Keywords
Mareka district; Prevalence; Packed cell volumes; Small ruminants; Trypanosomosis
Abbreviations
PCV: Packed Cell Volumes; CSA: Central Statistical Authority; FAO: Food and Agricultural Organization; AAU: Addis Ababa University; FVM: Faculty of Veterinary Medicine; NTTICC: National Tsetse and Trypanosomosis Investigation and Control Center; ILRI: International Livestock Research Institute; UK: United Kingdom; SNNPRS: Southern Nation, Nationalities and People Regional State
Introduction
Tsetse transmitted trypanosomosis affect 37 sub-Saharan countries of Africa. An estimate of 160 million cattle and 200 million sheep and goats are kept in this area of risk extending over 10 million Km2 of land [1]. In susceptible cattle breeds, the disease reduces calving by up to 20% and causes the death of another 20% of young stock. Ethiopia possesses the largest livestock population in Africa with its greatest variation in climate and physiographic factors. Among them 10-14 million heads of cattle and an equivalent number of equine, 18 million goats and 24 million sheep are exposed to the risk of acquiring trypanosomosis [2]. Morbidity and mortality losses from ruminant livestock alone are estimated to be USD 200 million [3].
Small ruminants become attractive assets for development because of low cost of production, requirement of little land and higher profitably. Small ruminants are kept in a very broad range of agro-ecological zones such as hot and dry condition and provide a golden opportunity to alternatively exploit potential of lowland areas [4,5]. Sheep and goats play a crucial role in providing protein (milk and meat) and non-food commodities (manure and hides). They equally serve as cash reserves and form of saving for a rural population and as a protection against agricultural failure; therefore, the number of animals is more important than individual productivity [6,7].
In spite of the presence of large number of small ruminant population in Ethiopia and it fails to utilize expected productivity due to many factors. Among the many factors which limit these economic returns from small ruminants is diseases that stand in the front line. One of those diseases which affect small ruminants is Trypanosomosis. This disease is the most devastating disease in terms of poverty and loss of agricultural production [8]. These losses include reduction of herd size, reduced market value due to loss in body condition, drop in milk production, drop in lambing rate and prevention of mixed farming [9].
Trypanosomosis is a parasitic disease caused by unicellular flagellated protozoa belonging to the genus Trypanosoma which in general is found in the blood stream, various body tissues and fluids of vertebrate hosts throughout the world [10,11]. The Genus Trypanosoma belongs to the order Kinetoplastidia, family Trypanosomatidea. Generally trypanosomes are classified into three main subgenera of economic and human health importance: Vivax (Duttonella), congolense (Nannomonas) and Brucei (Trypanozoon) [12]. Its mode of transmission is cyclically by tsetse flies (Glossina species) and mechanically by biting flies (Tabanids, Stomoxys etc). Tsetse transmitted trypanosomes species namely: Trypanosoma congolense, T. vivax and T. brucei comprise the major disease agents that affect livestock including large and small ruminants [13]. The severity of the trypanosomosis disease determined by infected animal species and age and species of trypanosomes involved in the disease process. Hence, pathogenesis of African trypanosomosis differs according to the species causing the infection. T. vivax and T. congolense appear to be strictly parasites of the blood plasma and produce tissue injury through the development of anemia. T. brucei is more widely distributed in the host affecting the intercellular fluids of the body cavities [14]. In T. brucei anemia is considered to be of secondary importance to extensive degenerative, necrotic and inflammatory changes. Regardless of the species of trypanosomes and host, the principal clinical signs are intermittent fever, progressive anemia and loss of condition. Also, the superficial lymph nodes are enlarged and prominent [15-17].
Trypanosomosis of cattle (nagana) is recognizable as a serious problem, but the situation with regard to small ruminant is less clear. Small ruminants, especially goats have been reported to be resistant to trypanosomes [18]. However, several studies on the prevalence infections of small ruminant trypanosomosis in Ethiopia revealed that these animals can acquire infections that result in economic losses. Though, these animals do not show significant signs of trypanosomosis, they could however act as a carrier of infection and endanger other livestock species and human beings [9]. Even trypanosomosis has long been reported, there is Scanty of information on the prevalence and effect of trypanosomes of sheep and goats in and around Mareka District of Dawro Zone, Southwestern Ethiopia.
Therefore, this study was conducted with the following objectives to:
• Determine the prevalence of trypanosomosis in small ruminants
• Assess the effects of trypanosome infection, in terms of packed cell volume (PCV) in sheep and goats.
Materials and Methods
Study area
The study was conducted from November 2009 to April 2010 at Mareka District, Dawro Zone. Mareka district is bounded with Loma district from North, Gana Bosa District from East, Tocha district from south and Isra district from west. The area is located 512 kms from Addis Ababa in the southwest of the country. The study area extends 6°37’ to 7°15’N Latitude and 38°12’ to 38°24’ longitude. Mareka district has an altitude range of 700-3000 meters above sea level with uni-modal rain fall. The yearly mean rainfall oscillates, between 1100 mm and 2050 mm distributed over all year months. The annual mean minimum and maximum temperature varies between 15.5°C to 35°C [19].
Also, the study area has about 114,508 cattle, 37,999 small ruminants, 26,655 poultry and 3,374 equine populations [19]. Totally, the topography of the served district is with steep slopes and chained mountains land features in case of highland whereas in case of lowland is almost flat plains and steeply hills and some typical slopes. The natural vegetation such as forest, shrub land, savanna grass land, Acacia Abyssinica, Piliotigma thonnigii and phonix are the dominant ones. Wild animals such Bush pig and green monkey are the common wild animals which are found in the study area.
Study animals
The study was carried out from November 2009 to April 2010 on 400 animals (222 sheep and 178 goats) selected by simple random sampling methods in the study area. Information on the sex, species and ages was obtained at the same time when the sample was collected.
Study design
Cross-sectional study design was used on sheep and goats, and the sample size was determined using 50% expected prevalence (no previous data for the area) and 95% precision [20]. Accordingly the sample size was expected to be 384 animals. For convenience, a total of 400 animals comprising 178 goats and 222 sheep were included in this study. Animals were grouped according to sex and age.
Sample collection
After obtaining informed consents of the owners on the purpose of the study, blood samples were collected from each animal by ear vein puncture using a sterile lancet in to a pair of heparinized microhematocrit centrifuge capillary tubes (75 × 1.2 mm). Each tube was filled until about three quarters of its length and one end sealed with crystal seal [21,22].
Measurement of Packed cell volume (PCV)
To get a valuable indication on the presence and degree of anemia, PCV was determined after centrifugation of the blood samples in capillary tubes at 12,000 rpm, for 5 minutes. PCV value was read directly by using Hawksley microhematocrit reader, which could be adjusted individually for the length of blood column as the percentage of the total volume in each tube [23].
Parasitological examination
Buffy coat examination: The centrifuged capillary tube after used for PCV determination was cut with a diamond a pointed per 1 mm below the buffy coat to include the upper most layer of RBC, and 3 mm above to include the plasma. The contents were gently expressed onto a microscope slide and covered with a cover slip (22 × 22 mm). The preparation was then examined using an X10 eye piece in combination with an X40 objective using the dark ground buffy coat examination technique [21]. Identification of trypanosomes species was made on the basis of movement behavior patterns observed in the buffy coat wet film preparation as follows:
Trypanosoma congolense: Recognized by its small size in relation to red blood cell (RBC) diameter, its sluggish activity and its invariable attachment to RBCs.
Trypanosoma vivax: Large and strinkgly apparent by the speed with which it whirred and transversed the microscopic field [10,24].
Trypanosoma brucei: Various sizes, rapid movement in a confined areas; undeviating membrane traps the light in to ‘pockets’ moving along body.
