Wei Jie Nicholas Hong1*, Hui Ling Huang2, Kumudhini Rajasegaran3, Jean Yin Oh4, Siobhan Kelly5, Seyed Ehsan Saffari6 and Chu Shan Elaine Chew7
1Department of Pediatrics, Pediatric Resident, KKH, Singapore
2National University of Singapore, Yong Loo Lin School of Medicine, Medical Officer (Ministry of Health Holdings), Singapore
3General Pediatrics and Adolescent Medicine Service, KKH, Singapore
4Consultant in General Pediatrics and Adolescent Medicine Service, KKH, Singapore
1Department of Psychology, Senior Psychologist, KKH, Singapore
2Research Fellow in Center for Quantitative Medicine, Duke-NUS Medical School, Singapore
3Consultant in General Pediatrics and Adolescent Medicine Service, KKH, Singapore
Corresponding Author:
Wei Jie Nicholas Hong
Department of Pediatrics, Pediatric Resident
KKH, Singapore, 100 Bukit Timah Road
Block 272A Sengkang Central #13-307 Singapore 541272, 229899
Tel: +65-90119201
E-mail: nickhong92@hotmail.com
Received Date: April 24, 2017; Accepted Date: May 26, 2017; Published Date: May 29, 2017
Citation: Nicholas HWJ, Ling Huang H, Rajasegaran K, et al. (2017) Presence of Obesity Related Comorbidities Associated with Lower Attrition Rate in Pediatric Weight Management Program. J child Obes 2:10 doi:10.21767/2572-5394.100031
Copyright: © 2017 Nicholas HWJ, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Keywords
Childhood obesity; Pediatric obesity service; Outcome; Body mass index; Lifestyle intervention
Abbreviations
BMI: Body Mass Index; OSA: Obstructive Sleep Apnea; NAFLD: Non-Alcoholic Fatty Liver Disease
Introduction
Global childhood obesity is increasing at an alarming rate in the 21st century [1]. In Asia, the estimated prevalence of childhood obesity is estimated at 4.9% in 2010 and is estimated to increase to 6.8% in 2020. However in absolute numbers, Asia has the highest number of children with obesity because more than half (approximately 18 million in 2010) live in this region [2]. Singapore is a good platform to study pediatric obesity in Asia with the majority representation from three main ethnicities: East Asians represented by the Chinese majority, South East Asians by the Malays and the South Asians by the Indians accounting for 74%, 13% and 9% of the population respectively. Over the past decade, the prevalence of obesity in Singapore has increased 0.7 percentage point a year since 2004 to reach nearly 11% in 2010 [3]. The total proportion of overweight students in Singapore schools has dropped from 11.1% in 1994 to 9.5% in 2007 but the percentage of children with severe obesity has seen a rise from 2.7% to 3.6% [4]. Adolescents who are overweight or obese have a moderate to substantial risk of remaining so in adulthood and they suffer from a range of immediate and long term medical and psychosocial complications [5,6].
The paediatric weight management program at KK Women’s and Children’s Hospital was developed in response to the rising rates of childhood obesity and associated obesity related comorbidities. Studies on paediatric weight management clinics have reported varied long-term outcomes in reduction of BMI ZScore in their patients with high attrition rate thus limiting generalizability of the results. Predictive factors for attrition in weight management clinics have also reported varied factors [7]. There is no literature looking at the outcome and attrition of the paediatric weight management program locally and in Asia. The aim of this study is to determine whether the pediatric weight management program effectively reduces Body Mass Index (BMI) Z-Scores and cardiovascular risk factors in children with obesity aged 2-16 years old pre post intervention and to examine the factors that predict attrition from the program.
Subjects, Materials and Methods
This is a single center retrospective study reviewing the medical records of patients enrolled in the pediatric weight management program between 1st January 2009 to 29th February 2010. All patients enrolled in the pediatric weight management program in the above time period presenting with Body Mass Index (BMI) above 90th percentile were included in the study. Patients with secondary causes for weight gain, such as uncontrolled thyroid disease or genetic syndrome associated with obesity were excluded. The study was approved by the Singhealth Centralized Institutional Review Board. In this retrospective review of the study institution’s pediatric obesity service, the following observations regarding its protocol and procedures were made:
Observations of the pediatric weight management program protocols and procedures
Medical consultation
Criteria for referral to pediatric obesity service included a Body Mass Index (BMI) above the 90th percentile based on the Centers for Disease Control and Prevention (CDC) growth chart [8]. Children and adolescents with their parents/guardians were seen by a pedatrician on the first visit to assess the severity of exogenous obesity, identify risk factors and screen for complications.
Mechanical complications of obesity screened included obstructive sleep apnea (OSA) where a colloborative history was obtained from parents of patients for symptoms of OSA, previous sleep studies and interventions if any. Metabolic screen for diabetes mellitus, dyslipidaemia and Nonalcoholic fatty liver (NAFLD) were recommended based on U.S. Preventive Services Task Force recommendations [9].
Patients were reviewed every one to six monthly depending on the paediatrician assessment. Patients were also routinely referred to the dieticians and exercise trainers for dietary and physical activity counselling respectively.
Physical activity and dietary assessment and counseling
Physical activity counselling was done by the exercise trainers according to the guidelines recommended by the Health Promotion Board [10]. A structured exercise program was available for patients who opt for an experiential intervention based on individual or group exercise program facilitated by exercise trainers.
Dietary recommendations and counseling for the child were done by dietitians according to the guidelines recommended by the Health Promotion Board [11].
Anthropometry and metabolic profile
Patients’ weight and height were measured at every clinic visit. Clinic assistants measured the height and weight of the subjects at each clinic visit. The height and weight were measured to the nearest 0.1 cm via a stadiometer (Seca, Model 220; Germany) and 0.1 Kg via a medical weighing scale (Tanita HD-316, Tanita Corp.; Tokyo, Japan) respectively without shoes and in light clothing. BMI was calculated as kg/m2 and BMI-Z Score was calculated using the L, M, S (lambda, mu, sigma) parameters published by the CDC [8]. Blood pressure was measured via an electronic sphygmomanometer (Dinamap model 8101, Critikon Inc.; Florida, USA). Metabolic blood tests (fasting lipid panel, oral glucose tolerance test) were collected after a minimum 10-hour fasting period and analyzed using standard automated techniques in the hospital’s laboratory. Hypertension is defined as systolic or diastolic blood pressure ≥ 95th percentile for age, sex and height [12]. Dyslipidemia is defined by high levels of total cholesterol (TC), low-density lipoprotein (LDL) and triglycerides (TG) with lower levels of highdensity lipoprotein (HDL) [13]. Elevated aspartate transaminases (AST) / alanine transaminases (ALT) were used as surrogate markers of Non-Alcoholic Fatty Liver Disease (NAFLD) Impaired glucose tolerance was diagnosed based on abnormal fasting glucose and/or oral glucose tolerance test (OGTT). Obstructive sleep apnea was diagnosed as sleep studies with apnea/hypoxia indices (AHIs) >5/hr.
Data collection and analysis
Two investigators reviewed the case records and relevant data was collected in a standardized form. Demographic data (e.g. age, gender, ethnicity and education level), risk factor for obesity, obesity related co-morbidities, number of dietician and exercise sessions were collected. Patients’ weight and height at each clinic visit were recorded and utilized as outcome measures in this study. The change in BMI Z-Score was classified into three categories: (1) BMI Z-Score increase or stable (2) BMI Z-Score decrease of <0.25 (3) BMI Z-Score decrease of >0.25. The above classification was made based on studies [14-17] that showed an improvement in insulin sensitivity, intima media thickness and cardiovascular risk factors based on the degree of BMI Z-Score change.
For analysis, patients were classified either as dropouts (those that had been seen in obesity service but had failed to come for any appointments without being formally discharged) and attenders (those who attended ≥ 2 appointments).
Data was extracted for statistical analysis using SAS version 9.3 for Windows (SAS, Inc., Cary, NC USA). Baseline demographic and clinical features were compared between those in dropout group versus those in attendee group using the Wilcoxon ranksum test and the two-sample-t-test for non-normal and normal continuous variables, respectively, and the Fisher’s exact test for categorical variables. Pearson’s correlation coefficient was calculated to investigate the relationship between continuous variables such as baseline BMI Z-Score and age. Multiple regression analysis for BMI Z-Score difference at baseline (first) and first follow up visit was performed. Variables included in the multiple regression analysis were gender, age, race, baseline BMI Z-Score, parental overweight, exercise session, presence of obesity co-morbidities, and dietician consultation, and the model was adjusted for the first follow-up visit interval time. Generalized linear mixed model was carried out on the BMI ZScore over multiple visits. The mean value of BMI Z-Score was considered as a function of time from the baseline. The variance component is used as the covariance structure in generalized linear mixed model. It models a different variance component for each random intercept to allow each child to have his/her own overall level of BMI Z-Score.
Results
Study sample and baseline characteristics
A total of 121 patients (68% males) were enrolled in the pediatric weight management program from 1st January 2009 to 29th February 2010. The sample comprised of patients aged 2 to 16 years old with a mean age of 9.2 ± 2.8 years. The majority of the patients were Chinese (46%), while 40 (33%) were Malay, and 16 (13%) were Indian. Four patients (3%) were overweight, 105 (87%) were obese and 12 (10%) were severely obese by CDC criteria [8]. 43 patients (35%) had 1 or parents who were overweight.
Physical activity and dietary interventions
Mean dietary consultation was 1.2 ± 0.96 (range 0-6). Thirty two (26.5%) patients signed up for the exercise program (range 1-123).
Baseline BMI and obesity related comorbidities
Obesity-related medical comorbidities were observed in 68% of the patients and summarized in Table 1.
| Co-morbidity |
No. of patients |
| Screened |
Positive screening |
% of total no. |
| Hypertension |
121 |
34 |
28.1 |
| Dyslipidaemia ¥ |
44 |
35 |
36 |
| Low HDL |
|
14 |
|
| High LDL |
|
6 |
|
| Low HDL and High |
|
9 |
|
| LDL |
|
4 |
|
| Low HDL and High |
|
1 |
|
| TG |
|
1 |
|
| Impaired glucose tolerance |
23 |
7 |
5.8 |
| Fatty liver |
10 |
6 |
8.3 |
| Obstructive sleep apnea |
21 |
17 |
18 |
| Reflux disease |
121 |
3 |
2.5 |
| Orthopaedic complications* |
121 |
3 |
2.5 |
| Polycystic ovarian syndrome |
39 |
1 |
2.6 |
*Includes pes planus, ankle fracture and knee pain
ǂ Includes only females screened
¥ There were no cases of high LDL and high TG or low HDL and high LDL and high TG
Table 1: Obesity-related co-morbidities.
Mean baseline BMI was 29.2 ± 5.4 kg/m2 and baseline BMI ZScore was 2.44 ± 0.52. At baseline, BMI Z-Score negatively correlated with increasing age (r=-0.58, p<0.01). There was no significant difference in baseline BMI Z-Score between the boys (2.46 ± 0.56) and girls (2.38 ± 0.43) (p=0.77). There was also no significant difference in baseline BMI Z-Score between the patients with one or two overweight parent (2.52 ± 0.61) versus patients with no overweight parents (2.39 ± 0.46) (p=0.3), and also no significant difference among different races (p=0.58).
Pre-post intervention outcomes and attrition
The mean BMI Z-Score of patients enrolled in pediatric weight management program was 2.5 ± 0.4 at baseline compared to 2.42 ± 0.35 at first follow up visit, a statistically significant decrease of 0.08 (95% CI: 0.03 to 0.13, p=0.003). Mean duration of time between baseline and first follow up visit was 6.4 ± 3.3 months. Univariate analysis for BMI Z-Score difference between the baseline and first follow up visit showed that age (p=0.037) and baseline BMI Z-Score (p<0.001) were significant. On multivariate analysis, no significant factor predicted a reduction in BMI Z-Score (Table 2).
| Parameter |
Adj Estimate
(95% CI) |
P value |
Adj Estimate¥
(95% CI) |
P value |
| Gender (Male) |
-0.02 (-0.13, 0.09) |
0.702 |
-0.01 (-0.12, 0.10) |
0.826 |
| Age (Years) |
0.01 (-0.01, 0.03) |
0.133 |
0.01 (-0.02, 0.02) |
0.774 |
| Race (Indians*) |
0.02 (-0.13, 0.17) |
0.768 |
0.04 (-0.10, 0.19) |
0.552 |
| Race (Malay*) |
0.05 (-0.07, 0.17) |
0.402 |
0.05 (-0.07, 0.16) |
0.417 |
| Race (Others*) |
0.16 (-0.06, 0.38) |
0.145 |
0.19 (-0.03, 0.40) |
0.083 |
| BMI Z-Score at baseline |
-0.16 (-0.28, -0.03) |
0.018 |
-0.14 (-0.3, 0.01) |
0.067 |
| Parental Overweight (Yes) |
0.06 (-0.04, 0.17) |
0.237 |
0.07 (-0.03, 0.17) |
0.187 |
| Exercise (Yes) |
0.03 (-0.07, 0.14) |
0.517 |
0.03 (-0.07, 0.13) |
0.579 |
| Obesity (Yes) |
-0.01 (-0.14, 0.11) |
0.842 |
0.01 (-0.12, 0.13) |
0.943 |
| Dietician (Yes) |
0.01 (-0.04, 0.06) |
0.563 |
0.02 (-0.03, 0.07) |
0.395 |
Adjusted for first follow-up visit interval time
¥ Adjusted for first follow-up visit interval time, age, and BMI Z-score at baseline
* Reference level is Chinese
Table 2: Summary of multivariate analysis for BMI Z-Score difference at baseline and first visit, using linear regression model.
Baseline characteristics of the dropouts compared to attenders were compared in Table 3. Absence of obesity related co-morbidities diagnosed on baseline visit were associated with increased dropout compared to patients with obesity-related comorbidities (p=0.05). A log rank test was performed to determine if there were differences in dropout rates over time for patients with and without obesity co-morbidities (Figure 1). The dropout distributions for the two groups of patients were statistically significant (p=0.002).
| Variable |
Dropout |
Attenders |
P value* |
| n |
Count (%)/ Mean ± SD |
n |
Count (%)/ Mean ± SD |
| Gender (Male) |
70 |
49 (69) |
51 |
33 (66) |
0.84 |
| Race |
70 |
|
51 |
|
0.57 |
| - Chinese |
|
35 (49.3) |
|
21 (42) |
|
| - Malays |
|
23 (32.4) |
|
17 (34) |
|
| - Indians |
|
7 (9.9) |
|
9 (18) |
|
| BMI Z-Score (SD) |
70 |
2.39±0.59 |
51 |
2.5±0.4 |
0.051 |
| Age (Years) |
70 |
10.1±2.7 |
51 |
9.5±3 |
0.27 |
| Parental overweight (Present) |
70 |
24 (33.8) |
51 |
19 (38) |
0.7 |
| Obesity related co-morbidities (Present) |
70 |
43 (60.6) |
51 |
39 (78) |
0.05 |
*Wilcoxon rank-sum test for non-normal continuous variables; Two-sample t-test for normal continuous variable; Fisher’s Exact test for categorical variables.
Table 3: Baseline characteristics of dropouts versus attenders.
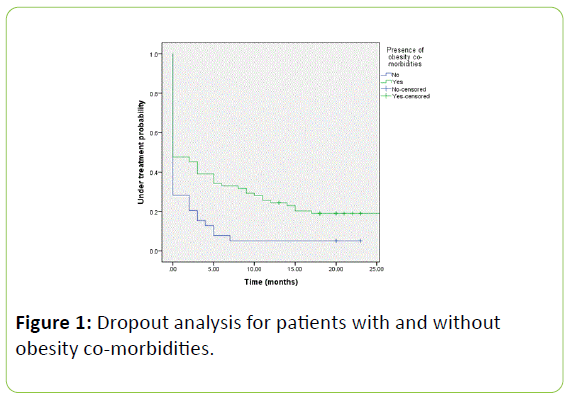
Figure 1: Dropout analysis for patients with and without obesity co-morbidities.
Complete follow up data was obtained for 48% of patients at 6 months and 21% of patients at 24 months. Using intention to treat analysis, 33%, 22% and 16% of patients achieved a reduction in their BMI Z-Score at 6 months, 12 months and 24 months, respectively (Figure 2).
