to be most associated with mortality. The mortality rate was 66.7% in the group with preoperative PVO but 15.3% in the group without preoperative PVO. This finding was statistically significant (p=0.0052, Table 2). The surgical mortality was 45.5% (5/11) before 2000 and 21.7% (5/23) after 2000, but no statistically significant difference was noted(p=0.2318).
| |
Alive |
Expired |
P value |
| Number of patients |
18 |
16 |
|
| Male/Female |
13/5 |
9/7 |
0.4754 |
| Age (days), mean |
209 ± 581.88 |
37.63 ± 42 |
0.2699 |
| Body weight <3% of age, numbers |
14 |
4 |
0.0051* |
| Pre-OP PVO, numbers |
7 |
14 |
0.0052* |
| Pre-OP blood gas acidosis, numbers |
4 |
6 |
0.4569 |
| Pre-OP ventilator FiO2 (%), mean |
41.22 ± 20.15 |
77.5 ± 28.6 |
0.0057* |
| Pre-OP inotropics use, numbers |
7 |
12 |
0.0454* |
| Post-OP ventilator FiO2 (%), mean |
51.39 ± 25.43 |
77.86 ± 23.26 |
0.005* |
| Post-OP epinephrine use, number |
5 |
13 |
0.0026* |
| Post-OP restenosis |
5 |
3 |
0.0421* |
| CPB times (mins), mean |
153 ± 52.26 |
158.46 ± 71.24) |
0.8173 |
| ACC times (mins), mean |
75 ± 28.58 |
70.3 ± 21.63 |
0.6602 |
| Hypothermia times (mins), mean |
111.39 ± 40.36 |
111.43 ± 50.21 |
0.998 |
| Post-OP ICU days, mean |
9.94 ± 5.61 |
14.87 ± 23.68 |
0.3985 |
| post-OP hospitalization days, mean |
16 ± 7.85 |
15.87 ± 23.59 |
0.9821 |
| OP: operation, CPB: cardiopulmonary bypass, ACC: aortic cross clamp, * P<0.05 |
Table 2: Characteristics of patients who underwent TAPVC correction.
Simple Cox regression revealed that the preoperative PVO was a significant predictor for the mortality of TAPVC patients (p=0.02, Table 3). The overall survival in patients of TAPVC patient without or with preoperative PVO is shown by Kaplan- Meier event-free curves (Figure 4). Patients with preoperative PVO had significantly higher mortality than patients without preoperative PVO (p=0.0047<0.05, Figure 4).
| Parameters |
HR |
95% CI |
P value |
| Age |
0.99 |
0.98 |
1.00 |
0.21 |
| Sex |
1.77 |
0.66 |
4.77 |
0.26 |
| Reintervention post OP days |
1.01 |
1.00 |
1.02 |
0.30 |
| Pre-OP ventilation days |
0.98 |
0.85 |
1.13 |
0.79 |
| Pre-OP ventilation setting (FiO2) |
1.03 |
1.01 |
1.05 |
0.01* |
| Pre-OP inotropics use |
0.26 |
0.07 |
0.92 |
0.04* |
| Post-OP ventilation days |
1.02 |
0.99 |
1.04 |
0.19 |
| Post-OP ventilation setting (FiO2) |
1.03 |
1.01 |
1.06 |
0.005* |
| Post-OP epinephrine |
0.12 |
0.03 |
0.54 |
0.01* |
| Bypass time |
1.00 |
0.99 |
1.01 |
0.91 |
| Hypothermia time |
1.00 |
0.99 |
1.01 |
0.77 |
| Aortic clamp time |
1.00 |
0.97 |
1.02 |
0.76 |
| Initial gas |
0.87 |
0.02 |
36.05 |
0.94 |
| Initial SpO2 |
0.93 |
0.88 |
0.97 |
0.002* |
| Post-OP ICU days |
1.00 |
0.98 |
1.03 |
0.76 |
| Post-OP hosp. days |
0.99 |
0.94 |
1.03 |
0.50 |
| Pre-OP PVO |
0.16 |
0.04 |
0.72 |
0.02* |
| Post-OP Reintervention |
0.52 |
0.15 |
1.82 |
0.30 |
| PV Restenosis |
1.83 |
0.52 |
6.48 |
0.35 |
| BW percentile (<3%) |
1.19 |
0.44 |
3.21 |
0.73 |
| TAPVC type |
0.78 |
0.19 |
3.26 |
0.73 |
| Post-OP infection |
1.29 |
0.37 |
4.52 |
0.70 |
| Emergent repair |
0.22 |
0.08 |
0.62 |
0.004* |
| PAH |
0.36 |
0.08 |
1.58 |
0.17 |
| *: p<0.05 |
Table 3: Simple cox regression analysis, outcome: Mortality.