Thin smear examination: When samples were found positive using the buffy coat technique, thin blood films were done on non-centrifuged blood samples for further species identification on trypanosome morphology. Air dried thin blood films were fixed with absolute methanol for 3-5 minute, and stained by immersing into a staining jar containing diluted Geimsa (1:20) solution for 30-45 minutes . Stained blood smears were then rinsed in distilled water, air dried and examined for better appreciation of trypanosome species identification based on morphology under oil immersion objective [22]. The following morphological characteristics were observed for species identification:
Trypanosoma congolense: Appreciated with inconspicuous undulating membranes, medium sized kinetoplast which is located marginally and blunt posterior end.
Trypanosoma vivax: Observed as somewhat longer size with inconspicuous undulating membrane, the large kinetoplast is found terminally and the posterior end is broad and rounded, short free flagellum presents [25].
Trypanosoma brucei: Undulating membrane is conspicuous, free flagellum present at the anterior end, posterior end pointed, kinetoplast sub-terminal.
Data Analysis
After entering all the information on excel spreadsheet, data were analyzed using chi-square statistics for comparison of prevalence of trypanosomosis between sheep and goat, male and female and between different age groups. T-test was used for comparison of PCV means on SPPS 15.0 software.
Result
General observation during the study time showed that very few animals have demonstrated clinical signs of trypanosomosis.
Prevalence of tryanosomosis
Based on buffy coat examination technique, the overall prevalence of trypanosomes was 9% in goats and 7.2% in sheep. No statistically significant difference (P>0.05) between sheep and goat in the prevalence of tryanosomosis. Based on movement behavior in buffy coat and morphology in thin smear, only two cases of Trypanosoma vivax were observed (one in sheep and one in goats), null infection of T. brucei and all the rest were due to T. congolense. On the other hand, in both sheep and goats, the prevalence of trypanosomes was significantly higher in females than in male animals (Figure 1).
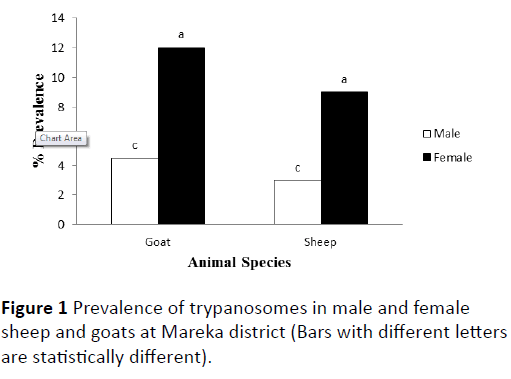
Figure 1: Prevalence of trypanosomes in male and female sheep and goats at Mareka district (Bars with different letters are statistically different).
Animals were also grouped according to their age (<2 years and ≥ 2 years). No significant difference in the prevalence of trypanosomosis was observed between animals <2 years and ≥ 2 years of age (Figure 2).
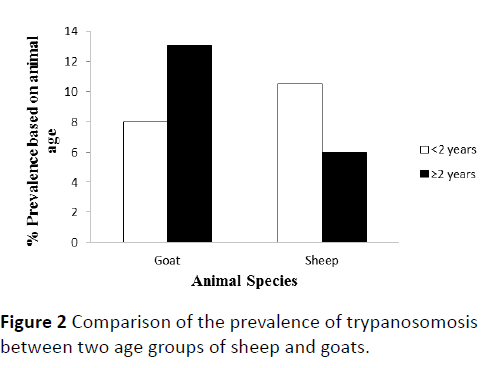
Figure 2: Comparison of the prevalence of trypanosomosis between two age groups of sheep and goats.
