Research Article - (2016) Volume 2, Issue 1
College of Materials Science and Engineering, Beijing University of Chemical Technology, China.
Received Date: May 26, 2016; Accepted Date: June 13, 2016; Published Date: June 16, 2016
Citation: Shi D, Li P, Cao B. Preparation of PDMS/PVDF Composite Pervaporation Membrane Modified with Hydrophobic TiO2 Nanoparticles for Separating Formaldehyde Solution. Polym Sci. 2016, 1:2.
In this paper, polydimethylsiloxane (PDMS) was blended with hydrophobic TiO2 nanoparticles and then coated on top of the polyvinylidene fluoride (PVDF) ultrafiltration membrane to form the (PDMS/TiO2)/PVDF composite membrane. The structure, morphology and physical property of the composite membrane were characterized by FTIR, SEM, contact angle measurement, and dry-wet swelling degree test. The TiO2 was uniformly distributed in the PDMS matrix, and the composite membrane showed higher affinity to formaldehyde than water. The membrane was used to separate the 1000 ppm formaldehyde water solution through a pervaporation (PV) process. The results showed that the membrane could selectively permeate formaldehyde over water and the best separation performance could be achieved at 50°C with a separation factor of 11.25, and a total flux of 187.72 g·m-2·h-1, which were better than the pristine PDMS/PVDF membrane with separation factor of 10.66 and total flux of 115.52 g·m-2·h-1. Hence, adding TiO2 not only increased the membrane flux, but also increased the separation factor of the composite membrane. This study showed that pervaporation technology had the potential for treating formaldehyde wastewater.
Formaldehyde/water separation; Pervaporation; (PDMS/TiO2)/PVDF composite membrane
Formaldehyde has been widely applied in both industrial and nonindustrial processes such as: the production of adhesives, hardboards, resins, fungicides, and paper, etc. [1-4]. However, formaldehyde is an allergic source and one of the most dangerous mutagens. Even at very low concentrations (<20 mg/L), formaldehyde may still cause cough, incitement to mucous membrane and disorder in gulping. Moreover, formaldehyde can cause death or inhibit biological activity of micro-organism by directly reacting with microbe protein, deoxyribonucleic acid (DNA) and ribonucleic acid (RNA) [5]. Hence, it is a severe pollutant to environment. When the formaldehyde concentration is higher than 200 mg/L, the microbial activity will be completely restrained. Therefore, biological degrading method is not applicable for treating high concentrated formaldehyde solution [6]. So, there is an urgent need to develop more eco-friendly and efficient technologies for the removal of formaldehyde, especially at high concentration.
Current methods for removing formaldehyde are Fenton reaction [7], chemisorption [8,9], photocatalytic oxidization [10], thermal catalytic oxidization [11-15], plasma catalysis [16-18], and biological/botanical filtration [19,20]. These methods share the same idea of decomposing formaldehyde through chemical or biological degradation processes in wastewater which typically demand high operation cost and large consumptions of oxidant and enzyme. In addition, the decomposed formaldehyde loses its application value. Hence, a low cost, high efficient, and formaldehyde recyclable technology is required for dealing with concentrated formaldehyde wastewater.
Pervaporation is an energy efficient technology and be widely applied or studied in the separations of liquid mixtures. Due to the separation mechanism of pervaporation membrane is solution-diffusion, the separation factor is not subject to vaporliquid equilibrium. Therefore, pervaporation can be applied in separating azeotropic mixtures, liquid compound nearing their boiling points, and removing trace organic matters in water [21-23]. In this study, we aim to explore the possibility of using pervaporation technology to separate formaldehyde solutions. Since formaldehyde is the minor component in the solution, it is more efficient for the pervaporation membrane to selectively permeate formaldehyde over water. Hence, a rubbery polymer, hydrophobic polydimethyl siloxane (PDMS), was chosen as the base material and a hydrophobic nanoparticle, titanium dioxide (TiO2), was used as an additive to further increase the hydrophobicity of PDMS in the purpose of achieving a higher separation factor of formaldehyde over water. Polyvinylidene fluoride (PVDF) ultrafiltration membrane was used as the substrate of the composite membrane in the regard of its better mechanical property and hydrophobic nature. Separation performance of the (PDMS-TiO2)/PVDF composite membrane for dealing with a 1000 mg/L formaldehyde solution was determined and impacts including temperature, TiO2 content, membrane hydrophobicity (contact angle) on the membrane flux and separation factor of formaldehyde/water were studied systematically.
Materials
Formaldehyde solution (37.0-40.0 wt% in water) was obtained from Xilong Chemical Co., