Vera Kolyovska1*, Iskra V Sainova1, Ivaila Ivanova-Pandourska1, Rumyana Eneva2, Stephan Engibarov2, Tzvetanka Markova3 and Dimitar Maslarov4
1Institute of Experimental Morphology, Pathology and Anthropology with Museum (IEMPAM), Bulgarian Academy of Sciences (BAS), Sofia, Bulgaria
2The “Stephan Angel off” Institute of Microbiology, Bulgarian Academy of Sciences (BAS), Sofia, Bulgaria
3Department of Pharmacology and Toxicology, Sofia Medical University, Sofia, Bulgaria
4Neurology Clinic, First MHAT, Medical University, Sofia, Bulgaria
Corresponding Author:
Vera Kolyovska
Institute of Experimental Morphology, Pathology and Anthropology with Museum (IEMPAM)
Bulgarian Academy of Sciences (BAS), Sofia, Bulgaria
Tel: +359 (2) 971 32 19
E-mail: verakol@abv.bg
Received date: July 19, 2018; Accepted date: August 07, 2018; Published date: August 09, 2018
Citation: Kolyovska V, Sainova IV, Pandourska II, Eneva R, Engibarov S, et al. (2018) Presence and Levels of Anti-GM3 Antibodies and GM3 Ganglioside Molecules in Probes from Experimental In Vitro-Models. Biochem Mol Biol J 4:18. doi: 10.21767/2471-8084.100067
Keywords
Anti-GM3 antibodies; Experimental in vitromodels; ELISA
Introduction
Gangliosides have been characterized as glycosphingolipids, which are among the main elements of the cell membranes [1]. These molecules have been found to mediate main biological processes as cell signaling pathways, especially cell differentiation, proliferation and death [2].
Ganglioside GM3 has been proposed as a tumor-associated antigen and specific marker in many malignant disorders [3-5]. According to other references a protective function of the increased values of this ganglioside in malignant cells has been proposed. This is based on the accumulation of the molecule in apoptotic malignant cells [6]. In this way, GM3 has been characterized as a target for novel anti-malignancy therapeutic strategies [7]. It can act as a regulator of membranetransmitted signals [8,9] and by modulation of the functions of tumor suppressors [10], or by its anti-angiogenic effects in some solid malignancies as well [11]. In addition, specific increase of ganglioside molecular species during immune cell differentiation of normal and malignant myeloid and lymphoid progenitors has been established [12-14].
In this aspect, the main goal of the current paper was to be investigated the differences in GM3 ganglioside levels in lysates of in vitro-incubated normal and malignant cells. The amounts of this molecule are intensively changing depending on the cell functions. The levels of anti-GM3 antibodies were assessed in parallel from all types of the cell cultures used.
Materials and Methods
Types of in vitro-cell cultures
Normal cells 3T3 from Balb/c mouse embryos, malignant cells from mouse myeloma, as well as mixed cultures from both were used. The so prepared cultures (1 × 106 cells/ml), were cultivated at 37°C in incubator with 5% ??2 and 95% humidity, in RPMI 1640, Dulbecco’s Modified Minimal Essential Medium (DMEM) or a mixture of both media (1:1) supplemented with 10% Fetal Calf Serum (FCS), 100 UI/ml Penicillin, 0.25 mg/ml Streptomycin and 0.25 mg/ml Amphotericin-B, in 24-well plaques. The observations were performed by inverted light microscope, supplied with megapixel CCD-camera. The cells of all three culture types were lysed by treatment of previously-prepared cell suspensions with 10% tri-chloroacetic acid (Cl3CCOOH) and 0.48 M solution of K3PO4. For isolation of specific bio-molecules with affinity to GSH from the so prepared protein extracts, the isolated total protein content from all three cell culture types was passed through GSH-Agarose columns.
