Review Article - (2023) Volume 9, Issue 4
Regulatory and Pathophysiological Roles of Reactive Oxygen Species in Skeletal Muscle
Anayt Ulla and
Takeshi Nikawa*
Department of Nutritional Physiology, Institute of Biomedical Sciences, Tokushima University Graduate School, Japan
*Correspondence:
Takeshi Nikawa,
Department of Nutritional Physiology, Institute of Biomedical Sciences, Tokushima University Graduate School,
Japan,
Email:
Received: 15-Aug-2023, Manuscript No. IPBMBJ-23-17318;
Editor assigned: 17-Aug-2023, Pre QC No. IPBMBJ-23-17318 (PQ);
Reviewed: 31-Aug-2023, QC No. IPBMBJ-23-17318;
Revised: 05-Aug-2023, Manuscript No. IPBMBJ-23-17318 (R);
Published:
12-Sep-2023, DOI: 10.36648/2471-8084-9.04.31
Abstract
Skeletal muscle is a complex organ in the body that has pivotal roles in movement, respiration, metabolism, and
other normal daily activities. Owing to its contractile activity, oxygen consumption and metabolic functions, oxidant
species, such as Reactive Oxygen Species (ROS) or Reactive Nitrogen Species (RNS) are continuously generated
in skeletal muscle. ROS/RNS have been reported to have dual functionality in skeletal muscle: They may induce
oxidative damage, tissue dysfunction, and myopathy, but they can also regulate various biochemical processes,
such as gene expression, calcium signaling, and contractility to confer beneficial effects on the organism. In skeletal
muscle, the precise action of ROS/RNS has been found to be linked to their concentration. At lower concentrations,
they generally act as regulatory molecules to enhance muscle force and muscle function; however, at higher concentrations,
they may cause oxidative damage leading to a decreased muscle performance and occurrence of atrophy.
The present review has shed light on the physiological and pathological roles of ROS/RNS in skeletal muscle
health and discussed their possible mechanisms of action.
Keywords
Skeletal muscle; Oxidative stress; Reactive oxygen species; Reactive nitrogen species; Proteolysis;
Mitochondrial dysfunction; Excitation-contraction coupling
Introduction
Skeletal muscle is a dynamic and plastic tissue in human body that contributes significantly to multiple bodily functions comprising mechanical and metabolic activities of the body. Mechanically, it maintains posture and supports health leading to functional independence; metabolically, it participates in basal energy metabolism, storage of amino acids and carbohydrates, thermogenesis, utilization of oxygen increased during physical activity and exercise etc [1]. Various factors including nutritional status, physical activity, age, exercise, and the activation of biomolecular pathways promoting function and metabolic activity of muscle are critical for muscle health [2]. Reactive oxygen species, commonly known as ROS, have long been considered entities harmful to skeletal muscle tissue. Research has determined the pathogenic role of ROS in inherited muscle health abnormalities and subsequently identified them a causative agent in multiple muscular diseases [3-5]. However, as the field of research has expanded, it has been proposed that the presence of ROS at physiological concentrations may play a beneficial role in physiology of skeletal muscle. ROS can regulate multiple processes in skeletal muscles such as transcription factor activity, ion transport, apoptosis, and metabolism, including various proteins critical for muscle cell function [6]. The major distinctions between physiological and pathological signaling exerted by ROS stem from their concentration, reactivity, and origin. At low concentrations, they increase muscle force and enhance adaptation to exercise, whereas at a high concentration, they lead to a decline in muscle performance [7]. During exercise, ROS promotes mitochondrial biogenesis via peroxisome Proliferator-activated receptor Gamma Coactivator- 1α (PGC-1α)-activated signal transduction pathway, but at higher concentrations, mitochondria and mitochondrial DNA (mtDNA) may be adversely affected by ROS, which impairs the myogenic differentiation process [8-10]. Therefore, the regulatory and pathological effects of ROS on skeletal muscle function are associated with the concentration of ROS targeting the muscle cells and the duration of exposure. Other factors that may impact the physio-pathological activities of muscle are the sources of ROS, their targets, reactivity of ROS, anti-oxidative capacity of cells, and the cells’ defense mechanisms [11]. Presently, ROS are known to trigger many signaling pathways related to skeletal muscle homeostasis and adaptation. Hence, the present review has been drafted to discuss the various roles of ROS on the physiological and pathological condition of skeletal muscle, along with their possible mechanisms.
GENERATION OF ROS IN SKELETAL MUSCLE
ROS are chemically reactive molecules containing an unpaired electron produced from the incomplete reduction of molecular oxygen. Reactive Nitrogen Species (RNS) are substances produced by the reaction of NO with compounds containing ROS. Most ROS are produced as by-products of the mitochondrial Electron Transport Chain (ETC) [12]. The leakage of electrons or the uncoupled transfer of an electron during its movement from complex I to complex III in the ETC leads to superoxide radicals formation (O2 •−) [13]. Furthermore, during muscle contraction, oxygen consumption is increased which is utilized in the ETC. Oxygen is the final acceptor of electron in ETC. After accepting electron, it turns reactive and undergoes reduction with the proton (H+) found in the matrix of mitochondria that ultimately forms water. Approximately 5% of O2 is reported to be converted to superoxide. In addition to this, superoxide can be produced from various locations within muscle fibers including the mitochondrion, sarcoplasmic reticulum, transverse tubules, sarcolemma, and the cytosol [14].
Although mitochondria are thought as the primary source of ROS, studies reported that Nicotinamide Adenine Dinucleotide Phosphate (NADPH) oxidases (NOXs), a multicomponent enzyme system located in the plasma membrane, sarcoplasmic reticulum, transverse tubules, and the sarcolemma also produce ROS [14]. NOXs catalyze the reduction of O2 to O2 •− by utilizing NADH or NADPH as electron donors. NOX2 and NOX4 are isoforms of NOX present in skeletal muscle and considered to be the major source of ROS in striated muscle [15]. Moreover, Xanthin Oxidase (XO), cytosolic in origin generates superoxide as a byproduct of oxidation of hypoxanthine to xanthine and uric acid in the cytosol of contracting skeletal muscle [16].
Phospholipase A2 (PLA2) also generates ROS. PLA2 acts on the cell membrane and releases arachidonic acid. The deoxygenation of arachidonic acid by lipoxygenase enzymes generates ROS [17]. Mechanistically, the activation of PLA2 can stimulate NOXs, which promotes ROS production in the mitochondria and cytosol of muscle and release ROS into the extracellular space [18,19]. Both the calcium-dependent and independent forms of PLA2 are present in skeletal muscle and participate to ROS generation in the muscle [6]. It is suggested that ROS activity under resting conditions is mediated via calcium-independent PLA2, whereas during inflammation, stress, and contractions, calcium-dependent PLA2 is activated to induce ROS production [19].
Superoxide molecules formed during the above processes act as a substrate for the generation of secondary ROS molecules. The dismutations of superoxide radical by Superoxide Dismutase (SOD) convert it to another non-radical ROS, namely hydrogen peroxide (H2O2). Hydroxyl radicals (•OH) are formed from H2O2 by Fenton reaction [20]. H2O2 is also converted to H2O by the enzymatic action of antioxidants catalase and Glutathione Peroxidase (GPx). Hence, in a nutshell, ROS/RNS include superoxide anions (O2 •−), hydrogen peroxide (H2O2), hydroxyl radicals (•OH), and the highly reactive peroxynitrite (ONOO−), which is formed from the reaction of nitric oxide (NO) and O2 •− (Table 1).
Table 1: ROS in skeletal muscle
| Species |
Producing site/enzyme |
| Superoxide (O2·-) |
Mitochondria, NADPH oxidase, Xanthin oxidase, PLA2 |
| Hydrogen peroxide (H2O2) |
Dismutation of superoxide by superoxide dismutase |
| Hydroxyl radicals (.OH) |
Produced from H2O2 by Fenton reaction |
| Peroxynitrite (ONOO-) |
Reaction of nitric oxide (NO) and O2·- |
ANTIOXIDANT DEFENSE IN SKELETAL MUSCLE
Antioxidants are the compounds that scavenge or neutralize the ROS and prevent the cellular damages induced by ROS. They are essential molecules to maintain the equilibrium between production of ROS and its neutralization i.e., redox homeostasis in the body [6]. There are two types of antioxidants exist: Enzymatic and non-enzymatic antioxidants.
Enzymatic Antioxidants
Primary enzymatic antioxidants include Superoxide Dismutase (SOD), catalase, and Glutathione-Peroxidase (GPx). Other secondary or ancillary enzymatic antioxidants that function to protect cells from oxidation are Peroxiredoxins (PRXs), Thioredoxin (TRX), and Glutaredoxins (GRX) etc. Superoxide Dismutase (SODs) dismutates superoxide radicals (O2 •−) and convert it to H2O2 and Oxygen (O2). They exist in three isoforms namely SOD1, SOD2 and SOD3 [21,22]. All these three isoforms of SOD contain the transition metal to their active sites to exert the dismutation reaction. SOD1 has Copper-Zinc as cofactor and found in the cytosol and mitochondrial intermembrane space. SOD2 contain manganese as a cofactor and situated in the mitochondrial matrix. SOD3 acquires copper-Zinc as cofactor and located in extracellular space [22]. In skeletal muscle, the maximum amount of SOD is found in cytosol (65%-85%) whereas (15%-35%) is present in the mitochondria of muscle [23]. Moreover, the activity of SODs in oxidative muscle fiber was found higher than glycolytic fibers [24,25]. The deficiency of SOD1 in skeletal muscle found to markedly increase oxidative stress whereas the contraction of myogenic fiber activates SOD1 and SOD2 [26]. Catalase, which incorporates iron as a cofactor, is ubiquitously distributed in the cells. It catalyzes the breakdown of H2O2 into H2O and O2 [27]. Like SOD, the activity of catalases is maximum in oxidative muscle fibre than the glycolytic muscle fibres [28]. It also shows lower affinity for H2O2 compared to GPx. Glutathione-Peroxidase (GPx) is a Se- dependent enzyme that catalyzes the reduction of H2O2 or organic hydroperoxide (ROOH) to water and alcohol (ROH) respectively [29]. It is found in both the cytosol and mitochondria of the cell. It shows maximum activity in type-I muscles fiber (slowtwitch muscle fiber) compared to fast-twitch muscle fibres [30]. Among secondary antioxidants, the TRX antioxidant system is situated in the cytosol and mitochondria and protects the proteins from being oxidized. It also prevents apoptosis and exerts protection against oxidative stress [31]. Thioredoxin reductase, exhibit antioxidant effect by reducing hydroperoxides [31]. GRX is also found in cytosol and mitochondria and participate in the protection and repair of protein and non-protein thiols under oxidative stress condition [32]. Lastly, PRX, a cysteine dependent peroxidase, located in skeletal muscle cells either in mitochondrion or in cytosol, peroxisome, and nuclei. It can reduce peroxides, hydroperoxides and peroxynitrite using electrons given by physiological thiols [33] (Table 2).
Table 2: Antioxidants in skeletal muscle (Enzymatic)
| Name |
Types |
Localization |
Functions |
| Superoxide Dismutase (SOD) |
SOD1 (Cu-Zn SOD) |
Cytosol, Mitochondria, Intermembrane space |
Superoxide neutralization |
| SOD2 (MnSOD) |
Mitochondrial matrix |
Superoxide neutralization |
| SOD3 (Cu-Zn SOD) |
Extracellular fluid |
Superoxide neutralization |
| Catalase |
|
Cytosol, Mitochondria, Peroxisome |
Breakdown of H2O2 into H2O and O2 |
| Glutathione Peroxidase (GPx) |
GPx1, GPx2, GPx3, GPx4, GPx5, GPx6, GPx7, GPx8 |
Cytosol, mitochondria, extracellular fluids, GIT, and kidney tissues |
Reduction of H2O2 or organic hydroperoxide (ROOH) to water and alcohol (ROH). |
| Secondary or ancillary enzymatic antioxidants |
| Peroxiredoxins (PRXs), |
PRDX1, PRDX2, PRDX3, PRDX4, PRDX5, PRDX6 |
Mitochondria, cytosol, Peroxisome, and nucleus |
Reduces peroxides, hydroperoxides and peroxynitrates |
| Thioredoxin (TRX) |
|
Cytosol, Mitochondria |
Prevent protein oxidation, apoptosis, and oxidative stress |
| Glutaredoxins (GRX) |
|
Cytosol, Mitochondria |
Protection and repair of protein and non-protein thiols under oxidative stress |
Non-Enzymatic Antioxidants
Non-enzymatic antioxidants include Glutathione (GSH), vitamin- C, vitamin-E, α-lipoic acid, uric acid, bilirubin, coenzyme Q10 (CoQ10), carotenoids, and polyphenols etc [34]. GSH is one of the most important non-enzymatic antioxidants in the muscle fibre. The availability of GSH in the cells corresponds to the extent of exposure of oxidants to that cell. Moreover, it was found that the concentration of GSH in slow-twitch muscle fiber (type-I) is 4-5-fold higher than fast twitch muscle fibre (type-II) [35]. Among various roles of GSH, it acts as a substrate for GPx to eradicate H2O2 and other organic hydroperoxide [36]. It is also associated with the reduction of antioxidants like vitamin-C and vitamin-E to maintain them in reduced state. The adaptation of skeletal muscle fiber to high intensity exercise in linked to improved GSH level [37]. Likewise, vitamin C and vitamin E also exert antioxidant effect. They play vital roles in protecting cellular membrane and plasma lipoprotein against lipid peroxidation, as they can scavenge ROS/RNS specially peroxyl radicals (ROO•) [38]. α lipoic acid is another non-enzymatic antioxidant. It is naturally found and distributed in various foods. It acts as a cofactor for α-dehydrogenase complex and associated with other cellular reactions. Various studies have suggested that α-lipoic acid can exert antioxidant effect by recycling vitamin-C [39]. Uric acid is a by-product of purine metabolism and a low molecular weight antioxidant [40]. It acts as an effective scavenger of peroxyl radical, hydroxyl radical, and singlet oxygen [41]. Bilirubin is the end-product of hemoprotein degradation. It shows antioxidant activity against peroxyl radicals and prevents cellular damage induced by H2O2 [42]. CoQ10 is a component of mitochondrial electron transport chain which functions as a non-enzymatic antioxidant by scavenging (ROO• radicals) and preventing lipid peroxidation [43]. Polyphenols (PPs) are naturally occurring organic compounds that are distributed in different plants, fruits, vegetables, nuts, seeds, flowers, tea, and beverages. They exert strong antioxidant activity by scavenging the free radicals and preventing the upregulation of oxidative stress-induced pathways [44] (Table 3).
Table 3: Antioxidants in skeletal muscle (non-enzymatic)
| Name |
Functions |
| Glutathione (GSH) |
Acts as a substrate of GPx to remove H2O2 and other organic hydroperoxide |
| Vitamin C and Vitamin E |
Scavenging of ROS/RNS specially peroxyl radicals (ROO·) |
| a-lipoic acid |
Acts as a cofactor for a-dehydrogenase complex |
| Uric acid |
Scavenging of peroxyl radical, hydroxyl radical, and singlet oxygen |
| Bilirubin |
Prevent cellular damage induced by H2O2 |
| Coenzyme Q10 |
Scavenging (ROO· radicals) and preventing lipid peroxidation |
| Polyphenols |
Scavenging (ROO· radicals) and preventing lipid peroxidation |
ROLE OF ROS AS REGULATORY MOLECULES
ROS act as signaling molecules to regulate various physiological activities. They are imperative to maintain oxidation-reduction homeostasis and correct functioning in the body [45]. They regulate signaling pathways for proper cell cycle and apoptosis and play vital roles in gene activation, cellular growth, and the modulation of chemical activities in the cells [46]. Skeletal muscle maintains redox equilibrium between ROS/RNS generation and antioxidant defense that is in constant equilibrium even after contraction [45]. At basal ROS level, they regulate protein phosphorylation, ion channels, transcription factors, immune defense, and muscle function [47]. ROS activate the PI3K-NFE2-like2 (Nrf2)-antioxidant response element to protect cells from oxidative damage and maintain the redox homeostasis [48]. Similarly, during exercise, numerous signaling pathways such as PGC-1α, 5′-AMP-Activated Protein Kinase (AMPK), mitogen-activated protein kinase, insulin-like growth factor-1 (IGF-1), and calcium are regulated by the physiological concentration of ROS [49]. Furthermore, ROS induce the phosphorylation of transcription factors and hence increase protein synthesis due to their kinase activation (e.g. ERK, JNK, and p38) and phosphatase deactivation [50]. ROS induced by H2O2 in C2C12 cells significantly enhanced IGF-I-induced phosphorylation of the IGF-I receptor (IGF-IR) which was attenuated by treatment of antioxidants, suggesting ROS are necessary for IGF-1 myocyte hypertrophy [51]. The pathways mentioned above are foundational for muscle adaptation owing to their modulation of mitochondrial biogenesis and function, anti-oxidative capacity, proliferation and differentiation of myoblast, and growth of muscle.
EXCITATION-CONTRACTION COUPLING
Excitation-Contraction (EC) coupling is a series of events that propagates the action potential through sarcolemma causing activation of Dihydropyridine Receptors (DHPRs) and Ryanodine Receptors (RyRs) to release calcium from sarcoplasmic reticulum into cytoplasm/sarcoplasm leading to force generation and contraction into sarcomere [52]. Dihydropyridine Receptors (DHPRs) are present in the t-tubules of sarcotubular system. The activation of DHPR activates Ryanodine Receptors (RyRs) closely located to t-tubules [52]. Mammalian cells express three types of RyRs: RyR1, RyR2, and RyR3. RyR1 is the dominant isoform in skeletal muscle [53]. The activation of RyR1 stimulates the release of calcium stored in sarcoplasmic reticulum into the cytoplasm/sarcoplasm. The released calcium then binds to the regulatory protein troponin and causes confirmational change to another protein tropomyosin to initiate the contraction, which is basically the interaction between actin and myosin [52]. This process requires ATP along with Ca2+ [54]. During relaxation, released calcium transported back to the sarcoplasmic reticulum through SR Ca2+ ATPase (SERCA) pumps. SERCA is an ATP-dependent Ca2+ pump located in the free sarcoplasmic reticulum [52]. Disturbance of calcium channels like RyRs and SERCA in the sarcoplasmic reticulum membrane can interrupt the Ca2+ regulation leading to uncoupling of excitation and contraction events [55]. The calcium (Ca2+) ion modulates various cellular functions such as contraction, secretion, metabolism, gene expression, cell survival etc. Therefore, proper Ca2+ handling in the muscle fiber is pivotal as calcium dysregulation may impair muscle force generation and with oxidative stress [56].
ROS IN EXCITATION-CONTRACTION COUPLING
The excitation-contraction coupling in skeletal muscle is modulated by ROS [57]. Depolarization of skeletal muscle upregulates NOX enzymes found in the t-tubules and sarcoplasmic reticulum of sarcotubular system. The activation of NOX generates ROS which modulates Ca2+ release by the RyR1 and regulate excitation-contraction coupling [58,59]. However, excessive increase of ROS can exert adverse effects to EC coupling. Oxidative stress-induced leaking of RyR1 Ca2+ and SERCA oxidation may induce muscle atrophy and muscle weakness as reported by Qaisar et al. [60,61]. Similarly, increased ROS (O2 •−, OH• and H2O2) due to high copper concentration impaired myocardial excitation-contraction coupling, decreased force generation capacity, Ca2+ release and reuptake and diminished myosin-ATPase activity [62].
High level of Ca2+ in cytosol can also promote mitochondrial ROS production and proteolysis by calpain activation [63]. Oxidative stress reduces the activity of SERCA pump as reported by Sharov et al. (2006) where aging-induced oxidative stress increased oxidation of cysteine residue of SERCA proteins [64]. In skeletal muscle, cysteine residues like Cys674 and Cys675 regulate the SERCA activity by reversible oxidation through peroxynitrite- induced glutathionylation; however, increased oxidative stress may cause irreversible oxidation of cysteines including sulfonylation leading to reduced SERCA activity [65,66]. Oxidative stress-induced mice lacking antioxidant enzyme CuZnSod (sod1-/-) showed reduced membrane excitability and RyR stability, decreased fiber Ca2+ sensitivity and suppressed SERCA activity via modification of the Cys674 residue, dysregulated SR and cytosolic Ca2+ homeostasis, and impaired mitochondrial Ca2+ buffering and respiration [67]. Thus, the above arguments suggest that physiological ROS regulate EC coupling however oxidative stress condition causes impairment of EC coupling.
ROS DURING MYOGENESIS
Myogenesis is an organized process that involves the activation of satellite cells into myoblasts followed by proliferation and differentiation to form myotubes [68]. It occurs both during neonatal growth and muscle injury. In adult skeletal muscle, tissue homeostasis is maintained via self-renewal of skeletal muscle satellite cells that compensate for the turnover of terminally differentiated cells [69]. Myogenic regulatory factors MyoD, Myf5, Myf6, myogenin, myocyte enhancer factors, and the serum response factor are the vital players for myogenesis regulation [70]. In the injured muscle signals such as Wnt signaling, JAK/STAT1/STAT3 Signaling, PI3K/AKT signaling etc. are stimulated which stimulate muscle satellite cells to migrate toward the injury site and initiate cell cycle to undergo proliferation [71]. In the early stages of regeneration, IL-6 signaling promotes muscle satellite cell proliferation by activating the JAK/ STAT1/STAT3 signaling pathway [72] and the PI3K/Akt signaling pathway which not only promotes muscle protein synthesis but also activate the proliferation of muscle satellite cells [73]. Both the IL-6 and PI3K/Akt signaling were found to be mediated by ROS in skeletal muscle [74,75]. ROS are likely to function as a double-edged sword in myogenesis.
POSITIVE EFFECTS OF ROS IN MYOGENESIS
At the physiological level, ROS modulate cellular proliferation, migration, differentiation, and muscle contractions [76]. However, under oxidative stress condition, it led to muscle damage and injury impairing muscle function [77]. Study conducted by Hansen et al. (2007) reported that lower intracellular redox potential promotes differentiation of skeletal muscle cell whereas higher redox potential exerts inhibitory effect [76]. Treatment of ROS scavenger, phenyl-N-tert-butylnitrone, decreased oxidative stress and increased C2C12 differentiation while addition of 25 μM H2O2 to cells in the presence of 20% O2 significantly impaired differentiation process [76]. Similarly, the physiological expression of Nrf2 attenuated the production of excessive ROS and enhanced myoblast proliferation and viability. However, overexpression of Nrf2 prevented C2C12 cell differentiation along with the downregulation of Myogenic Regulatory Factors (MRFs) [78]. Furthermore, differentiation of C2C12 cells was found to be associated with the increased ROS as indicated by increased expression of ROS related molecules such as hypoxia inducible factor1-alpha (HIF1-α), hypoxia inducible factor1-beta (HIF1-β), Von Hippel-Lindau (VHL), lysyl oxidase (Lox), EGL-9 family hypoxia-inducible factor 1 (EGLN1), proline 4-hydroxylase alpha 1 (P4HA1) and decreased level of heme oxygenase-1 (HOMX1) [79]. The moderate production of ROS during exercise or regeneration induces myogenic differentiation of satellite cells and myoblasts, while excessive accumulation of ROS results in their senescence, apoptosis, and regenerative failure in muscle repair [80,81]. These arguments suggest that moderate level of ROS is involved in regulation of myoblast differentiation while its inhibition by overactivation of antioxidants may interfere myogenesis.