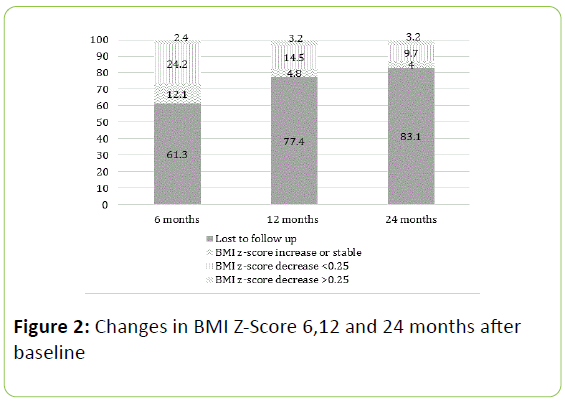
Figure 2: Changes in BMI Z-Score 6,12 and 24 months after baseline
Studying the 51 attenders showed a reduction in their BMI ZScore which was accompanied by improvement in their obesityrelated co-morbidities (Table 4).
| Variable |
n |
Mean ± SD |
P value |
| BMI z-scores |
| - Initial mean (SD) |
51 |
2.51 ± 0.37
2.43 ± 0.39 |
0.001 |
| - Final mean (SD) |
| - Mean change (95% CI) |
| Waist-height ratio |
| - Initial mean (SD) |
25 |
0.67±0.065
0.67±0.053
0.001(-0.017 to 0.019) |
0.881 |
| - Final mean (SD) |
| - Mean change (95% CI) |
| Body Fat % |
16 |
44.45 ±8.86
41.88 ± 11.12
-2.56 (-7.29 to 2.16) |
0.265 |
| - Initial mean (SD) |
| - Final mean (SD) |
| - Mean change (95% CI) |
| Fasting glucose (mmol/L) |
17 |
5.01 ± 1.07
4.77 ± 0.27
-0.22 (-0.75 to 0.317) |
0.401 |
| - Initial mean (SD) |
| - Final mean (SD) |
| - Mean change (95% CI) |
| Glucose (120 min) (mmol/L) |
17 |
7.39 ± 3.55
6.58 ± 1.35
-0.82 ( -2.27 to 0.633) |
0.249 |
| - Initial mean (SD) |
| - Final mean (SD) |
| - Mean change (95% CI) |
| HDL (mmol/L) |
17 |
0.97 ± 0.21
1.05 ± 0.20
0.088 (0.014 to 0.16) |
0.023 |
| - Initial mean (SD) |
| - Final mean (SD) |
| - Mean change (95% CI) |
| TC/HDL ratio |
17 |
5.09 ± 1.21
4.70 ± 1.22
-0.39 (-0.64 to -0.14) |
0.005 |
| - Initial mean (SD) |
| - Final mean (SD) |
| - Mean change (95% CI) |
| Triglycerides (mmol/L) |
17 |
1.32 ± 0.82
1.34 ± 0.64
0.011( -0.25 to 0.28) |
0.926 |
| - Initial mean (SD) |
| - Final mean (SD) |
| - Mean change (95% CI) |
| LDL (mmol/L) |
17 |
3.09 ± 0.58
3.17 ± 0.72 |
0.572 |
| - Initial mean (SD) |
| - Final mean (SD) |
| - Mean change (95% CI) |
| SBP percentile |
52 |
78.5 ± 19.8
72.8 ± 21.3
-5.8 (-12.7 to 1.14) |
0.100 |
| - Initial mean (SD) |
| - Final mean (SD) |
| - Mean change (95% CI) |
| DBP percentile |
52 |
58.8 ± 23.7
54.3 ± 23.0 |
0.277 |
| - Initial mean (SD) |
| - Final mean (SD) |
| - Mean change (95% CI) |
Table 4: Change in BMI Z-Scores, body composition, blood pressure, glucose metabolism and lipid profile in attenders (N=51).
The analysis of the changes in BMI Z-Score over the time in the 28 patients who attended for at least 24 months is performed using a generalized linear mixed model. The age at presentation and time were taken into account to adjust the mixed model for changes in BMI Z-Score over a 24 month period. The results showed that the mean BMI Z-Score difference was -0.05 (-0.09, -0.02) after 12 months of treatment (p=0.004) and -0.15 (-0.23, -0.06) after 24 months of treatment (p=0.001) in the 28 children.
Discussion
The findings of this study suggest that a multidisciplinary weight management program is effective in improving weight status and cardiovascular risk factors in a multiracial population under real-life conditions. The results showed a significant proportion of patients with obesity related comorbidities with hypertension being the most prevalent. However, there is a lower proportion of patients with dyslipidemia, impaired glucose tolerance and fatty liver in this study, compared to other studies [14,18,19], which is related to lower numbers of patients being screen for the metabolic complications. The expert committee’s recommendations on childhood obesity suggest for fasting glucose, lipid panel. ALT and AST screen for children more than 10 years old with obesity [20], 55% of the children meeting the above criteria did not undergo the above recommended metabolic screening. Hence there must be a greater awareness amongst physicians and families alike to screen for obesity related complications. This is especially significant in view of the study findings that presence of obesity related comorbidities is associated with lower attrition rate.
In the present study, the mean reduction in BMI Z-Score was greater after 24 months, suggesting that staying in treatment is beneficial. However, as per study design, a control group was not included. Thus the reduction in BMI Z-Score cannot be concluded to be due to the pediatric obesity service but potentially be due to factors not analyzed in these patients. However, childhood obesity is a significant predictor of adult obesity [21] and the reduction in BMI-Z Score while in pediatric obesity service is thus less likely to be sporadic in nature.
The percentage of patients with reduction in BMI Z-Score was 27% at 6 months and 18% at 12 months and 12.9% at 24 months. These findings are comparable with pediatric obesity service in Middle Europe [22]. It is disappointing that only 3.2% of patients presenting to the service had a reduction in BMI ZJournal Score by 0.25 which has been shown to be clinically relevant to improve body composition and cardiometabolic risk factors [15]. The best five treatment centers in Europe had reported a much better reduction in BMI Z-Score and drop-out rates, presumably attributed to higher intensity of treatment [22]. This study thus serves as an audit for our service to improve the effectiveness of obesity intervention.
The higher prevalence of obesity amongst Malays in our study is consistent with the local pediatric and adult population with obesity [23]. The higher prevalence of obesity amongst Malays is a complex issue. Besides higher energy intake with lower physical activity rate [24] other factors involving cultural, genetics and socioeconomics are likely in play. This is a worrying trend as the children with obesity become adults with obesity with higher prevalence of chronic diseases [3]. The Singapore government has introduced some programs at the primary level amongst the Malay community, with particular focus on healthier versions of Malay dishes and promotion of culturally relevant physical activities, to reduce the obesity rate amongst the Malays with some success [25] but this study has highlighted the need for more research into effective culturally-modified intervention amongst children with obesity.
The poor take up rate of the structured exercise program at 26% was consistent with that found in other studies who encountered similar difficulties in engaging patients with obesity in exercise programs [26].
The study findings are limited by the large dropout rates which can lead to potential drop out bias. High dropout rates had been similarly observed in other similar studies on pediatric obesity services [22] High attendance rate had previously been predictive of success [27], it is thus important to examine the clinic process to reduce dropout rate. Interestingly, we found a significant association between dropout and absence of obesity related comorbidities. The dropout rate amongst patients without obesity co-morbidities continues to increase throughout the course of intervention. One postulated reason is due to lack of urgency on the need for obesity intervention in the absence of comorbidities. This further highlights the importance of raising awareness amongst physicians, patients and their families to actively screen for obesity related comorbidities and heighten the urgency for obesity intervention. We also need to urgently evaluate how best to engage patients with obesity and their families to reduce drop out.
There are several limitations with our study. Firstly, in the same year 2009, there was a concurrent weight management program offered by our hospital known as the Health Service Development Program for Adolescent Weight management which offered a structured group exercise intervention with dietary counseling sessions and 38 patients in the study period took up the program and these patients are analyzed separately. This can lead to selection bias in our study population, which failed to recruit the highly motivated patients thus accounting for the high default rate. As per our study design, we are limited by information bias such as missing data. This was reduced by reviewing all available paper and electronic health records. All cases with missing data were verified by a second investigator before being declared as incomplete.
In conclusion, a multidisciplinary weight management program is effective in reducing the overweight status and cardiovascular risk factors in a multiracial population. The significant association of presence of obesity related comorbidities with lower attrition rate highlights the need for future more targeted studies looking into reducing attrition rates and increasing the awareness of screening for obesity related co-morbidities to address the childhood obesity epidemic.
References
- Organisation WH. Global Strategy on Diet, Physical Activity and Health 2015.
- de Onis M, Blossner M, Borghi E (2010) Global prevalence and trends of overweight and obesity among preschool children. The Am J Clin Nutr. 92:1257-64.
