Case Report - (2019) Volume 3, Issue 1
Mouahamadou Nazirou Doddo Siddo1*, Bako Harouna1, Maman Sani Chaibou2, Habibou Harouna1, Hama Idrissa1, Ibrahim Arzika1, Ali Bonkano1, Moustapha Sanoussi1, Issoufou Coulidiaty1, Ibrahim Harou1, Altine Ramatou1 and Ali Ibrahim Toure3
1Department of Cardiology, Cardiology Center of National Hospital of Niamey Niger, Hospital in Niamey, Niger
2Department of Anesthesia and Resuscitation, Hospital of Niamey Niger, Hospital in Niamey, Niger
3Department of Cardiology, Center of Lamorde Hospital of Niamey, Niger
*Corresponding Author:
Mouahamadou Nazirou Doddo Siddo
Department of Cardiology
Cardiology Center of National Hospital of Niamey Niger
Hospital in Niamey, Niger
Tel: +22790659622
E-mail: nazdodo@yahoo.fr
Received date: February 04, 2019; Accepted date: February 27, 2019; Published date: March 09, 2019
Citation: Siddo MND, Bako H, Chaibou MS, Harouna H, Idrissa H, et al. (2019) Rhythmic Collapse and Significant Pulmonary Hypertension Indicative of Arrhythmogenic Dysplasia of Right Ventricle: A Case Report. Cardiovasc Investig. Vol.3 No.1:6
Keywords
Rhythmic collapse; Pulmonary hypertension; Right ventricular arrhythmogen dysplasia
Arrhythmogenic right ventricular dysplasia (ARVD) or Right ventricular arrhythmogenic cardiomyopathyis a genetically determined of cardiac muscle disease. It can lead to serious rhythmic and hemodynamic consequences in direct relation to the progressive adipose transformation of the most often right ventricular myocardium. We report a case of a 35-year-old woman hospitalized for rhythmic collapse. The electrocardiogram showed a rapid atrial fibrillation reduced by an external electric shock. The echocardiography showed an important dilatation of the right heart chambers and a significant pulmonary hypertension on tricuspid regurgitation. Thoracic angioscanner has eliminated a thromboembolic cause that could be the cause of pulmonary hypertension. Cardiac MRI diagnosed ADRV by confirming the dilatation of the right cavities and revealed thinning and fibro-fatty transformation of the RV walls. The evolution under treatment with bases of phosphodiesterase inhibitors, amiodarone was favorable with a follow-up of 12 months.
Introduction
Arrhythmogenic dysplasia of the right ventricle (ADRV) is a disease that selectively affects the right ventricle, in which cardiac muscle cells are gradually replaced by fat and fibrous tissue [1,2]. Some authors prefer the term of right ventricular arythomogenic cardiomyopathy because the etiology of the disease is not known [2-5]. It exposes the risk of haemodynamic instability and ventricular rhythm disorders [6] with sometimes sudden death [3] and the risk of progression to heart failure. Its frequency is estimated between 1 and 2 out of 10,000 in the general population [7,8]. The evolution of the disease can be schematized into four phases: asymptomatic, ventricular rhythmic, right heart failure and finally, biventricular disease. This evolution is difficult to predict over time and sudden death can occur at all stages, making the management of this disease difficult [9].
Observation
A 35-year-old woman who was seen for NYHA stage IV dyspnea associated with palpitations. Clinical examination revealed auscultatory tachyarrhythmia, arterial hypotension (BP at 70 mmHg), rapid peripheral pulsations, poorly perceived but symmetrically cold extremities.
The emergency electrocardiogram performed in the field of application (Figure 1) revealed a complete tachyarrhythmia with atrial fibrillation with an average ventricular rate of 135 cycles / min and a right axial deviation.
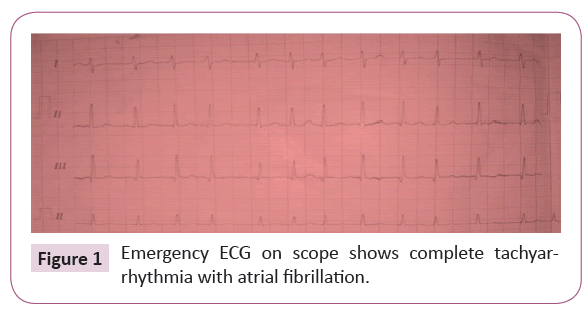
Figure 1: Emergency ECG on scope shows complete tachyarrhythmia with atrial fibrillation.