PV restenosis was identified in 8 patients (8/21, 38.1%) during follow-up. The freedom from pulmonary vein restenosis was calculated by using the Kaplan-Meier technique. As shown in Figure 2A, the freedom from PV restenosis were 81.9%, 56.1% at 1 year, 5 years after first TAPVC correction repair, respectively.
The rates of freedom from PV restenosis decreased rapidly within the five post-operative year and remained stable in the subsequent years. Among patients with PV restenosis, three had symptoms of significant heart failure and required PV surgical reintervention to relieve PV stenosis. These three patients’ pulmonary venous flow to LA pressure gradients were 6, 26 and 38.2 mmHg, respectively. The Figure 2B show higher rates of freedom from PV restenosis in patients without preoperative PVO, but no significant difference was identified statistically.
The freedom from PV reintervention is shown in Figure 3A. Mean number of days to reintervention was 112.3 days. These three patients’ PV reintervention procedures were performed within six months after the first surgery. However, these patients all died after PV reintervention.
More than half of the patients (19/34, 55.9%) initially presented with signs of heart failure. Sixteen patients need mechanical ventilation and 19 patients required inotropic support preoperatively. A case review revealed that patients who had obvious signs of heart failure and ventilation or inotropic agent support had a higher mortality rate and more frequently needed emergent repair. After surgical correction, we also found patient who need higher ventilator O2 and epinephrine support also had higher mortality rate.
When including late mortality within the statistical analysis of investigated variables, we found several statistically significant correlations (Table 2). Besides preoperative pulmonary venous obstruction, these associated risk factors were as follows: pre-OP ventilator FiO2, pre-OP inotropics use, post-OP ventilator FiO2, post-OP epinephrine use, post-OP restenosis and emergent repair. Simple cox regression analysis also showed that these associated factors mentioned above were the significant predictor for the mortality of TAPVC patients (Tables 3 and 4).
| Parameters |
HR |
95% CI |
P value |
| Age |
0.98 |
0.95 |
1 |
0.09 |
| BW |
0.24 |
0.04 |
1.28 |
0.09 |
| Sex |
2.24 |
0.52 |
9.64 |
0.28 |
| Expired after OP days |
0.99 |
0.98 |
1 |
0.14 |
| Pre-OP ventilation days |
0.89 |
0.68 |
1.16 |
0.39 |
| Pre-OP ventilation setting |
1 |
0.97 |
1.04 |
0.8 |
| Pre-OP inotropics use |
0.82 |
0.21 |
3.31 |
0.78 |
| Post-OP ventilation days |
0.98 |
0.88 |
1.1 |
0.77 |
| Post-OP ventilation setting |
0.98 |
0.94 |
1.01 |
0.16 |
| Post-OP epinephrine |
1.09 |
0.26 |
4.63 |
0.91 |
| Bypass time |
1 |
0.99 |
1.01 |
1 |
| Hypothermia |
0.99 |
0.97 |
1.01 |
0.32 |
| Aortic cross clamp time |
1 |
0.97 |
1.03 |
0.79 |
| Initial SpO2 |
0.9 |
0.83 |
0.98 |
0.02* |
| Post-OP ICU days |
0.98 |
0.89 |
1.08 |
0.69 |
| Post-OP hosp. days |
1.01 |
0.95 |
1.07 |
0.8 |
| Pre-OP PVO |
0.51 |
0.12 |
2.16 |
0.36 |
| Reintervention post OP |
0 |
0 |
0 |
1 |
| Mortality |
0.04 |
0 |
0.41 |
0.01* |
| BW percentile (<3%) |
1.05 |
0.26 |
4.2 |
0.95 |
| TAPVC Type |
2.1 |
0.23 |
18.83 |
0.51 |
| Post-OP infection |
3.17 |
0.39 |
26.06 |
0.28 |
| Emergent |
0.72 |
0.14 |
3.56 |
0.68 |
| PAH |
1.95 |
0.43 |
8.82 |
0.39 |
| * P<0.05 |
Table 4: Simple cox regression analysis, outcome: restenosis.