Packed cell volume (PCV) values
In both sheep and goats, trypanosome positive animals had significantly lower mean PCV than those negative animals (P<0.001). Mean PCVs for positive and negative sheep were 23.9 ± 2.4% and 26.3 ± 4.1%, respectively. Similarly, Mean PCVs for positive and negative goats were 23.9 ± 2.6% and 27.4 ± 4.1%, respectively. On the other hand, comparison of mean PCVs between trypanosome-positive sheep and trypanosome-positive goats did not show statistical difference (Figure 3). On the other hand, among the trypanosome positive animals, 94% of sheep and 25% of goats had PCV values below the normal range (27-45% for sheep and 24-38% for goats) described in Radostitis et al. [10] while, among trypanosome negative animals, 47% of sheep and 15% of goats had PCV values below normal.
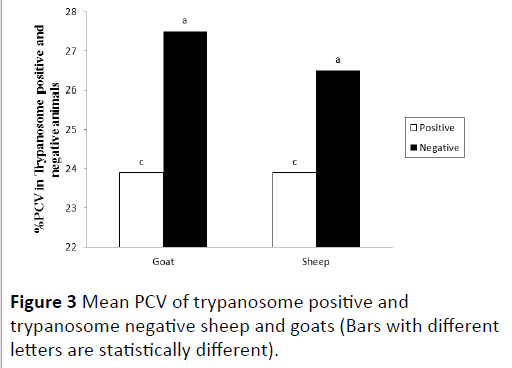
Figure 3: Mean PCV of trypanosome positive and trypanosome negative sheep and goats (Bars with different letters are statistically different).
Discussion
This study was undertaken with the objectives to show the prevalence of trypanosomosis in sheep and goat and see if the parasite causes significant anemia in those affected animals. The findings revealed an overall prevalence of 8% in small ruminants at Mareka District, Dawro Zone, Southwestern Ethiopia. It was significantly higher than the result previously reported by Dinka [26] for Didesa and Gibe Valley (5.1%) and Shukure [27] for Wolaita Zone (5.45%).
In the present investigation, there was no statistically significant difference in the prevalence of trypanosomosis between the two species of hosts even though it tends to be higher (9%) in goats than (7.2%) in sheep. On the contrary, Shukure [27] reported a 7.79% and 2.18% prevalence of typanosomosis in goats and sheep respectively in Wolaita. Similarly, Ahmadin [28] in Wolaita and Tekele and Getachew [29] in Gamogofa respectively reported 6.3% and 31% in goats but none in sheep.
The high incidence of trypanosomosis in goats in the previous studies may be attributed to the feeding habit of tsetse. It has been reported that caprine trypanosomiasis is more common in East than in West Africa. This has been attributed to differences in feeding preferences between riverine and savannah species of Glossina, the latter being more inclined to feed on goats [30]. The second reason might be the difference in hair coat structure in that sheep have more body hair coverage than goats which in turn reduces the risk of exposure to the bite of tsetse or other non-cyclical trypanosomosis transmitter arthropods. The third reason could be due to the feeding habit of goats as they are browser, goats cross the savannah and riverine areas where tsetse is highly infested with objective of looking for leaves this exposes to the trypanosomosis infection. None of these explanations could justify the relatively high incidence of trypanosomosis in goats compared to the previous findings. On the other hand, Ameen et al. [31] in Nigeria reported a prevalence of 4.7%, 3.5% and 3.9% T. congolense infection in sheep, goat and cattle respectively by using wet and stained thin blood film; techniques with lower sensitivity than the Buffy coat method [16]. Similarly relatively higher prevalence of trypanosomes in sheep (8.7%) than in goats (4.1%) was reported in Northwest Ethiopia [32].