Determination of the presence and levels of ganglioside GM3 and anti-GM3 antibodies in in vitro-cell cultures by ELISA technique
Slight modifications of the method of Mitzutamari et al., were made [15]. For measuring the values of antibodies to GM3, 1000 ng of the ganglioside (Sigma) in 100 ml of methanol were pipetted into microtiter plate wells, containing the tested biological material. For determination the values of GM3, instead of ganglioside solution, the tested probes were mixed with sera, previously proved to contain specific antibodies to this ganglioside. Subsequently, the wells were air-dried and blocked with BSA-PBS (Sigma) (1% bovine serum albumin in phosphate-buffered saline) for 1 hour. After six-fold washing with PBS, 100 ml of each one of the prepared lysates from the cell cultures, described above, diluted 1: 20 to 1: 5000 in PBS were added to each well and incubated overnight. After that, the plates were washed six-fold with PBS. Binding was detected by following 2 hours incubation period with BSAPBS (Sigma) diluted (1/3200) peroxidase-conjugated goat antihuman IgG antibodies (Bul Bio Ltd., NCIPD, Sofia). All the incubations were performed at 4°C. The plates were then washed six times with PBS. Color development was achieved in a substrate solution, previously prepared from 15mM O-Phenylenediamine and 0.015% H2O2 in 0.1 M sodium acetate buffer (0.2 M CH3COONa/0.2 M CH3COOH; pH 5.0) at 20°C. The reaction was stopped after 30 minutes with 50 ml of 1N H2SO4, and the optical density (OD) was assessed spectrometrically at 490 nm on ELISA-reader (TECAN TM, Sunrise, Austria). Non-specific antibody bindings (OD value in a well not containing the specific molecule in the respective probe) were subtracted for each measurement.
The data were considered strongly positive, when the mean OD exceeded 2 ± SD (standard deviation), compared with the controls. The standard error of mean varied between ± 0.01 and ± 0.1. For the best reliability the procedure was performed three times.
Results
In the total (control) lysates from probes of the all three types of cell cultures, anti-GM3 antibodies were assessed only at dilutions 1: 200 and 1: 400 (Figure 1A), and ganglioside GM3 – only at dilutions 1: 100 and 1: 400, respectively (Figure 1B). No statistically significant differences were in the levels established in this case. At both 1: 200 and 1: 400 dilutions, the highest values of anti-GM3 antibodies were assessed in the lysate from the culture of normal cells, and the lowest – in the lysate from the mixed culture (Figure 1A). At dilution 1: 100, the lowest level of GM3 was found in the lysate from the culture of malignant cells and the highest – in the lysate from the mixed cell culture, unlike of the result at dilution 1: 400, where the highest amount of this ganglioside was established in the lysate from the culture of normal cells (Figure 1B). At dilution 1: 100, the highest value of the same ganglioside was assessed in the extract from the culture of normal cells, and the lowest – again in the lysate from the mixed culture.
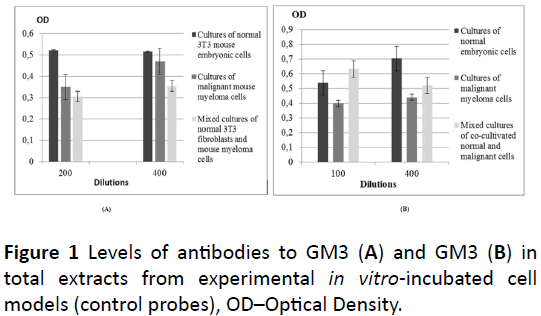
Figure 1: Levels of antibodies to GM3 (A) and GM3 (B) in total extracts from experimental in vitro-incubated cell models (control probes), OD–Optical Density.
Different data were observed in the lysates from the same three types of cell cultures, determined as containing “selected” molecules, possessing affinity to GSH (Figure 2). Here, increased values of both GM3 and anti-GM3 antibodies were established in the lysates of malignant cells and mixed cultures, in comparison with that from the normal cells. At all dilutions of the lysates from the three types of cell cultures, the lowest value of the anti-GM3 antibodies was established in the lysate from the culture of normal cells (Figure 2A). At dilutions 1: 40 and 1: 100, the highest value of these antibodies was assessed in the lysate from the culture of malignant cells, and at dilutions 1: 200 and 1: 400 – in the lysate of the mixed cell culture, respectively. Also, at all dilutions of the extracts from the cell cultures, the highest level of GM3 was established in the lysate of the culture of malignant cells, but only at dilution 1: 200 the difference between this value and the value in the lysate from the culture of normal cells was not statistically significant (Figure 2B). At the same dilution, as well as at dilution 1: 100, the lowest level of this ganglioside was noted in the lysate from the mixed cell culture, unlike at dilutions 1: 40 (the lowest) and 1: 400 (the highest), where the lowest value of GM3 was assessed in the lysate from the culture of normal cells.
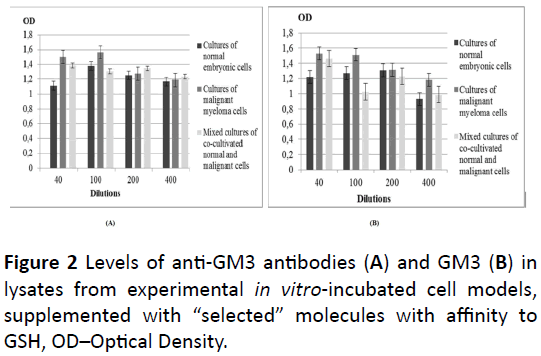
Figure 2: Levels of anti-GM3 antibodies (A) and GM3 (B) in lysates from experimental in vitro-incubated cell models, supplemented with “selected” molecules with affinity to GSH, OD–Optical Density.