The clinical state and the electrical abnormalities evoke a rhythmic collapse, thus justifying an emergency electric shock of 200 joules allowing to slow the heart rate and to find a sinus rhythm and presenting a microvoltage and in the precordial straight line of the repolarization disorders of type Negative T waves beyond V3 and right full-length depolarization with QRS are extended to 120 ms (Figure 2).
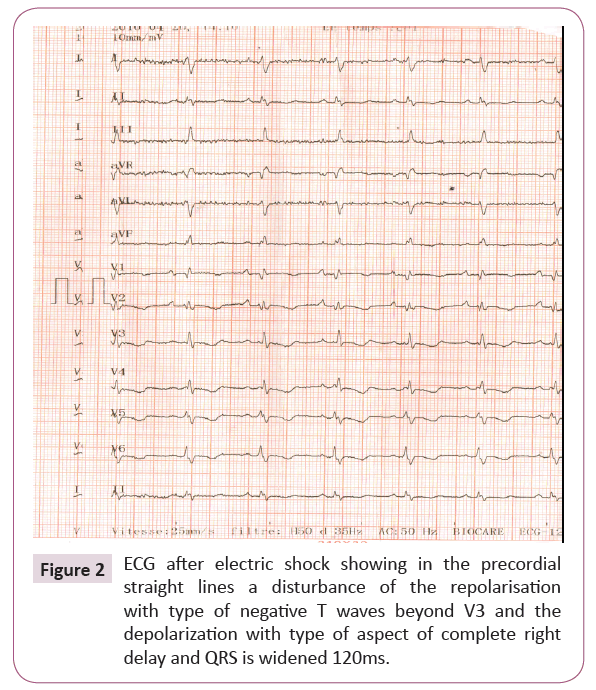
Figure 2: ECG after electric shock showing in the precordial straight lines a disturbance of the repolarisation with type of negative T waves beyond V3 and the depolarization with type of aspect of complete right delay and QRS is widened 120ms.
The chest x-ray revealed cardiomegaly with a high diaphragmatic point (Figure 3). Transthoracic echocardiography (Figure 4) showed significant dilatation of the right heart chambers, hypocontractile of right ventricle wall, and grade 3 tricuspid insufficiency with significant arterial hypertension at 65 mmHg.
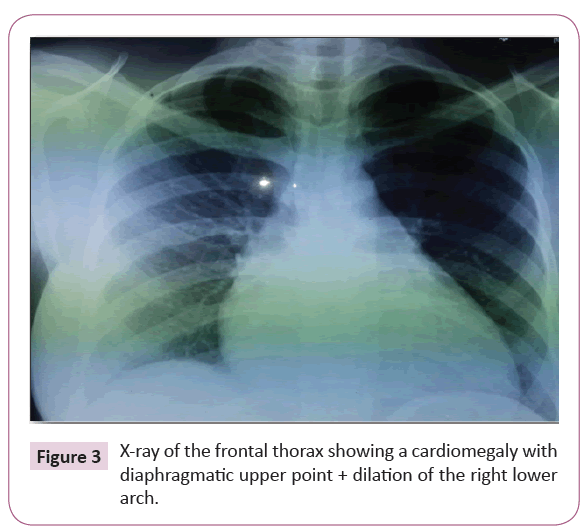
Figure 3: X-ray of the frontal thorax showing a cardiomegaly with diaphragmatic upper point + dilation of the right lower arch.