Discussion
The natural course of TAPVC without surgical treatment is frequently progressive heart failure, which is associated with >80% mortality in the first year of life after surgery [3]. Surgical correction is needed after the diagnosis is confirmed. Improvement in surgical technique, early diagnosis and well post-operative patient care have led to improved results of TAPVC correction patient.
The surgical mortality of TAPVC patients decreased from 80% in early series to 5% in recent reports [5,6]. In our series, the surgical mortality decreased from 45.5% before 2000 to 21.7% after 2000. We collected the associated data of these cases to find the possible risk factors for mortality.
There are four types of TAPVC including supracardiac, coronary sinus, infracardiac and mixed type. In our series, supracardiac and coronary sinus type account for about 82% cases. Several studies have analyzed the relationships between mortality and type of TAPVC. In our studies, we find no association between the type of TAPVC and mortality (p=0.73, Table 3). But previous associated studies showed no consistent results [7]. The impact of connection type remains controversial [5-8].
Preoperative pulmonary vein obstruction was reported about 20% to 60% of TAPVC patient [1,2]. The preoperative PVO can be diagnosed by echocardiography with fair sensitivity and specificity [9]. In our series, preoperative PVO was present in 61.8% of patients. Supra-cardiac type had the highest rate (71.4%). Preoperative PVO was found to be associated with overall mortality (p =0.02<0.05, by simple Cox regression, Table 3).
Previous reports have revealed that preoperative PVO was also a risk factor for mortality and pulmonary vein reintervention [8,10]. However, not all studies revealed the relationship between PVO and overall mortality [1,2]. In our opinion, preoperative PVO is an important risk factor for overall mortality.
Younger age at repair was noted to be a risk factor for mortality of TAPVC patients in a previous study [1,11]. In early report, Wukasch et al. state that surgical treatment should be delayed until at least 6 months of age [12]. However, Lupinetti et al. found that operative treatment can be performed on TAPVC patients in infancy with low mortality [13]. We also noted that age at the time of surgery is not a risk factor for mortality of TAPVC patient (p=0.2699, Table 2). In our studies, better outcomes were found among patients who had early surgical correction. However, previous studies showed no consistent results [1,11,12].
In our series, we state that low body weight (<3% for a given age) is a protective factor (p=0.0051<0.05, Table 2). Low body weight at repair is not a risk factor for mortality, and such observation has also been described in other reports [4,14,15]. In our series, surgery was performed as soon as possible after diagnosis was confirmed, regardless of body weight and age.
Among the 34 TAPVC correction surgery in our hospital, 13 surgeries were performed under emergent conditions. An operation was classified as emergent if the patient was taken to the operating room within 24 hours after arriving at our hospital due to hemodynamic or ventilation compromise.