Discussion
The presented data were in support with many literature sources about the higher values of the tested molecules (GM3 and antibodies to it) in neo-plastic cells and tumors as markers for malignancy [3-5]. Moreover, the role of this ganglioside on important signaling pathways has been proved, which has delineated it as appropriate target for anti-neoplastic therapeutic directions [7]. On the other hand, the data, in particular the noted higher values in the lysate from the mixed culture compared with this of the culture from normal cells, both determined as containing “selected” bio-molecules with affinity to GSH, confirm the reported increased levels of GM3 ganglioside in the process of cell differentiation in myeloid direction [12]. One of the reasons for the observed deviations, as well as the often lack of certain levels in the extracts, containing also non-specific molecules, non-possessing affinity to GSH from the lysates of all cell culture types could be a decreased concentration of the specific GM3 and anti-GM3 antibodies at the expense of other non-specific bio-molecules, including of cross-linked to the antibodies [15], in equal volume of biological material. Another hypothesis could be the eventual existence of GM3 besides in free, non-bound form, also in different bound forms with various bio-molecules [16], depending of the respective function, in which this ganglioside participates.
The role of Glutathione (L-γ-glutamyl-L-cysteinyl-glycine) in the cell and its redox-status, determining its physiological functions, has been underlined [17]. This molecule regulates many cascade mechanisms, underlining the living cell processes, as well as in many inflammatory and proinflammatory.
The current paper is in confirmation of literature findings that non-lymphoid cell types are able to express and produce immunoglobulins (in the concrete case, IgG antibodies) in appropriate conditions [18,19], including of such with malignant characteristics [20]. Two main explanations about the established presence of anti-GM3 antibodies in the lysates from cells cultures, could be proposed:
• Indicated in many studies cells with lymphoid functions, derived from separate stem-like embryonic cells [21,22].
• Proved action of some cell-produced proteins as antibodies or antibody domains to specific bio-molecules [23].
On this basis, possibilities for development of novel immunotherapeutic strategies against malignancies have been proposed [24]. So, variations in the location and distribution of gangliosides and antibodies (from class IgG) to them in separate regions of the cell could be suggested, on the basis of literature sources [25].
Conclusion
Only in separate dilutions of the total extracts from the used three types of in vitro-experimental models certain values of both ganglioside GM3 and antibodies to it were established. In the lysates from the same probes, determined as containing “selected” molecules, possessing affinity to GSH in all dilutions from the cell cultures the lowest values in both GM3 and antibodies to it were established in the extract from the culture of the normal cells. These differences can be explained by the presence of molecules, non-possessing affinity to GSH and the reduction of bio-molecules with such affinity in an equal volume of biological material. The results could be explained with the presence of cross-linked antibodies. These data were in support of cited literature sources about higher values of GM3 in malignant cells as a marker for neo-plasticity, including in its participation in protective functions. On the other hand, the higher values in the mixed culture of cocultivated normal and malignant cells confirm the proved increasing levels of this ganglioside in the process of myeloid differentiation. Another explanation of the observed deviations could be the existence of GM3 in free, nonbounded form, also bounded with different molecules, in dependence of the biological function, which it performs. The observed presence of antibodies to this ganglioside in the probes of the cell cultures, supplemented with “selected” biomolecules, possessing affinity to GSH, could be interpreted with established by other investigations presence of lymphoidlike cells. Another hypothesis could be showed from other references the possibility of non-lymphoid cells to express immunoglobulin genes and to produce antibodies (from IgG type).
References
- Moussavou G, Kwak D, Lim MU, Kim JS, Kim SU, et al. (2013) Role of gangliosides in the differentiation of human mesenchymal-derived stem cells into osteoblasts and neuronal cells. BMB Rep 46: 527-532.
- Zurita A, Maccioni H, Daniotti J (2001) Modulation of epidermal growth factor receptor phosphorylation by endogenously expressed gangliosides. Biochem J 355: 465-472.
- Nicolae I, Caragheorgheopol A, Schipor S, Nicolae C, Paun D, et al. (2011) Gangliosides and sex hormones in human melanoma. Acta Endocrinol 7: 337-344.
- Miura Y, Kainuma M, Jiang H, Velasco H, Vogt P, et al. (2004) Reversion of the Jun-induced oncogenic phenotype by enhanced synthesis of sialosyllactosylceramide (GM3 ganglioside). Proc Natl Acad Sci USA 101: 16204-16209.
- Pe’guet-Navarro J, Popa M, Berthier O, Schmitt D, Portoukalian J (2003) Gangliosides from human melanoma tumors impair dendritic cell differentiation from monocytes and induce their apoptosis. J Immunol 170: 3488-3494.
- Watanabe R, Ohyama C, Aoki H, Takahashi T, Satoh M, et al. (2002) Ganglioside GM3 overexpression induces apoptosis and reduces malignant potential in murine bladder cancer. Cancer Res 62: 3850-3854.
- Zheng C, Terreni M, Sollogoub M, Zhang Y (2018) Ganglioside GM3 and its role in cancer. Curr Med Chem 25: 1.
- Hashiramoto A, Mizukami H, Yamashita T (2006) Ganglioside GM3 promotes cell migration by regulating MAPK and c-Fos/AP-1. Oncogene 25: 3948-3955.
- Iwabuchi K, Yamamura S, Prinetti A, Handa K, Hakomori S (1998) GM3-enriched microdomain involved in cell adhesion and signal transduction through carbohydrate-carbohydrate interaction in mouse melanoma B16 cells. J Biol Chem 273: 9130-9138.
- Toledo M, Suzuki E, Handa K, Hakomori S (2004) Cell growth regulation through GM3-enriched microdomain (glycosynapse) in human lung embryonal fibroblast WI38 and its oncogenic transformant VA13. J Biol Chem 279: 34555-34664.
- Choi H-J, Chung TW, Kang SK, Lee YC, Ko JH, et al. (2006) Ganglioside GM3 modulates tumor suppressor PTEN-mediated cell cycle progression-transcriptional induction of p21WAF1 and p27kip1 by inhibition of PI-3K/AKT pathway. Glycobiology 16: 573-583.
- Garofalo T, Sorice M, Misasi R, Cinque B, Mattei V, et al. (2002) Ganglioside GM3 activates ERKs in human lymphocytic cells. J Lipid Res 43: 971-978.
- Jiiu HN, Takakut F, Terui Y, Miurat Y, Saito M (1986) Ganglioside GM3: An acidic membrane component that increases during macrophage-like cell differentiation can induce monocytic differentiation of human myeloid and monocytoid leukemic cell lines HL-60 and U937. Proc Natl Acad Sci USA 83: 782-786.
- Sorice M, Longo A, Garofalo T, Mattei V, Misasi R, et al. (2004) Role of GM3-enriched microdomains in signal transduction regulation in T lymphocytes. Glycoconjugate J 20: 63-70.
- Mitzutamari R, Wiegandt H, Nores G (1994) Characterization of anti-ganglioside antibodies present in normal human plasma. J Neuroimmunol 5: 215-220.
- Krengel U, Olsson LL, Martinez C, Talavera A, Rogas G, et al. (2004) Structure and molecular interaction of a unique antitumor antibody specific for N-glycolyl GM3. J Biol Chem 279: 5597-5603.
- Meister A (1983) Selective modification of glutathione metabolism. Science 220: 472-477.
- Bebbington C (1991) Expression of antibody genes in nonlymphoid mammalian cells. Methods 2: 136-145.
- Nematpour F, Mahboudi F, Khalaj V, Vaziri B, Ahmadi S, et al. (2017) Optimization of monoclonal antibody expression in CHO cells by employing epigenetic gene regulation tools. Turk J Biol 2018: 622-628.
- Chen Z, Qiu X, Gu J (2009) Immunoglobulin expression in non-lymphoid lineage and neoplastic cells. Am J Pathol 174: 1139-1148.
- Sorice M, Parolini I, Sansolini T, Garofalo T, Dolo V, et al. (1997) Evidence for the existence of ganglioside-enriched plasma membrane domains in human peripheral lymphocytes. J Lipid Res 38: 969-980.
- Cho S, Webber T, Carlile J, Nakano T, Lewis S, et al. (1999) Functional characterization of B lymphocytes generated in vitro from embryonic stem cells. Proc Natl Acad Sci USA 96: 9797-9802.
- Ponomarenko N, Durova O, Vorobiev I, Belogurov A, Kurkova I, et al. (2006) Autoantibodies to myelin basic protein catalyze site-specific degradation of their antigen. Proc Natl Acad Sci USA 103: 281-286.
- https://www.seminoncol.org/article/S0093-7754(18)30042-3/pdf.
- Uemura S, Yoshida S, Shishido F, Inokuchi J (2009) The cytoplasmic tail of GM3 synthase defines its subcellular localization, stability, and in vivo activity. Mol Biol Cell 20: 3088-3100.