In the study area T. congolense was the dominant species with both techniques of buffy coat and stained thin blood film examinations. Abebe and Jobre [3] described the predominance of T. congolense as it indicates the increased contact of animals with tsetse vector and the lesser importance of biting flies in the transmission of trypanosomosis in cattle. It may also suggest that sheep and goats are more preferred hosts by tsetse than by other biting flies. The findings of the current study are in accord with the above reports because infection by T. Congolese accounts the highest proportion (93.75%) both in sheep and goats. In addition, the highest infection rate by the T. congolense and T. vivax in small ruminants in this study agrees with the reports of Uilenberg [23] who concluded that T. congolense and T. vivax are the possible species of trypanosomes responsible for severe disease in cattle, sheep and goats. On the other hand, Kebede et al. [32] indicated that T. vivax was the dominate species followed by T. congolense and T. brucei in sheep and goats in Northwest Ethiopia (Awi Zone) suggesting the significance of mechanical transmission even though cyclical transmission could not be ruled out. Detection of trypanosomes in small ruminants and pigs using the more advanced technique, PCR, in western Kenya has also shown prevalence of 25.3% in sheep, 20% in goat and 19.2% in pig but no statistical difference between the host species [33]. This may suggest that the prevalence rate reported in our study by using buffy coat technique though better than other parasitological techniques may have undermined the true extent of the problem in the area.
There was no significant difference in infection rate between small ruminants below two years and those above two years of age although it tends to be lower in younger goats and adult sheep. Reduced incidence of trypansomosis infection in young animals has been noted by Leak [34], Murray and Dexter [35] and Bealby et al. [36], who conclude that young ruminants are less exposed to trypanosome infections than adult animals due to lack of long association of young animals with the vectors of trypanosomosis. It may also be attributed to the chronicity of the disease that obscures detection of trypanosomes and phenomenon of self-cure in older animals [34,37,38].
The findings in this study have shown that females have higher infection rates 9% and 11.7% in sheep and goat respectively than males 3% and 4.48% in sheep and goat, respectively. Similar results were reported by Samdi et al. [39] where female small ruminants exhibited higher incidence of trypanosome infection than males. Animals were sampled irrespective of their physiological status. It has been reported that pregnant and lactating animals are more susceptible to parasitic diseases than non-pregnant and non-lactating animals [40]. Furthermore, the sample composition shows that more than two-third of the animals are females kept for breeding purposes. Therefore, such physiological status of the animals might have contributed for the higher prevalence of trypanosomes in female sheep and goats in the study area.
The damage of tissues due to trypanosomosis is probably multifactorial in etiology, but the main underlying feature is the progressive anaemia throughout the course of the disease. The cause of anemia is complex, but it is believed to be hemolytic, caused primarily by erythrophagocytosis due to stimulation and expansion of the monoculear phagocytic systems [12]. This study clearly revealed that trypansomosis causes considerable anaemia in sheep and goats. This was ascertained by the significantly lower PCV and high proportion of parasitemic small ruminants with pale mucous membrane (data not shown) than those without detectable trypanosomes. The overall mean PCV value of parasitemic sheep is significantly lower than those of the aparasitaemic sheep and goats. The normal PCV value of sheep and goats ranges from 27-45% and 24-38%, respectively [10]. Significant proportion of infected sheep and surprisingly lower number of goats showed depression in PCV at the time of observation. This finding is partly in agreement with the statement of Uilenberg [23] that trypanosomosis causes anemia in cattle, sheep and goats. Furthermore, Kebede et al. [32] and Samdi et al. [39] have also reported significant reduction of PCV in parasitemic animals. However, the presence of infected sheep and mainly goats with PCV value in a normal range indicates that PCV value alone could not be considered as diagnostic criteria for trypansomosis. Similarly, the presence of aparasitemic animals with PCV values in normal ranges may show the low sensitivity of the diagnostic test in chronically infected animals and/or the existence of other causes of anemia (other diseases and nutritional deficiencies).
Conclusion and Recommendation
This research work was intended to obtain data on the prevalence and pathogenic effect of trypanosomosis in small ruminants. It also addressed the effect of the disease in terms of the development of anemia. Accordingly, significant number of sheep and goats were found equally infected with mainly the cyclically transmitted trypanosome, T. congolense. The infection has resulted in the development of anemia characterized by reduced PCV. Female animals appear to be more susceptible than males while all ages are equally susceptible. From the data it was observed that female animals are the most abundant at the study areas suggesting altogether that more attention be given to this section of the small ruminant population. Based on the above conclusion, the following recommendation can be forwarded for further consideration in research and implementation of control programs:
• Further research is required as to the factors contributing to susceptibility and pathogenic significance of the disease.