NEGATIVE IMPACTS OF ROS IN MYOGENESIS
High ROS may target mitochondria and mitochondrial DNA causing blockage of myogenesis [9]. Excess ROS inhibited myogenesis with decreased expression of satellite cell markers, reduced expression of myoblast differentiation markers and reduced phosphorylation of MAPK signaling pathways [77,82]. The p38-MAPK signaling pathway is crucial in regulating skeletal muscle gene expression at different stages of the myogenic process [83]. ROS induced NF-kB activation lowers expression of MyoD, thereby inhibiting myogenesis [84]. Moreover, NF-κB was also found to suppress myofibrillar gene expression by mediating the regulation of myogenic transcriptional repressor Yin Yang 1 (YY1) [85,86]. Oxidative stress and p66ShcA, a mammalian adaptor protein localized in the mitochondria and functions as a redox enzyme that generates mitochondrial ROS, in skeletal muscle negatively modulate myogenic differentiation; in contrast, p66ShcA deletion enhances skeletal muscle regeneration after ischemia [87]. The deficiency of Nrf2 gene increased oxidative stress and decreased regeneration with decreased expression of Pax7/MyoD along with apoptosis [88,89]. Mild ROS produced by low dose gelatin in mice skeletal muscle stimulated ROS production from NOX2 with increasing antioxidant defense. It increased myokine IL-6 expression that enhance myogenesis and muscle regeneration. In contrast, high dose of gelatin produced excessive ROS (O2−, •OH) from NOX2 and mitochondrial chain complex, and suppressed antioxidant defense. This in turn released TNF-α preventing myogenesis and muscle regeneration [90]. Therefore, for proper understanding of the role of ROS in myogenesis, future studies are warranted to define an optimal intracellular redox environment that could facilitate the activation of muscle stem cells and promote differentiation of myoblasts into myotubes, thereby triggering the regeneration process in response to injury or damage.
ROS DURING EXERCISE
ROS stimulate antioxidant response and adaptations to exercise by activating redox signaling pathways such as peroxisome Proliferator- activated receptor Gamma Coactivator-1α (PGC-1α), Mitogen-Activated Protein Kinase (MAPK) and NFκB [91,92]. Exercise-induced ROS participates in improving muscle regeneration and recovery from muscle damage with insulin sensitivity [93,94]. Oxidative stress induced by exercise depends on the type, intensity, and duration of the exercise. Many studies have found that regular exercise and endurance training attenuates exercise-mediated oxidative stress along with improving the antioxidant status [95,96]. Regular exercise enhances angiogenesis, mitochondrial biogenesis, and muscle hypertrophy with improved physical fitness [97,98].
A clinical investigation found that ROS produced by physical exercise increase insulin sensitivity showing an adaptive response with the upregulation of ROS-mediated transcriptional coactivators PGC1-α, PGC1-β, and the transcription factor PPAR-gamma along with their target antioxidant SOD1, SOD2, GPx1 [99]. Surprisingly, the supplementation of antioxidants (vitamin C and E) revoked the beneficial effect of physical exercise as well as ROS mediated transcriptional activation [99]. Moreover, moderate-intensity exercise activates NOX2 that increased cytosolic ROS in human and mice. This in turn promoted the muscle glucose uptake via GLUT4 translocation [100]. Strikingly, lack of NOX2 subunits (either p47phox or Rac1) impaired ROS production and the glucose uptake via GLUT4 [100]. Exercise increases the concentration of FGF23 (Fibroblast growth factors) in skeletal muscle which improves endurance performance by controlling the ROS produced by exercise and enhancing mitochondrial functions [101].
Signaling pathways such as Adenosine Monophosphate-activated Protein Kinase (AMPK), Mitogen Activated Protein Kinase (MAPK), nuclear respiratory factor2 (Nrf2), and PGC-1α are regulated by exercise-mediated ROS production and participates in skeletal muscle responses [102,103]. Nrf2, a redox sensitive transcription factor, activates in response to exercise [104]. During its activation, it dissociates from its cytoplasmic inhibitor Keap1 and move to the nucleus to interact with Antioxidant Response Element (ARE) for transactivating downstream antioxidant genes such as oxidase Cytochrome Oxidase (COX), Superoxide Dismutase (SOD), Glutathione Peroxidase (GPX) activities and Glutathione (GSH) thereby alleviating the oxidative damage and promoting exercise-induced adaptations [105]. Merry et al. (2016) further reported that exercise-induced ROS and Nitric Oxide (NO) can activate Nrf2 which regulates skeletal muscle mitochondrial biogenesis markers such as Nuclear Respiratory Factor 1 (NRF-1), mitochondrial Transcription Factor A (mtTFA), and antioxidant defense gene, SOD1, SOD2 and catalase expression [106]. Deficiency of Nrf2 reduced mitochondrial biogenesis as well as decreased antioxidant defense [106]. Similarly, lactate produced by exercise can be coupled with ROS generation which can increase PGC1-α expression. To this connection, Nalbandian et al. (2019) has shown that treatment of C2C12 cells with lactate increased PGC1-α expression which was diminished by treatment of antioxidant NAC [107]. Exercise-induced ROS can stimulate generation of myokines in human skeletal muscle as treatment of antioxidants (Vitamins A, C and E) abrogated the myokines release such as IL-6 which regulates glucose homeostasis and inhibit pro-inflammatory effects of cytokines like TNF-a [108-110].
Exercise may also cause oxidative stress in skeletal muscle [111]. Various studies suggest that exercise particularly high-intensity exercise increased accumulation of ROS due to increased metabolic rate [112-114]. Intense or exhaustive exercise can produce excessive ROS and weakens the antioxidant defense system in skeletal muscle leading to muscle damage by modifying lipid, protein, DNA etc. [115,116]. Oxidative stress induced by exercise also cause post-exercise proteinuria [117]. Similarly, intense bicycling raised oxidative DNA damage along with DNA strand break down [118]. Therefore, intense or overtraining would elevate respiration rate and imbalance the redox homeostasis causing abnormality to physiological functions. Balanced and well-designed exercise training should be adopted as a preferred way to acquire the best benefits exerted by exercise (Figure 1).
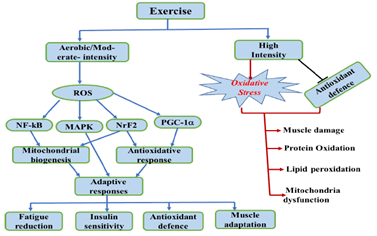
Figure 1: Diagram illustrating exercise-mediated ROS production and oxidative stress under moderate and high intensity exercise and their roles in muscle adaptation. Pointing arrows indicates induction/stimulation. ?? Indicates inhibition
Discussion
ROS in Pathophysiological Conditions
High level of ROS and RNS has been closely linked with many
pathophysiological conditions including skeletal muscle aging,
inflammation, and atrophy [119]. Chronic exposure of ROS/RNS
lead to macromolecular degradation, redox deregulation, impaired
calcium signaling, chronic inflammation, mitochondrial
dysfunction and activation of the apoptotic pathway through
NF-κB causing muscle atrophy, sarcopenia, and aging [120].
Moreover, it can damage skeletal muscle and cause metabolic
syndrome [121,122]. Selenium (Se), an essential trace element
plays vital role against oxidative stress induced pathologies.
Se is involved in Selenoprotein, the main form of selenium in
animal, synthesis in the form of Selenocysteine [123]. Se- deficiency
increased generation of ROS and lipid peroxidation,
and decreased activities of Glutathione Peroxidase (GPx), Total
Antioxidant Capacity (T-AOC), Superoxide Dismutase (SOD),
and Catalase (CAT) in skeletal muscle [124]. Moreover, Se- deficiency
showed decreased level of muscle differentiation genes
like Myog, Myod, Myh2, Myh3, and Myf5 [124]. It impairs antioxidant
activities of Glutathione Peroxidase (GPx) and thioredoxin
reductase, and also reduced the level of other antioxidant
selenoproteins [125]. Glutathione Peroxidase (GPx) like
GPx1, GPx2, GPx3, GPx4 are Selenium containing enzymes and
involved in the protection against oxidative stress [126]. GPx
reduces lipid hydroperoxide to their corresponding alcohol,
and hydrogen peroxide (H2O2) to water. Selenoprotein, particularly
Selenoprotein N (SelN) contributes to oxidative and
calcium homeostasis, with a potential role in the regulation of
the ryanodine receptor activity [127,128]. SelN1 is ubiquitously
expressed in the body and found early in muscle precursors
[129]. The mutation or deficiency of SelN was associated with
muscular disorder. SelN deficient myotubes showed increased
intracellular oxidant activity (ROS and Nitric oxide), excessive
oxidation of proteins including the contractile proteins actin
and myosin. Moreover, SelN-knockout myotubes exhibited impaired
Ca2+ homeostasis, potentially by dysfunction of the redox-
sensor Ca2+ channel RyR1 [130].
ROS in Age Related Sarcopenia
Sarcopenia, a term coined by Rosenberg in 1989, has been derived
from the Greek phrase means “loss of flesh.” It is a progressive
and generalized disorder of skeletal muscle identified
by reduced muscle strength, muscle mass or quality with impaired
physical performance [131], generally regarded as a geriatric
syndrome. It leads to significant difficulties in performing
routine daily activities conferring a high risk of fall and fractures
causing loss of independence and mortality. There are multifarious
causative factors for aging-induced sarcopenia such as diminished
regenerative potency, imbalance of protein degradation
and synthesis equilibrium, oxidative stress, mitochondrial
dysfunction, and inflammation etc. [132]. ROS accumulation is
increased in muscles during aging which instigate oxidation of
macromolecules (lipid, DNA, and protein), mitochondrial dysfunction,
inhibition of muscle cells differentiation, suggesting
that oxidative stress plays role in sarcopenia-induced muscle loss [133-135].
Skeletal muscle commonly maintains its mass by the homeostasis
of protein synthesis and breakdown; however, during aging
this equilibrium is disrupted due to various signaling induced
by oxidative stress and inflammation. In aging, ROS production
is increased as well as mitochondrial dysfunction occurs due
to mitochondrial DNA mutation and damage [136]. Moreover,
aged muscle cells unable to replace dysfunctional mitochondria
that further stimulate ROS accumulation [137]. A study by
Sullivan-Gunn et al. (2013) reported that aging triggers NOX2
mediated H2O2 production and reduces antioxidant enzymes
such as catalase and Glutathione Peroxidase (GPx) [138]. H2O2 activates calpain system of myofibrillar protein degradation
[139]. Calpain system may also be activated due to increased
intracellular Ca2+ concentration during aging [140]. Agarwal et
al. (2020) showed that redox remodeling induces a leaky ryanodine
receptor that disturbs Ca2+ reuptake via Sarcoplasmic
Reticulum Ca2+-ATPase (SERCA) pumps [141-143], and increase
Ca2+ concentration that lead to increased ROS which may activate
calpain that increases susceptibility of protein degradation
[144].
The CuZn-superoxide dismutase (SOD1) knockout aged mice
(Sod1(-/-)) showed increased oxidative stress, decreased muscle
mass and gait disturbance leading to sarcopenia [26]. Furthermore,
the study of same sod1(-/-) deficient mice model
reported that oxidative stress induced by SOD1 deficiency enhanced
oxidative damage with upregulation of cysteine proteases,
calpain and caspase-3 [145]. The activation of calpain and
caspase-3 subsequently initiate proteolytic process in skeletal
muscle [146]. In aged female Balb/c mice, the expression of
NOX component was increased significantly followed by increment
of superoxide (O2 ‾), H2O2 and reduction of key endogenous
antioxidant enzymes SOD1, catalase and GPx. This study
further suggests the contribution of H2O2 in the development
of sarcopenia [138]. Aging wistar rat muscle increased mitochondrial
H2O2 generation in the tibialis anterior muscle with
sarcopenia compared to their younger counterparts [147]. A
similar result was reported in vastis lateralis muscle of aged human
subjects [148]. Neuralization of H2O2 by muscle specific
Peroxiredoxin3 (PRX3) overexpression decreased mitochondrial
H2O2 production and improved mitochondrial function
along with alleviating loss of muscle mass and quality in murine
model of redox-dependent sarcopenia [149]. Sarcopenia
is also induced due to imbalance of protein turnover in elderly
age. Muscle protein synthesis is regulated by well-known anabolic
pathway PI3K/AKT/mTOR. Anabolic resistance develops in
aging that prevents protein synthesis in sarcopenic condition.