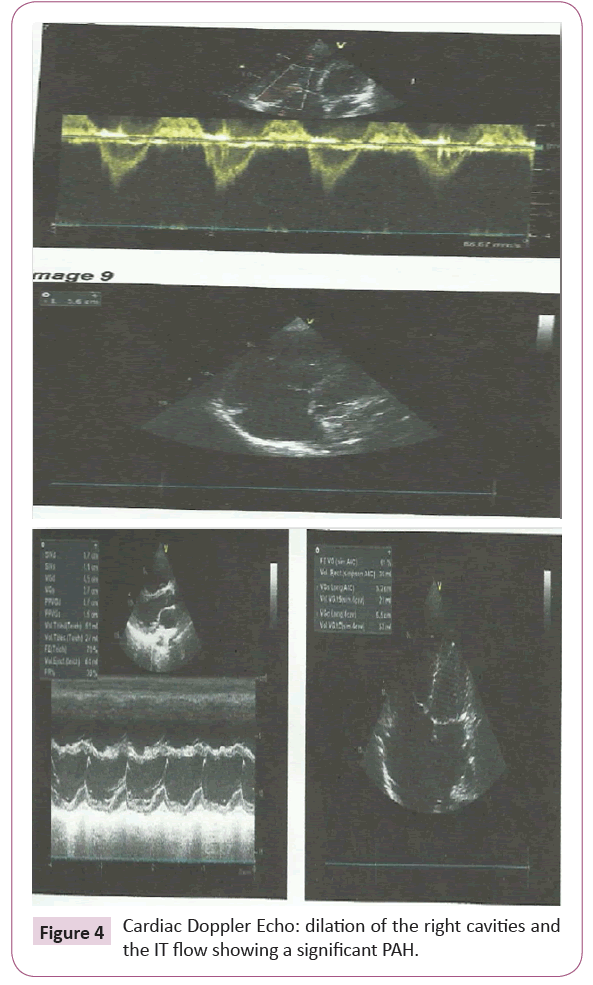
Figure 4: Cardiac Doppler Echo: dilation of the right cavities and the IT flow showing a significant PAH.
Echo-cardiac abnormalities suggested a secondary cause of arterial hypertension justifying the performance of a thoracic angioscan that eliminated a thromboembolic cause that could be the cause of pulmonary hypertension, thus justifying cardiac MRI (Figure 5) which poses the diagnosis of ADRV by confirming the dilatation of the right cavities and brings out a thinning and a fibro-adipose transformation of the walls of right ventricle. The family survey does not mention the concept of next death in the family.
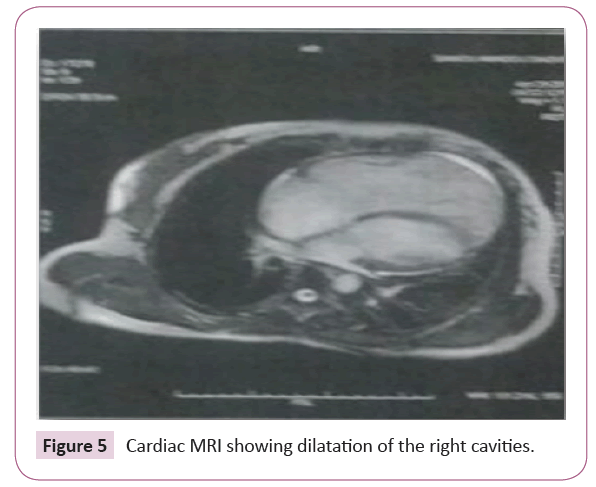
Figure 5: Cardiac MRI showing dilatation of the right cavities.
The evolution under treatment based on inhibitors of phosphodiesterase, amiodarone and long-term anti-coagulation was favorable with a decline of 12 months, waiting to discuss the insertion of an implantable defibrillator.
Comment
Arrhythmogenic right ventricle dysplasia is a progressive cardiomyopathy that mainly affects the right ventricle (RV). The condition is most often diagnosed in a young male subject [10,11]. In our case, it is a young lady of 35 years. In the French series of 130 cases, the average age is 31.8 ± 14 of which 77% are male [12]. The etiology of this condition is genetic. Indeed, a family history is found in 30 to 50% of patients with ADRV [10,13]. No family history of ADRV has been reported in our patient.
Electrocardiographic abnormalities are the first manifestation of the disease. These abnormalities are present in 90% of ADRV. These anomalies are represented by disturbances of repolarization and depolarization in the right precordial shunts (V1, V2, V3); inversion of the T waves (in the absence of a major straight branch block) and a right-sided delay often incomplete respectively. The upward slope of the S wave at V1, V2, V3 is decreased, sometimes crooked and prolonged by a series of socalled "epsilon wave" deflections (33% of cases) [7,11]. In our patient, we found a T wave inversion and a complete straight leg block.