• Studies on the extent and distribution of the vectors and application of tsetse control programs should be of paramount importance.
• Any treatment and control program should take into account the physiological states of these animals.
• Sheep and goats should be frequently examined for the presence of trypanosomes in their blood and their role as reservoir hosts should be investigated.
Disclosure Statement
No potential conflict of interest was reported by the authors.
Funding
No funding body.
References
- Erkelens AM, Dwinger RH, Bedane B, Slingenbergn JHW, Wint W (2000) Selection of priority Areas for Tsetse control in Africa; A decision tool using GIS in Didessa Valley, Ethiopia, as a pilot study. In: animal Trypanosomes Diagnosis and Epidemology. FAO/IAEA coordinated Research program, Vienna, Austria.
- CSA (1998) Ethiopian agricultural sample enumeration 1996/1997. Central statistic Authority, Federal democratic republic of Ethiopia.
- Abebe G, Jobre Y (1996) Trypanosomosis: threat to cattle production in Ethiopia. Revue Med Vet 147: 879-902.
- Oyeyemi M (2002) Response of multiparous primiparous West Africa Dwarf goat to Concentrate supplementation. Veterinarsiki Archiv 72: 29-38.
- Yibeltal M (2005) A seroprevalence of small ruminant Brucellosis in selected site of Afar and Somali region. DVM Thesis, Faculty of veterinary Medicine, Addis Ababa University, DebreZeit, Ethiopia.
- Pannin A, Mahbile M (1997) Profitability and household income contribution of Small ruminant to small scale farmers in Botswana. Small Ruminant Res 25: 9-15.
- Itty PP, Ankers J, Zinnsta, S, Trawally, Pfister K (1997) Productivity and profitability of sheep production in Gambia: Implication for livestock development in West Africa. J Intern Agric 36: 153-172.
- Hursey BS (2000) PAAT: The program against African Trypanosomosis. Trends Parasitol 4.
- Swallow BM (2000) Impacts of Trypanosomiasi on African Agriculture. TAAT Technical and Scientific Series, FAO, Rome.
- Radostitis OM, Gray CC, Blood DC, Hinchcliff KKI (2000) Disease causes by Trypanosomes. In: A text book of the diseases of cattle, sheep, pigs, goats and horse, W.B sounders Company Ltd.
- Finelle D (1983) African animal trypanosomosis. Part one-Disease and Chemotherapy. FAO World Animal Health Review,p:4.
- Seifert HSH (1996) Trypanosomes In: Horst Seifert. Tropical animal health, pordrecht/London, Kluwer academic Publisher,pp: 151-168.
- Mulligan HW (1970) The African Trypasonomosis. Georgs and Unwin Ltd, London.
- Robertson A (1976) Trypanosomiasis in Domestic Animals. In: Robertson A (ed.)Hand book on Animal Disease in the tropics. British veterinary Association, London, Overseas.
- Hall HTB (1985) Diseases and parasites of livestock in the tropics. Intermediate tropical Agricultures series, Longman London and New York, NY.
- Paris J, Murray M, Mcodim BF (1982) A comparative evaluation of the Parasitiological techniques currently available for the diagnosis of African Trypanosomiasis in cattle. Acta Trop 39: 307-316.
- Molyneux DM, Ashford RW (1983) The biology of trypanosome and leishmania Parasite of man and animals. Taylor and Francis,London.
- Kristjanson PM, Swallow BM, Rowland’s GJ (1999) Measuring the costs of animal Tryposomosis, the potential benefits to control and returns to research. Agr System 59: 75-98.
- NTTICC (2010) Annual report MOA. National tsetse and trypanosomosis control centre, Bedelle, Ethiopia.
- Thrusfield M (2005) Veterinary Epidemiology. London: Blackwell Publishing.