Increased ROS may prevent phosphorylation of components of
synthesis pathways such as Akt, mTOR and mTOR downstream
targets p70S6K and 4E-BP1 [150] (Figure 2).
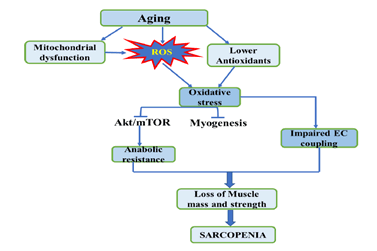
Figure 2: Diagram illustrating aging-induced ROS production and their roles in sarcopenia. Pointing arrows indicates induction/stimulation. ꟷꞁ Indicates inhibition.
Ros in Disuse-Induced Muscle Atrophy
Disused/inactivity conditions like limb immobilization, chronic
bed rest, spaceflight, spinal nerve injury, unloading etc. increases
ROS productions and decreases antioxidants enzymes
[151-153]. ROS are generated by several oxidants producing
pathways such as xanthine oxidase, nitric oxide synthase, NADPH
oxidase, and mitochondria. Although both xanthine oxidase
and NADPH oxidase participates to disuse-induced oxidants
production in skeletal muscle, mitochondria considered
to be the dominant site of ROS production [154-156]. Inactivity
results to decreased skeletal muscle mass and functions due to increased protein breakdown and decreased protein synthesis
[157,158]. We previously reported that denervation-induced
disused muscle significantly produced mitochondrial H2O2 followed
by muscle atrophy in mice [159]. The Ubiquitin-Proteasome
System (UPS) is one of the major proteolytic systems
in the body. Ubiquitin ligases such as Atrogin-1, MuRF-1, Cbl-b
participates in protein degradation in UPS and causes muscle
atrophy [160]. Previously, we found that microgravity and
clinorotation increased ROS production and ROS mediated
Cbl-b upregulation via ERK1/2 early-growth response protein
(Egr)1/2-Cbl-b signaling pathway to induce muscle atrophy in
L6 myotubes. Notably treatment with antioxidants like N-acetylcysteine
and TEMPOL significantly decreased ROS mediated
activation of ERK1/2 [161]. Cbl-b prompts degradation of IGF-
1 signaling intermediate Insulin Receptor Substrate-1 (IRS-1)
which ultimately activates FoxO3 dependent Atrgoin-1 and
MuRF-1 expression and initiates muscle protein degradation
[162]. Treatment of myotubes derived from COPD patient by
H2O2 induced ROS production and myotube atrophy by increasing
component of UPS such as Atrogin-1 and MuRF-1 [163]. In
a nutshell, Inactivity-induced oxidative stress upregulates the
expression of vital components of the ubiquitin-proteasome
system of proteolysis.
Secondly, inactivity increases autophagy in skeletal muscle suggesting
its role in disused induced muscle proteolysis [164].
ROS induces autophagy and increases autophagy related genes
by activating various ROS-mediated signaling like mitogen-activated
kinase (p38-MAPK), AMPK etc. ROS-induced activation
of p38-MAPK increased myotube atrophy and level of multiple
autophagy-related genes (e.g., Atg7, LC3 and Beclin-1) [165].
Mitochondrial targeted antioxidant SS-31 treatment decreased
autophagy related genes LC3, cathepsin etc. in soleus and
plantaris muscle of inactivity induced rat muscle [156]. Similarly,
ventilator-induced oxidative stress inhibition significantly
reduced autophagy markers in muscle fibers. Reduction of
oxidative stress in diaphragm by antioxidant during mechanical
ventilation decreased levels of LC3, Atg7, Atg12, Beclin-1,
cathepsin B, cathepsin D, and cathepsin L in diaphragm muscle
[164]. The above facts suggest that oxidative stress is involved
in activating autophagy that contributes to inactivity induced
proteolysis.
Calpains, a Ca2+ dependent proteolytic system is also involved
in disuse-induced muscle loss [166], as muscle-specific overexpression
of calpastatin (endogenous inhibitor of calpains) prevented
muscle mass loss in hindlimb unloading mediated disuse
model [167]. Oxidative stress increases the expression of
Calpain-1 and Calpain-2 [139,168]. Treatment of mitochondrial
targeted antioxidant SS-31 decreased calpain induced muscle
proteolysis by suppressing oxidative stress in immobilization
and mechanical ventilation-induced muscle atrophy in rodents
[169]. Based on all above reports it can be summarized that
inactivity and disuse mediates muscle atrophy via activating
different proteolytic system due to increased oxidative stress,
and supplementation of antioxidant decreases the severity of
muscle loss during prolong inactivity, although some contradiction
exist [170] (Figure 3).
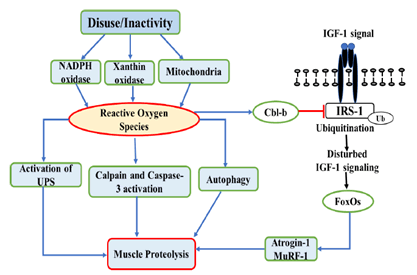
Figure 3: Diagram illustrating disuse/inactivity-induced ROS production and their roles in muscle proteolysis. Pointing arrows indicates induction/stimulation. ꟷꞁ Indicates inhibition.
ROS in Glucocorticoid-Induced Muscle Atrophy
Glucocorticoids (GCs) are steroidal drugs commonly prescribed
in inflammatory and autoimmune diseases. Prolong or high
intake of GCs is associated with multiple side effects such as
osteoporosis, adrenal gland dysfunction, hyperglycemia, and
muscle atrophy [171]. The level of GCs increased during different
pathological states e.g., sepsis, cachexia, starvation, metabolic
acidosis, and severe insulinopenia, that are characterized
by muscle atrophy, indicating the possibility that GC could trigger
muscle atrophy. GCs induce muscle atrophy by decreasing
protein synthesis and increasing protein degradation [171].
The major proteolytic pathway for GC-induced muscle atrophy
is the activation of the ubiquitin proteasomal system (UPS) and
the lysosomal system through the increased expression of several atrophy-inducing genes such as FOXOs, Atrogin-1, MuRF-1
etc. Moreover, the reduction of protein synthesis is mediated via inhibition of PI3K/AKT/mTOR pathway of protein synthesis
[171].
GCs have been reported to increase the production of ROS in
skeletal muscle in various cell and animal models [172-174]. In
humans, chronic GC administration increased 8-OHdG level (an
indicator of DNA damage) along with mitochondrial DNA damage
mediated by ROS in skeletal muscle [175]. Hydroxyl radicals
(OH•) were also increased by GC treatment in steroid myopathy
induced by dexamethasone (Dex) [176]. Previously, we reported
that Dex increases ROS production via glucocorticoid receptor-
mediated pathway that induced muscle atrophy by increasing
Atrogin-1 and MuRF-1 with the upregulation of Cbl-b
[177]. Cbl-b is an oxidative stress-sensitive ubiquitin ligase that
is upregulated by ROS and causes muscle atrophy by increasing
Atrogin-1 and MuRF-1 [161,162]. Dex also decreased antioxidant
enzymes such as SOD1 and catalase. A study conducted
by Espinoza et al. reported that GC treatment upregulates NADPH
oxidase (NOX) mRNA expression which generates ROS by
catalyzing the transfer of electrons to O2, producing superoxide
or H2O2, using NADPH as an electron donor [178]. NOX1 is upregulated
by Dex in smooth muscles [179]. Moreover, Dex-induced
hydroxyl free radical (OH•) production decreased cell
viability and increased apoptosis to C2C12 cells which was reversed
by treatment with the antioxidant quercetin [180]. Dex
increased cellular ROS levels and induced oxidative damage to
proteins and lipids in skeletal muscle [181]. Moreover, it causes
mitochondrial dysfunction by impairing mitochondrial respiration
and decreasing the activities of mitochondrial complexes
I, II, and IV, and ATP synthase [181,182]. Supplementation of
antioxidant compound Psoralea corylifolia L. seed extract decreased
Dex-induced muscle atrophy by decreasing oxidative
stress and inflammation [183]. Based on above reports, it can
be suggested that ROS may be a causative agent in glucocorticoid
induced muscle atrophy. The schematic diagram of glucocorticoid-
induced muscle atrophy has been shown below
(Figure 4).
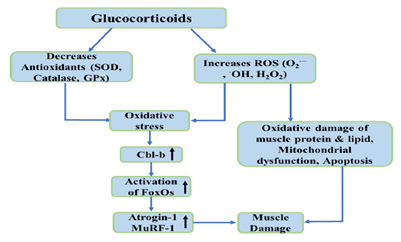
Figure 4: Diagram illustrating ROS generation during glucocorticoid induced muscle atrophy. Pointing arrows indicates induction/stimulation. ↑ Indicates increase of shown component.
Conclusion
Critical Overview and Future Perspective
This review has summarized the role of ROS as regulatory and
pathological agent in skeletal muscle. The production of ROS
in the skeletal muscle occurs via multiple mechanisms. This review
has tried to explain both the positive and negative effects
of ROS on skeletal muscle health along with their mechanism.
According to the results discussed above, ROS act as a dual
agent in performing physiological and pathological functions of
muscle health. The precise action of ROS is intricately associated
with concentration and origin of ROS. At physiological concentrations,
ROS stimulate the pathways that induce myogenesis,
muscle differentiation, exercise adaptation, EC coupling
etc. However, at high concentrations, ROS initiate macromolecular
damage, mitochondrial dysfunction, activation of proteolytic
pathways, impaired calcium signaling, and altered redox
regulation that led to muscle dysfunction such as sarcopenia and muscle atrophy. So, our knowledge gap to understand the
physiological and pathological functions of ROS lies on defining
the boundaries of the redox window. Further research focusing
measurements of the precise nature of ROS and their effect on
muscular cells is required along with the deeper study of redox-
sensitive pathways to clearly differentiate the pathological
and physiological roles of ROS in skeletal muscle.
Author Contribution
A.U.: Conceptualization, data curation, writing-original draft,
and visualization; T.N.: Conceptualization, writing-review and
editing, visualization and Funding acquisition.
Funding
This work was supported by Cabinet Office, Government of Japan,
Cross ministerial Moonshot Agriculture, Forestry and Fisheries
Research and Development Program, “Technologies for
Smart Bio-industry and Agriculture” (funding agency: Bio-oriented
Technology Research Advancement Institution), Grand
Number JPJ009237.
Acknowledgement
The authors would like to thank Enago for the English language
editing of this manuscript.
Conflict Of Interest
The authors declare no conflict of interest.
References
- Frontera WR, Ochala J (2014) Skeletal muscle: A brief review of structure and function. Calcif Tissue Int. 96(3): 183-195.
[Crossref] [Google Scholar]
- Michaelson LP, Iler C, Ward CW (2013) ROS and RNS signaling in skeletal muscle: Critical signals and therapeutic targets. Annu Rev Nurs Res. 31: 367-87.
[Crossref] [Google Scholar]
- Muller FL, Song W, Jang C, Liu Y, Sabia M, et al. (2007) Denervation-induced skeletal muscle atrophy is associated with increased mitochondrial ROS production. Am J Physiol Regul Integr Comp Physiol. 293(3): R1159-R1168.
[Crossref] [Google Scholar]
- Petrillo S, Pelosi L, Piemonte F, Travaglini L, Forcina L, et al. (2017) Oxidative stress in duchenne muscular dystrophy: Focus on the NRF2 redox pathway. Hum Mol Genet. 26(14): 2781-2790.