Recently, emerging evidence has suggested the contribution of acute and chronic stress to the burden of arrhythmia. Xianlong Gao [14] revealed a pivotal role of JNK in the development of atrial arrhythmias in the aged heart, while cardiac CaMKIId, another pro-arrhythmic molecule, was also known to enhance atrial arrhythmogenicity. Jiajie Yan and coll [15] in their study had shown that the stress kinase c-jun N-terminal kinase (JNK) is critical in the pathogenesis of cardiac diseases associated with an increased incidence of atrial fibrillation (AF), the most common arrhythmia in the elderly. They recently discovered that JNK activation is linked to the loss of gap junction connexin43 (Cx43) and enhanced atrial arrhythmogenicity. However, direct evidence for JNK-mediated impairment of intercellular coupling (cell-cell communication) in the intact aged atrium is lacking, as is evidence for whether and how JNK suppresses Cx43 in the aged human atrium.
The structural abnormalities of RV are evaluated preferentially by echocardiography, right ventriculography and nuclear magnetic resonance (MRI). They include alterations in regional kinetics and dilatation of RV with anevrysmal areas.
Another study of Jiajie and coll [16], had mentioned that an excessive binge alcohol drinking has acute cardiac arrhythmogenic effects, including promotion of atrial fibrillation (AF), which underlies “Holiday Heart Syndrome.” The mechanism that couples binge alcohol abuse with AF susceptibility remains unclear.
Also they had identified that JNK2-driven CaMKII activation as a novel mode of kinase crosstalk and a causal factor in atrial arrhythmic remodeling, making JNK2 a compelling new therapeutic target for AF prevention and treatment [17].
In our case, we were guided by clinical and electrical signs including a complete atrial fibrillation tachyarrhythmia accompanied by arterial hypotension, Doppler echo showing right cavitary dilatation and pulmonary arterial hypertension and CT scan. Thoracic eliminating a secondary cause that may explain this pulmonary arterial hypertension it is the cardiac MRI which poses the diagnosis of ADRV by confirming the dilation of the right cavities and shows a thinning and a fibro-fatty transformation of the walls of the RV. In our patient, no genetic analysis has been performed but it is not imperative to make the diagnosis.
Sudden death can occur at any stage of the disease. Its incidence is estimated at 1-3% per year in the known ADRV [11] and it may be the first manifestation of the disease as described a series of Lyonnais systematic autopsies of sudden deaths on 1,930 autopsies between 1980 and 1999 of which 10.4% are ADRV and the mean age at death is 32.5 years for men and 34 years for women [18].
In half of the cases reported in the literature, the circumstances of sudden death are related to stress or stress. The risk factors for sudden death identified include: history of ventricular arrhythmias, syncope, young age, exercise, LV involvement, and lack of effective antiarrhythmic therapy [11]. It is therefore recommended to avoid any intense physical activity given its role in the induction of arrhythmias. The therapeutic management consists of an antiarrhythmic drug treatment and to treat cardiac insufficiency when it is present.
Antiarrhythmic drugs decrease the frequency and duration of arrhythmias, but no current data suggests reducing the risk of sudden death. The use of a single antiarrhythmic drug in the ADRV is not effective for the control and abolition of arrhythmias. Only sotalol and amiodarone are the recommended antiarrhythmic drugs in ADRV [19-21]. In our case, the cardiac rhythm disorder was reduced by an external electric shock of 200 joules, and then stabilized by amiodarone with a one-year follow-up, waiting to discuss the insertion of an implantable defibrillator.
Conclusion
Rhythmic disorders are a serious and unpredictable complication of arrhythmogenic dysplasia of the right ventricle related to adipose transformation of the heart muscle.
The diagnosis is often difficult and based on a bundle of clinical and especially para-clinical arguments. The importance of the diagnosis lies in the prevention of sudden death because a better knowledge of the pathology makes it possible to reduce the deaths by rhythm disorder, aggravating factor and bad prognosis during AD RV. This clinical case illustrates hemodynamic instability secondary to specific electrical disorders of arrhythmogenic dysplasia of the right ventricle. At present, only the implantation of an implantable defibrilator compared to an antiarrhythmic treatment has shown its effectiveness in the primary prevention of sudden death.