- Murray M (1977) An improved parasitological technique for the diagnosis of African Trypasonomosis. Trans R Soc Trop Med Hyg 74: 389-432.
- Hoare CA (1972) The trypanosomes of mammals. J Small Anim Pract 13: 671-672.
- Uilenberg G (1998) A field guide for the diagnosis, treatment and prevention of African Animal Trypanosomiasi. FAO of the United Nations,Rome,pp: 158.
- Fischer MS, Say RR (1989) Methods for studying Trypanosomosis and their Vectors. In: Fischer MS, Say R Manual of Tropical Veterinary Parasitology,pp: 251-270.
- Urquhart GM, Armour J, Duncan JL, Dunn AM, Jennings FW (1996) Veterinary Parassitology. Blackwell Science Scotland, Scotland,pp: 212-218.
- Dinka H (2003) Prevalence of Trypanosomosis in small ruminants in South West Ethiopia (Didessa and Ghibe Valleys). DVM Thesis, Faculty of Veterinary Medicine, Addis Ababa University, DebreZeit, Ethiopia.
- Shukure M (1995) Small ruminant trypanosomiasis: prevalence and Evaluation of field diagnostic Methods in Wolita. DVM Thesis, Faculty of Veterinary Medicine, Addis Ababa University, DebreZeit, Ethiopia.
- Ahmadin J (1986) Survey of Trypanosomosis in WolaitAwraja. DVM Thesis, Faculty of Veterinary Medicine, Addis Ababa University, DebreZeit, Ethiopia
- Tekele A, Getachew A (1988) A survery of Trypanosomiasi in Gamogofa area. Rev Elev Med Vet Parasit Trop 41: 271-276.
- Smith MC, Sherman DM (1994) Blood, Lymph and immune system. In: SmithMC, ShermanDM(eds.) Goat Medicine. Lea & FA biger,Philadelphia,pp: 193-230.
- Ameen SA, Joshua RA, Adedeji OS, Raheem AK, Akingbade AA, et al. (2008) Preliminary studies on prevalence of ruminant trypanosomosis in Ogbomoso Area of OYO state, Nigeria. J Sci Res 3: 214-218.
- Kebede N,Fetene T, Animut A (2009) Prevalence of trypanosomsis of small Ruminants in Guangua district of Awi Zone, North Western Ethiopia. J Infect Develop Countries 3: 245-24.
- Ng’ayo MO, Njiru ZK, Kenya EE, Muluv GM, Osir EO, et al. (2005) Detection of trypanosomes in small ruminants and pigs in Western Kenya; Important reservoirs in the Epidemiology of sleeping sickness. J kinetoplastid Biol Dis 4: 5.
- Leak SE (1996) Trypasonomosis: A veterinary prospective pergramon press, New York,p: 551.
- Murray M, Dexter TM (1988) Anemia in bovine African Trypanosomosis. Acta Tropica 45: 389-432.
- Bealby KA, Connor RJ, Rowlands GJ (1996) Trypansomosis in Goats in Zambia. ILRI, Nairobi, Kenya,p: 88.
- Getahun B (1987) The prevalence of small ruminant trypanosomosis in amogofa. DVM Thesis, Faculty of Veterinary Medicine, Addis Ababa University, DebreZeit, Ethiopia.
- Roede PL, Scott JM, Pergramon RG (1984) Acute Trypanosome vivax infections of Ethiopia cattle in the apparent absence of tsetse. Trop Anim Health Prod 16:141-149.
- Samdi S, Abegna JN, Fajinmi A, Kalgo A, Idowu T, et al. (2008) Seasonal Variation in Trypanosomiasi rates in small ruminants at the kudana Abattoir, Nigeria. Afr BiomedRes11: 229-232.
- Fleming MW, Conard SD (1989) Effects of exogenous progesterone and/or Prolactin on Haemonchuscontortus infections in ovariectomized ewes. Vet Parasitol 34: 57-62.