[Crossref] [Google Scholar]
- Sullivan-Gunn MJ, Lewandowski PA (2013) Elevated hydrogen peroxide and decreased catalase and glutathione peroxidase protection are associated with aging sarcopenia. BMC Geriatr. 13(1): 104.
[Crossref] [Google Scholar]
- Barbieri E, Sestili P (2012) Reactive oxygen species in skeletal muscle signaling. J Signal Transduct. 2012: 982794.
[Crossref] [Google Scholar]
- He F, Li J, Liu Z, Chuang C, Yang W, et al. (2016) Redox mechanism of reactive oxygen species in exercise. Front Physiol. 7: 486.
[Crossref] [Google Scholar]
- Kang C, O'Moore KM, Dickman JR (2009) Exercise activation of muscle peroxisome proliferator-activated receptor-γ coactivator-1α signaling is redox sensitive. Free Radic Biol Med. 47(10): 1394-1400.
[Crossref] [Google Scholar]
- RochardP, Rodier A, Casas F, Malek IC, Victorion SM, et al. (2000) Mitochondrial activity is involved in the regulation of myoblast differentiation through myogenin expression and activity of myogenic factors. J Biol Chem. 275(4): 2733-2744.
[Crossref] [Google Scholar]
- Crane JD, Abadi A, Hettinga BP, OgbornDI, MacNeil LG, et al. (2013) Elevated mitochondrial oxidative stress impairs metabolic adaptations to exercise in skeletal muscle. PLoSOne. 8(12): e81879.
[Crossref] [Google Scholar]
- Yoon SO, Yun CH, Chung AS (2002) Dose effect of oxidative stress on signal transduction in aging. Mech Ageing Dev. 123(12): 1597-1604.
[Crossref] [Google Scholar]
- Neurohr JM, Paulson ET, Kinsey ST (2021) A higher mitochondrial content is associated with greater oxidative damage, oxidative defenses, protein synthesis and ATP turnover in resting skeletal muscle. J Exp Biol. 224(19): jeb242462.
[Crossref] [Google Scholar]
- Barja G (1999) Mitochondrial oxygen radical generation and leak: Sites of production in states 4 and 3, organ specificity, and relation to aging and longevity. J Bioenerg Biomembr. 31(4): 347-66.
[Crossref] [Google Scholar]
- Sakellariou GK, Jackson MJ, Vasilaki A (2014) Redefining the major contributors to superoxide production in contracting skeletal muscle. The role of NAD(P)H oxidases. Free Radic Res. 48(1): 12-29.
[Crossref] [Google Scholar]
- Ferreira LF, Laitano O (2016) Regulation of NADPH oxidases in skeletal muscle. Free Radical Bio Med. 98: 18-28.
[Crossref] [Google Scholar]
- Gomez-Cabrera MC, Close GL, Kayani A, McArdle A, Vina J, et al. (2010) Effect of xanthine oxidase-generated extracellular superoxide on skeletal muscle force generation. Am J Physiol Regul Integr Comp Physiol. 298(1): R2-8.
[Crossref] [Google Scholar]
- Zuo L, Christofi FL, Wright VP, Bao S, Clanton TL (2004) Lipoxygenase-dependent superoxide release in skeletal muscle. J Appl Physiol. 97(2): 661-668.
[Crossref] [Google Scholar]
- Nethery D, Callahan LA, Stofan D, Mattera R, DiMarco A, et al. (2000) PLA2 dependence of diaphragm mitochondrial formation of reactive oxygen species. J Appl Physiol. 89(1): 72-80.
[Crossref] [Google Scholar]
- Gong MC, Arbogast S, Guo Z, Mathenia J, Su W, et al. (2006) Calcium-independent phospholipase A2 modulates cytosolic oxidant activity and contractile function in murine skeletal muscle cells. J Appl Physiol. 100(2): 399-405.
[Crossref] [Google Scholar]
- Ahmad MI, Bensalah N (2022) Insights into the generation of hydroxyl radicals from H2O2 decomposition by the combination of Fe2+ and chloranilic acid. Int J Environ Sci Technol. 19(10): 10119-10130.
[Crossref] [Google Scholar]
- Mehta SK, Gowder SJT (2015) Members of antioxidant machinery and their functions. IntechOpen: Rijeka.
[Crossref] [Google Scholar]
- Wang Y, Branicky R, Noe A, Hekimi S (2018) Superoxide dismutases: Dual roles in controlling ROS damage and regulating ROS signaling. J Cell Biol. 217(6): 1915-1928.
[Crossref] [Google Scholar]
- Powers SK, Criswell D, Lawler J, Ji LL, Martin D, et al. (1994) Influence of exercise and fiber type on antioxidant enzyme activity in rat skeletal muscle. Am J Physiol Regul Integr Comp Physiol. 266(2): R375-R380.
[Crossref] [Google Scholar]
- Criswell D, Powers S, Dodd S, Lawler J, Edwards W, et al. (1993) High intensity training-induced changes in skeletal muscle antioxidant enzyme activity. Med Sci Sports Exerc. 25(10): 1135-1140.
[Crossref] [Google Scholar]
- Ji LL, Dillon D, Wu E (1990) Alteration of antioxidant enzymes with aging in rat skeletal muscle and liver. Am J Physiol. 258(4): R918-R923.
[Crossref] [Google Scholar]
- MullerFL, Song W, Liu Y, Chaudhuri A, Dahl SP, et al. (2006) Absence of Cu Zn superoxide dismutase leads to elevated oxidative stress and acceleration of age-dependent skeletal muscle atrophy. Free Radic Biol Med. 40(11): 1993-2004.
[Crossref] [Google Scholar]
- Alfonso-Prieto M, Biarnes X, Vidossich P, Rovira C (2009) The molecular mechanism of the catalase reaction. J Am Chem Soc. 131(33): 11751-11761.
[Crossref] [Google Scholar]
- Riley DA, Ellis S, Bain JL (1988) Catalase-positive microperoxisomes in rat soleus and extensor digitorum longus muscle fiber types. J Histochem Cytochem. 36(6): 633-637.
[Crossref] [Google Scholar]
- Lubos E, Loscalzo J, Handy (2011) DE Glutathione peroxidase-1 in health and disease: From molecular mechanisms to therapeutic opportunities. Antioxid Redox Signal. 15(7): 1957-1997.
[Crossref] [Google Scholar]
- Sen CK, Marin E, Kretzschmar M, Hanninen O (1992) Skeletal muscle and liver glutathione homeostasis in response to training, exercise, and immobilization. J Appl Physiol 73(4): 1265-1272.
[Crossref] [Google Scholar]
- Arner ES, Holmgren A (2000) Physiological functions of thioredoxin and thioredoxin reductase. Eur J Biochem. 267(20): 6102-6109.
[Crossref] [Google Scholar]
- Fernandes AP, Holmgren A (2004) Glutaredoxins: Glutathione-dependent redox enzymes with functions far beyond a simple thioredoxin backup system. Antioxid Redox Signal. 6(1): 63-74.
[Crossref] [Google Scholar]
- Lee KP, Shin YJ, Cho SC, Lee SM, Bahn YJ, et al. (2014) Peroxiredoxin 3 has a crucial role in the contractile function of skeletal muscle by regulating mitochondrial homeostasis. Free Radic Biol Med. 77: 298-306.
[Crossref] [Google Scholar]
- Ziad M, Zaher MAJ, Saleh AA (2019) Nonenzymatic exogenous and endogenous antioxidants. IntechOpen: Rijeka.
[Crossref] [Google Scholar]
- Leeuwenburgh C, Hollander J, Leichtweis S, Griffiths M, Gore M, et al. (1997) Adaptations of glutathione antioxidant system to endurance training are tissue and muscle fiber specific. Am J Physiol. 272(1): R363-R369.
[Crossref] [Google Scholar]
- Meister A, Anderson ME (1983) Glutathione. Annu Rev Biochem. 52: 711-760.
[Crossref] [Google Scholar]
- Ferreira JCB, Bacurau AV, Bueno CR, Cunha TC, Tanaka LY, et al. (2010) Aerobic exercise training improves Ca2+ handling and redox status of skeletal muscle in mice. Exp Biol Med. 235(4): 497-505.
[Crossref] [Google Scholar]
- Huang HY, Appel LJ, Croft KD, Miller ER, Mori TA, et al. (2002) Effects of vitamin C and vitamin E on in vivo lipid peroxidation: Results of a randomized controlled trial. Am J Clin Nutr. 76(3): 549-555.
[Crossref] [Google Scholar]
- Kagan VE, Shvedova A, Serbinova E, Khan S, Swanson C, et al. (1992) Dihydrolipoic acid-a universal antioxidant both in the membrane and in the aqueous phase: Reduction of peroxyl, ascorbyl and chromanoxyl radicals. Biochem Pharmacol. 44(8): 1637-1649.
[Crossref] [Google Scholar]
- Ames BN, Cathcart R, Schwiers E, Hochstein P (1981) Uric acid provides an antioxidant defense in humans against oxidant-and radical-caused aging and cancer: A hypothesis. Proc Natl Acad Sci. 78(11): 6858-6862.
[Crossref] [Google Scholar]
- Sautin YY, Johnson RJ (2008) Uric acid: The oxidant-antioxidant paradox. Nucleosides Nucleotides Nucleic Acids. 27(6): 608-619.
[Crossref] [Google Scholar]
- Stocker R, Glazer AN, Ames BN (1987) Antioxidant activity of albumin-bound bilirubin. Proc Natl Acad Sci. 84(16): 5918-22.
[Crossref] [Google Scholar]
- Littarru GP, Tiano L (2007) Bioenergetic and antioxidant properties of coenzyme Q10: Recent developments. Mol Biotechnol. 37(1): 31-37.
[Crossref] [Google Scholar]
- Pandey KB, Rizvi SI (2009) Plant polyphenols as dietary antioxidants in human health and disease. Oxid Med Cell Longev. 2(5): 270-278.
[Crossref] [Google Scholar]
- Ray PD, Huang BW, Tsuji Y (2012) Reactive Oxygen Species (ROS) homeostasis and redox regulation in cellular signaling. J Cell Sig. 24(5): 981-990.
[Crossref] [Google Scholar]
- Meo SD, Reed TT, Venditti R, Victor MV (2016) Role of ROS and RNS sources in physiological and pathological conditions. Oxid Med Cell Longev. 2016: 1245049.
[Crossref] [Google Scholar]
- Holmstrom KM, Finkel T (2014) Cellular mechanisms and physiological consequences of redox-dependent signalling. Nat Rev Mol Cell Biol. 15(6): 411-421.
[Crossref] [Google Scholar]
- Papaiahgari S, Kleeberger SR, Cho HY, Kalvakolanu DV, Reddy SP (2004) NADPH oxidase and ERK signaling regulates hyperoxia-induced Nrf2-ARE transcriptional response in pulmonary epithelial cells. J Biol Chem. 279(40): 42302-42312.
[Crossref] [Google Scholar]
- Bouviere J, Fortunato RS, Dupuy C, Castro JPW, Carvalho DP, et al. (2021) Exercise-stimulated ROS sensitive signaling pathways in skeletal muscle. Antioxidants. 10(4): 537.
[Crossref] [Google Scholar]
- Powers SK, Duarte J, Kavazis AN, Talbert EE (2010) Reactive oxygen species are signalling molecules for skeletal muscle adaptation. Exp Physiol. 95(1): 1-9.
[Crossref] [Google Scholar]
- Handayaningsih AE, Iguchi G, Fukuoka H, Nishizawa H, Takahashi M, et al. (2011) Reactive oxygen species play an essential role in igf-i signaling and IGF-I-induced myocyte hypertrophy in C2C12 myocytes. Endocrinology. 152(3): 912-921.
[Crossref] [Google Scholar]
- Calderon JC, Bolanos P, Caputo C (2014) The excitation-contraction coupling mechanism in skeletal muscle. Biophys Rev. 6(1): 133-160.