In our series, emergent surgery is a risk factor for mortality (p=0.004, Table 3). After arriving our hospital, we found most of these patients was unstable and need higher ventilator setting and inotropic agent support. Then, we recorded the associated data and analyzed them. We found higher ventilator FiO2 and pre-operative inotropic use were risk factors for mortality of TAPVC. We noted that lower initial SpO2 when arriving at our hospital was also a risk factor for mortality after TAPVC. We put these risk factors to simple Cox regression analysis and noted a significant correlation (p=0.002, Table 3). In previous studies, low cardiac output syndrome was also reported as the main causes of early postoperative death [14,16-18].
In conclusion, the patient who needed such support to compensate for heart failure had a higher mortality rate. So, the patient who had obvious heart failure condition before surgery could be the higher risk patient.
After surgical correction, these patients exhibited a wide range of clinical conditions. Some patient needed higher setting of ventilation but some didn’t. The patient who need epinephrine support had more critical condition. The use of epinephrine postoperatively is a risk factor for mortality [1]. In our series, we also found that use of epinephrine postoperatively is a risk factor for mortality of TPAVC patient (p=0.0026, Table 2). The patient also had higher FiO2 ventilator settings (p=0.005, Table 2). We put these two factors into simple Cox regression analysis and noted a significant correlation (p=0.01 and p=0.005, respectively, Table 3).
Pulmonary vein restenosis after repair of TAPVC is a significant risk factor for mortality in previous study [1,19-21]. In our series, there were eight patients with pulmonary vein restenosis after surgical correction. Among these eight patients, three patients need reintervention due to obvious heart failure.
In a previous study, patients who had restenosis later presented with significant heart failure and required reintervention within 6 months post-operatively [13]. These three patients of our study also had pulmonary vein restenosis episodes within six months, and all died after surgical reintervention.
In our study, post-operative pulmonary restenosis is also a significant risk factor for the mortality of TAPVC patients. We analyzed the associated factors by simple Cox regression. The results revealed that post-operative restenosis was also a significant risk factor for mortality (p=0.01, Table 4). However, we noted that initial SpO2 of patients when arriving our hospital is a protective factor. Higher SpO2 of patient initially had lower rates of post-operative stenosis (Table 4).
Study limitation
There were only 34 cases enrolled in our study from 1990 to 2015. Multivariate analysis was limited by small sample size and thus produces a clear limitation of the study power.
Conclusion
Although overall mortality decreased over time in isolated TAPVC patients, there were still a considerable number of mortality cases among this patient population. Overall mortality in our series was 47.1%. Preoperative pulmonary venous obstruction is an obvious risk factor. Besides, TAPVC correction surgery under emergent condition is also an important risk factor. A greater need for ventilator support and inotropic agent use before or after TAPVC correction are considered risk factors in our studies. Patients with obvious heart failure sign may be associated with the risk factors mentioned above. Pulmonary vein restenosis after repair of TAPVC also contribute to higher mortality rate. Low body weight and younger age at repair were not risk factors. However, low body weight was a protective factor. Surgical correction should be done as soon as possible regardless of body weight and age.
Acknowledgements
We wish to thank Shang-Chi Lee at the Biostatistics Consulting Center, National Cheng Kung University Hospital, for performing the statistical analysis.
Disclosure
The authors declare no conflicts of interest.
References
- Karamlou T, Gurofsky R, Al Sukhni E, Coles JG, Williams WG, et al. (2007) Factors associated with mortality and reoperation in 377 children with total anomalous pulmonary venous connection. Circulation 115: 1591e8.
- Kelle AM, Backer CL, Gossett JG, Kaushal S, Mavroudis C (2010) Total anomalous pulmonary venous connection: results of surgical repair of 100 patients at a single institution. J Thorac Cardiovasc Surg 139: 1387e94.
- Burroughs JT, Edwards JE (1960) Total anomalous pulmonary venous connection. Am Heart J 59: 913-931.
- Reardon MJ, Cooley DA, Kubrusly L, Ott DA, Johnson W, et al. (1985) Total anomalous pulmonary venous return: report of 201 patients treated surgically. Tex Heart Inst J 12: 131e41.