- Ministry of Health Singapore EaDCD (2010) National Health Survey 2010.
- MOH TST (2008) Parliamentary remarks: DPM Teo Chee Hean.
- Reilly JJ,Methven E, McDowell ZC, Hacking B, Alexander D, Stewart L, et al. (2003) Health consequences of obesity. Arch Dis Child. 88: 748-52.
- Singh AS MC, Twisk JW, van Mechelen W, Chinapaw MJ (2008) Tracking of childhood overweight into adulthood: a systematic review of the literature. Obes Rev. 9: 474-88.
- Skelton JA, Beech BM (2011) Attrition in paediatric weight management: a review of the literature and new directions. Obes Rev.12: e273-81.
- Kuczmarski RJ, Ogden CL, Grummer-Strawn LM, Flegal KM, Guo SS, Wei R, et al. (2000) CDC growth charts: United States. Adv data. 8: 1-27.
- Screening and interventions for overweight in children and adolescents: recommendation statement. (2005) Pediatrics. 116: 205-9.
- Board HP (2011) National Physical Activity Guidelines Professional Guide.
- Lee BL (2011) Dietary guidelines in singapore. Asia Pacific journal of clinical nutrition. 20:472-476.
- Riley M, Bluhm B (2012) High blood pressure in children and adolescents. Am Fam Physician. 85: 693-700.
- Executive Summary of The Third Report of The National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, And Treatment of High Blood Cholesterol In Adults (Adult Treatment Panel III). (2001) Jama. 285: 2486-2497.
- Giorgio V, Prono F, Graziano F, Nobili V (2013) Pediatric non alcoholic fatty liver disease: Old and new concepts on development, progression, metabolic insight and potential treatment targets. BMC Pediatrics. 13:1-10.
- Ford AL, Hunt LP, Cooper A, Shield JP (2010) What reduction in BMI SDS is required in obese adolescents to improve body composition and cardiometabolic health? Arch Dis Child. 95:256-261.
- Reinehr T, Andler W (2004) Changes in the atherogenic risk factor profile according to degree of weight loss. Arch Dis Child. 89: 419-422.
- Ji CY (2008) The prevalence of childhood overweight/obesity and the epidemic changes in 1985-2000 for Chinese school-age children and adolescents. Obes Rev: 9.
- Cook S, Kavey RE (2011) Dyslipidemia and pediatric obesity. Pediatr Clin N Am. 58:1363-73, ix.
- Shalitin S, Abrahami M, Lilos P, Phillip M (2005) Insulin resistance and impaired glucose tolerance in obese children and adolescents referred to a tertiary-care center in Israel. Int J Obesity 29:571-578.
- Barlow SE (2007) Expert committee recommendations regarding the prevention, assessment, and treatment of child and adolescent overweight and obesity: Summary report. Pediatrics. 120: S164-92.
- Falkner B (2008) Children and adolescents with obesity-associated high blood pressure. Journal of the American Society of Hypertension : JASH. 2:267-74.
- Reinehr T, Widhalm K, l'Allemand D, Wiegand S, Wabitsch M, Holl RW (2009) Two-year follow-up in 21,784 overweight children and adolescents with lifestyle intervention. Obesity (Silver Spring, Md). 17:1196-9.
- Lee YS, Kek BL, Poh LK, Saw SM, Loke KY (2008) Association of raised liver transaminases with physical inactivity, increased waist-hip ratio, and other metabolic morbidities in severely obese children. J Pediatr Gastr Nutr. 47:172-178.
- Board HDP (2010) National Nutrition Survey.
- Nidhi Gupta MKC, Jingzhen Yang, Govindasamy Balasekaran, Michael Chia, Robert N, Girandola, et al. (2010) Obesity prevention in Singapore: Collaborative efforts among government, health professionals and the community. Asian Council of Exercise and Sports Science. 7:61-70.
- Sabin MA, Ford A, Hunt L, Jamal R, Crowne EC, Shield JP (2007) Which factors are associated with a successful outcome in a weight management programme for obese children? J Eval Clin Pract. 13:364-368.
- Kalarchian MA, Levine MD, Arslanian SA, Ewing LJ, Houck PR, Cheng Y, et al. (2009) Family-based treatment of severe pediatric obesity: randomized, controlled trial. Pediatrics. 124:1060-1068.