[Crossref] [Google Scholar]
- Meissner G (2002) Regulation of mammalian ryanodine receptors. Front Biosci. 7(4): 2072-2080.
[Crossref] [Google Scholar]
- Li J, King NC, Sinoway LI (2003) ATP concentrations and muscle tension increase linearly with muscle contraction. J Appl Physiol. 95(2): 577-583.
[Crossref] [Google Scholar]
- Schartner V, Laporte J, Bohm J (2019) Abnormal excitation-contraction coupling and calcium homeostasis in myopathies and cardiomyopathies. J Neuromuscul Dis. 6(3): 289-305.
[Crossref] [Google Scholar]
- Treves S, Jungbluth H, Voermans N, Muntoni F, Zorzato F (2017) Ca2+ handling abnormalities in early-onset muscle diseases: Novel concepts and perspectives. Semin Cell Dev Biol. 64: 201-212.
[Crossref] [Google Scholar]
- Xu H, Ahn B, Remmen HV (2022) Impact of aging and oxidative stress on specific components of excitation contraction coupling in regulating force generation. Sci Adv. 8(43): eadd7377.
[Crossref] [Google Scholar]
- Espinosa A, Leiva A, Pena M, Muller M, Debandi A, et al. (2006) Myotube depolarization generates reactive oxygen species through NAD (P)H oxidase; ROS-elicited Ca2+ stimulates ERK, CREB, early genes. J Cell Physiol. 209(2): 379-388.
[Crossref] [Google Scholar]
- Hidalgo C, Sanchez G, Barrientos G, Parks PA (2006) A transverse tubule NADPH oxidase activity stimulates calcium release from isolated triads via ryanodine receptor type 1 S-glutathionylation. J Biol Chem. 281(36): 26473-26482.
[Crossref] [Google Scholar]
- Agrawal A, Rathor R, Kumar R, Suryakumar G, Singh SN, et al. (2020) Redox modification of ryanodine receptor contributes to impaired Ca2+ homeostasis and exacerbates muscle atrophy under high altitude. Free Radic Biol Med. 160: 643-656.
[Crossref] [Google Scholar]
- Qaisar R, Bhaskaran S, Ranjit R, Sataranatarajan K, Premkumar P, et al. (2019) Restoration of SERCA ATPase prevents oxidative stress-related muscle atrophy and weakness. Redox Biol. 20: 68-74.
[Crossref] [Google Scholar]
- Filetti FM, Vassallo DV, Fioresi M, Simoes MR (2018) Reactive oxygen species impair the excitation-contraction coupling of papillary muscles after acute exposure to a high copper concentration. Toxicol In Vitro. 51: 106-113.
[Crossref] [Google Scholar]
- Rossi A, Pizzo P, Filadi R (2019) Calcium, mitochondria and cell metabolism: A functional triangle in bioenergetics. Biochim Biophys Acta Mol Cell Res. 1866(7): 1068-1078.
[Crossref] [Google Scholar]
- Sharov VS, Dremina ES, Galeva NA, Williams TD, SchÃÂ?¶neich C (2006) Quantitative mapping of oxidation-sensitive cysteine residues in SERCA in vivo and in vitro by HPLC-electrospray-tandem MS: Selective protein oxidation during biological aging. Biochem J. 394(3): 605-615.
[Crossref] [Google Scholar]
- Adachi T, Weisbrod RM, Pimentel DR, Ying J, Sharov VS, et al. (2004) S-Glutathiolation by peroxynitrite activates SERCA during arterial relaxation by nitric oxide. Nat Med. 10(11): 1200-1207.
[Crossref] [Google Scholar]
- Xu H, Remmen HV (2021) The SarcoEndoplasmic Reticulum Calcium ATPase (SERCA) pump: A potential target for intervention in aging and skeletal muscle pathologies. Skelet Muscle. 11(1): 25.
[Crossref] [Google Scholar]
- Xu H, Ahn B, Remmen HV (2022) Impact of aging and oxidative stress on specific components of excitation contraction coupling in regulating force generation. Sci Adv. 8(43): eadd7377.
[Crossref] [Google Scholar]
- Rudnicki MA, Grand FL, McKinnell I, Kuang S (2008) The molecular regulation of muscle stem cell function. Cold Spring Harb Symp Quant Biol. 73: 323-31.
[Crossref] [Google Scholar]
- Pellettieri J, Alvarado AS (2007) Cell turnover and adult tissue homeostasis: From humans to planarians. Annu Rev Genet. 41: 83-105.
[Crossref] [Google Scholar]
- Hernandez-Hernandez JM, Garcia-Gonzalez EG, Brun CE, Rudnicki MA (2017) The myogenic regulatory factors, determinants of muscle development, cell identity and regeneration. Semin Cell Dev Biol. 72: 10-18.
[Crossref] [Google Scholar]
- Charge SBP, Rudnicki MA (2004) Cellular and molecular regulation of muscle regeneration. Physiol Rev. 84(1): 209-238.
[Crossref] [Google Scholar]
- Xiao F, Wang H, Fu X, LiY, Ma K, et al. (2011) Oncostatin M inhibits myoblast differentiation and regulates muscle regeneration. Cell Res. 21(2): 350-364.
[Crossref] [Google Scholar]
- Fu Y, Li S, Tong H, Li S, Yan Y (2019) WDR13 promotes the differentiation of bovine skeletal muscle-derived satellite cells by affecting PI3K/AKT signaling. Cell Biol Int. 43(7): 799-808.
[Crossref] [Google Scholar]
- Kosmidou I, Vassilakopoulos T, Xagorari A, Zakynthinos S, Papapetropoulos A, et al. (2002) Production of interleukin-6 by skeletal myotubes: Role of reactive oxygen species. Am J Respir Cell Mol Biol. 26(5): 587-93.
[Crossref] [Google Scholar]
- Kosmidou I, Xagorari A, Roussos C, Papapetropoulos A (2001) Reactive oxygen species stimulate VEGF production from C2C12 skeletal myotubes through a PI3K/Akt pathway. Am J Physiol Lung Cell Mol Physiol. 280(4): L585-L592.
[Crossref] [Google Scholar]
- Hansen JM, Klass M, Harris C, Csete M (2007) A reducing redox environment promotes C2C12 myogenesis: Implications for regeneration in aged muscle. Cell Biol Int. 31(6): 546-553.
[Crossref] [Google Scholar]
- Ikeda Y, Satoh A, Horinouchi Y, Hamano H, Watanabe H, et al. (2019) Iron accumulation causes impaired myogenesis correlated with MAPK signaling pathway inhibition by oxidative stress. Faseb J. 33(8): 9551-9564.
[Crossref] [Google Scholar]
- Rajasekaran NS, Shelar SB, Jones DP, Hoidal JR (2020) Reductive stress impairs myogenic differentiation. Redox Biol. 34: 101492.
[Crossref] [Google Scholar]
- Li X, Zhang S, Zhang Y, Liu P, Li M, et al. (2021) Myoblast differentiation of C2C12 cell may related with oxidative stress. Intractable Rare Dis Res. 10(3): 173-178.
[Crossref] [Google Scholar]
- Abruzzo PM, Esposito F, Marchionni C, Tullio SD, Belia S, et al. (2013) Moderate exercise training induces ROS-related adaptations to skeletal muscles. Int J Sports Med. 34(8): 676-87.
[Crossref] [Google Scholar]
- Pal S, Chaki B, Chattopadhyay S, Bandyopadhyay A (2018) High-intensity exercise induced oxidative stress and skeletal muscle damage in postpubertal boys and girls: A comparative study. J Strength Cond Res. 32(4): 1045-1052.
[Crossref] [Google Scholar]
- Reardon TF, Allen DG (2009) Iron injections in mice increase skeletal muscle iron content, induce oxidative stress and reduce exercise performance. Exp Physiol. 94(6): 720-30.
[Crossref] [Google Scholar]
- Lluis F, Perdiguero E, Nebreda AR, Canoves PM (2006) Regulation of skeletal muscle gene expression by p38 MAP kinases. Trends Cell Biol. 16(1): 36-44.
[Crossref] [Google Scholar]
- Ardite E, Albert BJ, Roca J, Fernandez-Checa JC (2004) Glutathione depletion impairs myogenic differentiation of murine skeletal muscle C2C12 cells through sustained NF-kappaB activation. Am J Pathol. 165(3): 719-728.
[Crossref] [Google Scholar]
- Wang H, Hertlein E, Bakkar N, Sun H, Acharyya S, et al. (2007) NF-kappaB regulation of YY1 inhibits skeletal myogenesis through transcriptional silencing of myofibrillar genes. Mol Cell Biol. 27(12): 4374-4387.
[Crossref] [Google Scholar]
- Zhou LZ, Johnson AP, Rando TA (2001) NF kappa, B and AP-1 mediate transcriptional responses to oxidative stress in skeletal muscle cells. Free Radic Biol Med. 31(11): 1405-1416.
[Crossref] [Google Scholar]
- Zaccagnini G, Martelli F, Magenta A, Cencioni C, Fasanaro P, et al. (2007) p66ShcA and oxidative stress modulate myogenic differentiation and skeletal muscle regeneration after hind limb ischemia. J Biol Chem. 282(43): 31453-31459.
[Crossref] [Google Scholar]
- Narasimhan M, Hong J, Atieno N, Muthusamy VR, Davidson CJ, et al. (2014) Nrf2 deficiency promotes apoptosis and impairs PAX7/MyoD expression in aging skeletal muscle cells. Free Radic Biol Med. 71: 402-414.
[Crossref] [Google Scholar]
- Shelar SB, Narasimhan M, Shanmugam G, Litovsky SH, Gounder SS, et al. (2016) Disruption of nuclear factor (erythroid-derived-2)-like 2 antioxidant signaling: A mechanism for impaired activation of stem cells and delayed regeneration of skeletal muscle. Faseb J. 30(5): 1865-1879.
[Crossref] [Google Scholar]
- LiuX, Zu E, Chang X, Ma X, Wang Z, et al. (2021) Bi-phasic effect of gelatin in myogenesis and skeletal muscle regeneration. Dis Model Mech. 14(12): dmm049290.
[Crossref] [Google Scholar]
- Kang C, O'Moore KM, Dickman JR, Ji LL (2009) Exercise activation of muscle peroxisome proliferator-activated receptor-gamma coactivator-1alpha signaling is redox sensitive. Free Radic Biol Med. 47(10): 1394-1400.
[Crossref] [Google Scholar]
- Ji LL (2002) Exercise-induced modulation of antioxidant defense. Ann N Y Acad Sci. 959: 82-92.
[Crossref] [Google Scholar]
- Teixeira VH, Valente HF, Casal SI, Marques AF, Moreira PA (2009) Antioxidants do not prevent post exercise peroxidation and may delay muscle recovery. Med Sci Sports Exerc. 41(9): 1752-1760.
[Crossref] [Google Scholar]
- Ristow M, Zarse K, Oberbach A, Kloting N, Kloting M, et al. (2009) Antioxidants prevent health-promoting effects of physical exercise in humans. Proc Natl Acad Sci. 106(21): 8665-8670.
[Crossref] [Google Scholar]
- Taysi S, Oztasan N, Efe H, Polat MF, Gumustekin K, et al. (2008) Endurance training attenuates the oxidative stress due to acute exhaustive exercise in rat liver. Acta Physiol Hung. 95(4): 337-347.
[Crossref] [Google Scholar]
- Chaumont SB, Maupoil V, Lahet JJ, Berthelot A (2001) Effect of exercise training on metallothionein levels of hypertensive rats. Med Sci Sports Exerc. 33(5): 724-728.
[Crossref] [Google Scholar]
- Oliveira AN, Richards BJ, Slavin M, Hood DA (2021) Exercise is muscle mitochondrial medicine. Exerc Sport Sci Rev. 49(2): 67-76.
[Crossref] [Google Scholar]
- Menshikova EV, Ritov VB, Ferrell RE, Azuma K, Goodpaster BH, et al. (2007) Characteristics of skeletal muscle mitochondrial biogenesis induced by moderate-intensity exercise and weight loss in obesity. J Appl Physiol. 103(1): 21-27.
[Crossref] [Google Scholar]
- Ristow M, Zarse K, Oberbach A, Kloting N, Birringer M, et al. (2009) Antioxidants prevent health-promoting effects of physical exercise in humans. Proc Natl Acad Sci. 106(21): 8665-8670.
[Crossref] [Google Scholar]
- Henriquez-Olguin C, Knudsen JR, Raun SH, Li Z, Dalbram E, et al. (2019) Cytosolic ROS production by NADPH oxidase 2 regulates muscle glucose uptake during exercise. Nat Commun. 10(1): 4623.
[Crossref] [Google Scholar]
- Li DJ, Fu H, Zhao T, Ni M, Shen FM (2016) Exercise-stimulated FGF23 promotes exercise performance via controlling the excess reactive oxygen species production and enhancing mitochondrial function in skeletal muscle. Metab Clin Exp 65(5): 747-756.
[Crossref] [Google Scholar]
- Ji L, Gomezcabrera MC, Steinhafel L, Vina J (2004) Acute exercise activates nuclear factor (NF)-?B signaling pathway in rat skeletal muscle. FASEB J. 18(13): 1499-1506.
[Crossref] [Google Scholar]
- Merry TL, Ristow M, (2016) Nuclear factor erythroid-derived 2-like 2 (NFE2L2, Nrf2) mediates exercise-induced mitochondrial biogenesis and the anti-oxidant response in mice. J Physiol. 594(18): 5195-5207.
[Crossref] [Google Scholar]
- Oh S, Komine S, Warabi E, Akiyama K, Ishii A, et al. (2017) Nuclear factor (erythroid derived 2)-like 2 activation increases exercise endurance capacity via redox modulation in skeletal muscles. Sci Rep. 7(1): 12902.
[Crossref] [Google Scholar]
- Ma Q (2013) Role of Nrf2 in oxidative stress and toxicity. Annu Rev Pharmacol Toxicol 53(1): 401-426.
[Crossref] [Google Scholar]
- Mallard AR, Spathis JG, Coombes JS (2016) Nuclear factor (erythroid-derived 2)-like 2 (Nrf2) and exercise. Free Radic Biol Med. 16: 471-479.
[Crossref] [Google Scholar]
- Nalbandian M, Radak Z, Takeda M (2019) M N-acetyl-L-cysteine prevents lactate-mediated PGC1-alpha expression in C2C12 myotubes. Biology. 8(2): 44.
[Crossref] [Google Scholar]
- Vassilakopoulos T, Karatza MH, Katsaounou P, Kollintza A, Zakynthinos S, et al. (2003) Antioxidants attenuate the plasma cytokine response to exercise in humans. J Appl Physiol. 94(3): 1025-1032.
[Crossref] [Google Scholar]
- Pedersen BK, Steensberg A, Schjerling P (2001) Muscle-derived interleukin-6: Possible biological effects. J Physiol. 536(2): 329-337.
[Crossref] [Google Scholar]
- Scheele C, Nielsen S, Pedersen BK (2009) ROS and myokines promote muscle adaptation to exercise. Trends Endocrinol Metab. 20(3): 95-99.
[Crossref] [Google Scholar]
- Saborido A, Naudi A, Otin MP, Pamplona R, Megias A (2011) Stanozolol treatment decreases the mitochondrial ROS generation and oxidative stress induced by acute exercise in rat skeletal muscle. J Appl Physiol 110(3): 661-669.
[Crossref] [Google Scholar]
- Sahlin K, Shabalina IG, Mattsson CM, Bakkman L, Fernstrom M, et al. (2010) Ultraendurance exercise increases the production of reactive oxygen species in isolated mitochondria from human skeletal muscle. J Appl Physiol. 108(4): 780-787.
[Crossref] [Google Scholar]
- Alessio HM, Hagerman AE, Fulkerson BK, Ambrose J, Rice RE, et al. (2000) Generation of reactive oxygen species after exhaustive aerobic and isometric exercise. Med Sci Sports Exerc. 32(9): 1576-1581.
[Crossref] [Google Scholar]
- Shi M, Wang X, Yamanaka T, Ogita F, Nakatani K, et al. (2007) Effects of anaerobic exercise and aerobic exercise on biomarkers of oxidative stress. Environ Health Prev Med. 12(5): 202-208.
[Crossref] [Google Scholar]
- Wang Y, Chen M, Gao Y, He K, Yang Z, et al (2022) Effect of one-time high load exercise on skeletal muscle injury in rats of different genders: Oxidative stress and mitochondrial responses. Acta Cir Bras. 37(8): e370805.
[Crossref] [Google Scholar]
- Kayatekin B, Gonenc S, Acikgoz O, Uysal N, Dayi A (2002) Effects of sprint exercise on oxidative stress in skeletal muscle and liver. Eur J Appl Physiol. 87(2): 141-144.
[Crossref] [Google Scholar]
- Kocer G, senturk UK, Kuru O, Gunduz F (2008) Potential sources of oxidative stress that induce postexercise proteinuria in rats. J Appl Physiol. 104(4): 1063-1068.
[Crossref] [Google Scholar]
- Moller P, Loft S, Lundby C, Olsen NV (2001) Acute hypoxia and hypoxic exercise induce DNA strand breaks and oxidative DNA damage in humans. Faseb J. 15(7): 1181-1186.
[Crossref] [Google Scholar]
- Kadoguchi T, Shimada K, Miyazaki T, Kitamura K, Kunimoto M, et al. (2020) Promotion of oxidative stress is associated with mitochondrial dysfunction and muscle atrophy in aging mice. Geriatr Gerontol Int. 20(1): 78-84.
[Crossref] [Google Scholar]
- Abdel-Rahman EA, Mahmoud AM, Khalifa AM, Ali SS (2016) Physiological and pathophysiological reactive oxygen species as probed by EPR spectroscopy: The underutilized research window on muscle ageing. J Physiol. 594(16): 4591-613.
[Crossref] [Google Scholar]
- Hassanin MM, Tolba M, Tadros M, Elmazar M, Singab A (2020) Wogonin hampers dexamethasone-induced oxidative imbalance in sprague dawely rats. Arch Pharm Sci Ain Shams Univ. 4(1): 70-78.
[Crossref] [Google Scholar]
- Yokota T, Kinugawa H, Yamato M, Hirabayashi K, Suga T, et al. (2013) Systemic oxidative stress is associated with lower aerobic capacity and impaired skeletal muscle energy metabolism in patients with metabolic syndrome. Diabetes Care. 36(5): 1341-1346.
[Crossref] [Google Scholar]
- Brown KM, Arthur JR (2001) Selenium, Selenoproteins and human health: A review. Public Health Nutr. 4(2b): 593-599.
[Crossref] [Google Scholar]
- Bao BW, Kang Z, Zhang Y, Li Y, Xu R (2022) Selenium deficiency leads to reduced skeletal muscle cell differentiation by oxidative stress in mice. Biol Trace Elem Res.
[Crossref] [Google Scholar]
- Li S, Sun W, Zhang K, Zhu J, Jia X, et al. (2021) Selenium deficiency induces spleen pathological changes in pigs by decreasing selenoprotein expression, evoking oxidative stress, and activating inflammation and apoptosis. J Anim Sci Biotechnol. 12(1): 65.
[Crossref] [Google Scholar]
- Brigelius-Flohe R, Flohe L (2020) Regulatory phenomena in the glutathione peroxidase superfamily. Antioxid Redox Signal. 33(7): 498-516.
[Crossref] [Google Scholar]
- Fodor J, Al-Gaadi D, Czirjak T, Olah T, Dienes B, et al. (2020) Improved calcium homeostasis and force by selenium treatment and training in aged mouse skeletal muscle. Sci Rep. 10(1): 1707.
[Crossref] [Google Scholar]
- Jurynec MJ, Xia R, Mackrill JJ, Gunther D, Crawford T, et al. (2008) Selenoprotein N is required for ryanodine receptor calcium release channel activity in human and zebrafish muscle. Proc Natl Acad Sci USA. 105(34): 12485-12490.
[Crossref] [Google Scholar]
- Castets P, Maugenre S, Gartioux C, Rederstorff M, Krol A, et al. (2009) Selenoprotein N is dynamically expressed during mouse development and detected early in muscle precursors. BMC Dev Biol. 9(1): 1-12.
[Crossref] [Google Scholar]
- Arbogast S, Beuvin M, Fraysse B, Zhou H, Muntoni F, et al. (2009) Oxidative stress in SEPN1-related myopathy: From pathophysiology to treatment. Ann Neurol. 65(6): 677-686.
[Crossref] [Google Scholar]
- Kim JW, Kim R, Choi H, Lee S, Bae G (2021) Understanding of sarcopenia: From definition to therapeutic strategies. Arch Pharm Res. 44(9-10): 876-889.
[Crossref] [Google Scholar]
- Walston JD (2012) Sarcopenia in older adults. Curr Opin Rheumatol. 24(6): 623-627.
[Crossref] [Google Scholar]
- Vasilaki A, Mansouri A, Remmen HV, Meulen JHV, Larkin L (2006) Free radical generation by skeletal muscle of adult and old mice: Effect of contractile activity. Aging cell. 5(2): 109-117.
[Crossref] [Google Scholar]
- Palomero J, Vasilaki A, Pye D, McArdle A, Jackson MJ (2013) Aging increases the oxidation of dichlorohydrofluorescein in single isolated skeletal muscle fibers at rest, but not during contractions. Am J Physiol Regul Integr Comp Physiol. 305(4): R351-R358.
[Crossref] [Google Scholar]
- Meng SJ, Yu LJ (2010) Oxidative stress, molecular inflammation and sarcopenia. Int J Mol Sci. 11(4): 1509-1526.
[Crossref] [Google Scholar]
- Bua E, Johnson J, Herbst A, Delong B, McKenzie D, et al. (2006) Mitochondrial DNA-deletion mutations accumulate intracellularly to detrimental levels in aged human skeletal muscle fibers. Am J Hum Genet. 79(3): 469-480.
[Crossref] [Google Scholar]
- Fulle S, Protasi F, Tano GD, Pietrangelo T, Beltramin A, et al. (2004) The contribution of reactive oxygen species to sarcopenia and muscle ageing. Exp Gerontol. 39(1): 17-24.
[Crossref] [Google Scholar]
- Sullivan-Gunn MJ, Lewandowski PA (2013) Elevated hydrogen peroxide and decreased catalase and glutathione peroxidase protection are associated with aging sarcopenia. BMC Geriatr. 13:104.
[Crossref] [Google Scholar]
- Dargelos E, Brule C, Stuelsatz P, Mouly V, Veschambre P, et al. (2010) Up-regulation of calcium-dependent proteolysis in human myoblasts under acute oxidative stress. Exp Cell Res. 316(1): 115-125.
[Crossref] [Google Scholar]
- Dargelos E, Brule C, Combaret L, Hadj-Sassi A, Dulong S, et al. (2007) Involvement of the calcium-dependent proteolytic system in skeletal muscle aging. Exp Gerontol. 42(11): 1088-1098.
[Crossref] [Google Scholar]
- Terentyev D, Gyorke I, Belevych AE, Terentyeva R, Sridhar A, et al. (2008) Redox Modification of Ryanodine Receptors Contributes to Sarcoplasmic Reticulum Ca2+ Leak in Chronic Heart Failure. Circ Res. 103(12): 1466-1472.
[Crossref] [Google Scholar]
- Viner RI, Ferrington DA, Huhmer AFR, Bigelow DJ, Schoneich C (1996) Accumulation of nitrotyrosine on the SERCA2a isoform of SR Ca-ATPase of rat skeletal muscle during aging: A peroxynitrite-mediated process? FEBS Lett. 379(3): 286-290.