- Kirshbom PM, Myung RJ, Gaynor JW, Ittenbach RF, Paridon SM, et al. (2002) Preoperative pulmonary venous obstruction affects long-term outcome for survivors of total anomalous pulmonary venous connection repair. Ann Thorac Surg 74: 1616-1620.
- Michielon G, Di Donato RM, Pasquini L, Giannico S, Brancaccio G, et al. (2002) Total anomalous pulmonary venous connection: long-term appraisal with evolving technical solutions. Eur J Cardiothorac Surg 22: 184-191.
- Delius RE, deLeval MR, Elliott MJ, Stark J (1996) Mixed total pulmonary venous drainage: still a surgical challenge. J Thorac Cardiovasc Surg 112: 1581-1588.
- Husain SA, Maldonado E, Rasch D, Curzon C, Milachek J, et al. (2012) Total Anomalous Pulmonary Venous Connection: Factors associated with mortality and recurrent pulmonary venous obstruction Ann Thorac Surg 94: 825-832
- Wang JK, Lue HC, Wu MH, Young ML, Wu FF, et al (1993) Obstructed total anomalous pulmonary venous connection. Pediatr Cardio 14: 28-32.
- Bando K, Turrentine MW, Ensing GJ, Sun K, Sharp TG, et al. (1996) Surgical management of total anomalous pulmonary venous connection: Thirty-year trends. Circulation 94: 12-16.
- Karaci AR, Harmandar B, Aydemir NA, Sasmazel A, Balci A Y, et al. (2012) Early and intermediate term results for surgical correction of total anomalous pulmonary venous connection. J Card Surg 27: 376-380
- Wukasch DC, Deutsch M, Reul GJ, Hallman GI, Cooley DA (1975) Total anomalous pulmonary venous return: a review of 125 patients treated surgically. Ann Thorac Surg 19: 622- 633.
- Lupinetti FM, Kulik TJ, Beekman RH, Crowley DC, Bove EL (1993) Correction of total anomalous pulmonary venous connection in infancy. J Thorac Cardiovasc Surg 106(5): 880-885.
- Hancock Friesen CL, Zurakowski D, Thiagarajan RR, Forbess JM, del Nido PJ, et al. (2005) Total anomalous pulmonary venous connection: an analysis of current management strategies in a single institution. Ann Thorac Surg 79: 596-606.
- Bové T, François K, de Groote K, Suys B, De Wolf D, et al. (2004) Outcome analysis of major cardiac operations in low weight neonates. Ann Thorac Surg 78: 181e7.
- Lincoln CR, Rigby ML, Mercanti C, Al-Fagih M, Joseph MC, et al. (1988) Surgical risk factors in total anomalous pulmonary venous connection. Am J Cardiol 61: 608-611.
- Cobanoglu A, Menashe VD. (1993) Total anomalous pulmonary venous connection in neonates and young infants: repair in the current era. Ann Thorac Surg 55: 43-49.
- Serraf A, Bruniaux J, Lacour-Gayet F, Chambran P, Binet JP, et al. (1991) Obstructed total anomalous pulmonary venous return: towards neutralization of a major risk factor. J Thorac Cardiovasc Surg 101: 601-606.
- van de Wal HJ, Hamilton DI, Godman MJ, Harinck E, Lacquet LK, et al. (1992) Pulmonary venous obstruction following correction for total anomalous pulmonary venous drainage: A challenge. Eur J Cardiothorac Surg 6: 545-49.
- Ricci M, Elliott M, Cohen GA, Catalan G, Stark J, et al. (2003) Management of pulmonary venous obstruction after correction of TAPVC: risk factors for adverse outcome. Eur J Cardiothorac Surg 24: 28e36.
- Lacour-Gayet F (2006) Surgery for pulmonary venous obstruction after repair of total anomalous pulmonary venous return. Semin Thorac Cardiovasc Surg Pediatr Card Surg Annu 45-50.