[Crossref] [Google Scholar]
- Klebl BM, Ayoub AT, Pette D (1998) Protein oxidation, tyrosine nitration, and inactivation of sarcoplasmic reticulum Ca2+-ATPase in low-frequency stimulated rabbit muscle. FEBS Lett. 422(3): 381-384.
[Crossref] [Google Scholar]
- Smuder AJ, Kavazis AN, Hudson MB, Nelson WB, Powers SK (2010) Oxidation enhances myofibrillar protein degradation via calpain and caspase-3. Free Radic Biol Med. 49(7): 1152-1160.
[Crossref] [Google Scholar]
- Jang YC, Rodriguez K, Lustgarten MS, Muller FL, Bhattacharya A, et al. (2020) Superoxide-mediated oxidative stress accelerates skeletal muscle atrophy by synchronous activation of proteolytic systems. Geroscience. 42(6): 1579-1591.
[Crossref] [Google Scholar]
- Du J, Wang X, Miereles C, Bailey JL, Debigare R, et al. (2004) Activation of caspase-3 is an initial step triggering accelerated muscle proteolysis in catabolic conditions. J Clin Invest. 113(1): 115-123.
[Crossref] [Google Scholar]
- Capel F, Buffiere C, Mirand PP, Mosoni L (2004) Differential variation of mitochondrial H2O2 release during aging in oxidative and glycolytic muscles in rats. Mech Ageing Dev. 125(5): 367-373.
[Crossref] [Google Scholar]
- Capel F, Rimbert V, Lioger D, Diot A, Rousset P, et al. (2005) Due to reverse electron transfer, mitochondrial H2O2 release increases with age in human vastus lateralis muscle although oxidative capacity is preserved. Mech Ageing Dev. 126(4): 505-511.
[Crossref] [Google Scholar]
- Ahn B, Ranjit R, Kneis P, Xu H, Piekarz KM, et al. (2022) Scavenging mitochondrial hydrogen peroxide by peroxiredoxin 3 overexpression attenuates contractile dysfunction and muscle atrophy in a murine model of accelerated sarcopenia. Aging Cell. 21(3): e13569.
[Crossref] [Google Scholar]
- Tan PL, Shavlakadze T, Grounds MD, Arthur PG (2015) Differential thiol oxidation of the signaling proteins Akt, PTEN or PP2A determines whether Akt phosphorylation is enhanced or inhibited by oxidative stress in C2C12 myotubes derived from skeletal muscle. Int J Biochem Cell Biol. 62: 72-79.
[Crossref] [Google Scholar]
- Lawler JM, Song W, Demaree SR (2003) Hindlimb unloading increases oxidative stress and disrupts antioxidant capacity in skeletal muscle. Free Radic Biol Med. 35(1): 9-16.
[Crossref] [Google Scholar]
- Gram M, Vigelso A, Yokota T, Helge JW, Dela F, et al. (2015) Skeletal muscle mitochondrial H2O2 emission increases with immobilization and decreases after aerobic training in young and older men. J Physiol. 593(17): 4011-4027.
[Crossref] [Google Scholar]
- Kumar A, Tahimic CGT, Almeida EAC, Globus RK (2021) Spaceflight modulates the expression of key oxidative stress and cell cycle related genes in heart. Int J Mol Sci. 22(16): 9088.
[Crossref] [Google Scholar]
- Steinbacher P, Eckl P (2015) Impact of oxidative stress on exercising skeletal muscle. Biomolecules. 5(2): 356-377.
[Crossref] [Google Scholar]
- Powers SK, Hudson MB, Nelson WB, Talbert EE, Min K, et al. (2011) Mitochondria-targeted antioxidants protect against mechanical ventilation-induced diaphragm weakness. Crit Care Med. 39(7): 1749-1759.
[Crossref] [Google Scholar]
- Talbert EE, Smuder AJ, Min K, Kwon OS, Szeto HH, et al. (2013) Immobilization-induced activation of key proteolytic systems in skeletal muscles is prevented by a mitochondria-targeted antioxidant. J Appl Physiol. 115(4): 529-538.
[Crossref] [Google Scholar]
- Wall BT, Snijders T, Senden JMG, Ottenbros CLP, Gijsen, et al. (2013) Disuse impairs the muscle protein synthetic response to protein ingestion in healthy men. J Clin Endocrinol Metab. 98(12): 4872-4881.
[Crossref] [Google Scholar]
- Powers SK, Smuder AJ, Criswell DS (2011) Mechanistic links between oxidative stress and disuse muscle atrophy. Antioxid Redox Signal. 15(9): 2519-2528.
[Crossref] [Google Scholar]
- Mukai R, Matsui N, Fujikura Y, Matsumoto N, Hou DX, et al. (2016) Preventive effect of dietary quercetin on disuse muscle atrophy by targeting mitochondria in denervated mice. J Nutr Biochem. 31: 67-76.
[Crossref] [Google Scholar]
- Bodine SC, Baehr LM (2014) Skeletal muscle atrophy and the E3 ubiquitin ligases MuRF1 and MAFbx/atrogin-1. Am J Physiol Endocrinol Metab. 307(6): E469-E484.
[Crossref] [Google Scholar]
- Uchida T Sakashita Y, KitahataK, Yamashita Y, Tomida C, et al. (2018) Reactive oxygen species upregulate expression of muscle atrophy-associated ubiquitin ligase Cbl-b in rat L6 skeletal muscle cells. Am J Physiol Cell Physiol. 314(6): C721-C731.
[Crossref] [Google Scholar]
- Nakao R, Hirasaka K, Goto J, Ishidoh K, Yamada, et al. (2009) Ubiquitin ligase Cbl-b is a negative regulator for insulin-like growth factor 1 signaling during muscle atrophy caused by unloading. Mol Cell Biol. 29(17): 4798-4811.
[Crossref] [Google Scholar]
- Pomies P, Blaquiere M, MauryJ, Mercier J, Gouzi F, et al. (2016) Involvement of the foxo1/murf1/atrogin-1 signaling pathway in the oxidative stress-induced atrophy of cultured chronic obstructive pulmonary disease myotubes. PLoS One 11(8): e0160092.
[Crossref] [Google Scholar]
- Smuder AJ, SollanekKJ, Nelson BW, Min K, Talbert EE, et al. (2018) Crosstalk between autophagy and oxidative stress regulates proteolysis in the diaphragm during mechanical ventilation. Free Radic Biol Med. 115: 179-190.
[Crossref] [Google Scholar]
- McClung JM, Judge AR, Powers SK, Yan Z (2010) p38 MAPK links oxidative stress to autophagy-related gene expression in cachectic muscle wasting. Am J Physiol Cell Physiol. 298(3): C542-C549.
[Crossref] [Google Scholar]
- Talbert EE, Smuder AJ, Min K, Kwon OS, Powers SK (2013) Calpain and caspase-3 play required roles in immobilization-induced limb muscle atrophy. J Appl Physiol. 114(10): 1482-1489.
[Crossref] [Google Scholar]
- Tidball JG, Spencer MJ (2002) Expression of a calpastatin transgene slows muscle wasting and obviates changes in myosin isoform expression during murine muscle disuse. J Physiol 545(3): 819-828.
[Crossref] [Google Scholar]
- McClung JM, Judge AR, Talbert EE, Powers SK (2009) Calpain-1 is required for hydrogen peroxide-induced myotube atrophy. Am J Physiol Cell Physiol 296(2): C363-C371.
[Crossref] [Google Scholar]
- Min K, Smuder AJ, Kwon OS, Kavazis AN, Szeto HH, et al. (2011) Mitochondrial-targeted antioxidants protect skeletal muscle against immobilization-induced muscle atrophy. J Appl Physiol. 111(5): 1459-1466.
[Crossref] [Google Scholar]
- Eshima H, Siripoksup P, Mahmassani ZS, Johnson JM, Ferrara PJ, et al. (2020) Neutralizing mitochondrial ROS does not rescue muscle atrophy induced by hindlimb unloading in female mice. J Appl Physiol. 129(1): 124-132.
[Crossref] [Google Scholar]
- Schakman O, Kalista S, Barbe C, Loumaye A, Thissen JP (2013) Glucocorticoid-induced skeletal muscle atrophy. Int J Biochem Cell Biol. 45(10): 2163-2172.
[Crossref] [Google Scholar]
- Kim YI, Hyunjung L, Farida SN, Seo HD, Ha TY, et al. (2022) Antioxidant activity of valeriana fauriei protects against dexamethasone-induced muscle atrophy. Oxid Med Cell Longev. 2022: 3645431.
[Crossref] [Google Scholar]
- Oh S, Chang HC, Lee BJ, Park JH, Son Kh, et al. (2021) Fermented oyster extract attenuated dexamethasone-induced muscle atrophy by decreasing oxidative stress. Molecules. 26(23): 7128.
[Crossref] [Google Scholar]
- Ohtsuka A, Hiroshi K, Takayuki O, Kunioki H (1998) Vitamin E reduces glucocorticoid-induced oxidative stress in rat skeletal muscle. J Nutr Sci Vitaminol. 44(6): 779-786.
[Crossref] [Google Scholar]
- Mitsui T, Yoshifumi U, Masakazu N, Masashi A, Kenji A, et al. (2002) Mitochondrial damage in patients with long-term corticosteroid therapy: Development of oculoskeletal symptoms similar to mitochondrial disease. Acta Neuropathol. 104(3): 260-266.
[Crossref] [Google Scholar]
- Konno S (2005) Hydroxyl radical formation in skeletal muscle of rats with glucocorticoid-induced myopathy. Neurochem Res. 30(5): 669-675.
[Crossref] [Google Scholar]
- Ulla A, Takayuki U, Yukari M, Kosuke S, Atsushi H, et al. (2021) Morin attenuates dexamethasone-mediated oxidative stress and atrophy in mouse C2C12 skeletal myotubes. Arch Biochem Biophys. 704: 108873.
[Crossref] [Google Scholar]
- Espinoza MB, Jorge EA, Rodrigo Z, Cristian V, Alfredo M, et al. (2017) Cortisol induces reactive oxygen species through a membrane glucocorticoid receptor in rainbow trout myotubes. J Cell Biochem. 118(4):718-725.
[Crossref] [Google Scholar]
- Siuda D, Silke T, Alma R, Ning X, Ulrich F, et al. (2014) Dexamethasone upregulates Nox1 expression in vascular smooth muscle cells. Pharmacology. 94(1-2): 13-20.
[Crossref] [Google Scholar]
- Chen C, Yang JS, Lu CC, Chiu YJ, Chen HC, et al. (2020) Effect of quercetin on dexamethasone-induced C2C12 skeletal muscle cell injury. Molecules. 25(14): 3267.
[Crossref] [Google Scholar]
- Huang Y, Ka C, Qingbo R, Long Y, Jundong Z, et al. (2018) Dihydromyricetin attenuates dexamethasone-induced muscle atrophy by improving mitochondrial function via the pgc-1a pathway. Cell Physiol Biochem. 49(2): 758-779.
[Crossref] [Google Scholar]
- Liu J, Yunhua P, Xun W, Yingying F, Chuan Q, et al. (2016) Mitochondrial dysfunction launches dexamethasone-induced skeletal muscle atrophy via AMPK/FOXO3 signaling. Mol Pharm. 13(1): 73-84.
[Crossref] [Google Scholar]
- Seo E, Truong CS, Jun HS (2022) Psoralea corylifolia L. seed extract attenuates dexamethasone-induced muscle atrophy in mice by inhibition of oxidative stress and inflammation. J Ethnopharmacol. 296: 115490.
[Crossref] [Google Scholar]
Citation: Ulla A, Nikawa T (2023) Regulatory and Pathophysiological Roles of Reactive Oxygen Species in Skeletal Muscle. Biochem Mol Biol J. 9:31.
Copyright: © 2023 Ulla A, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.





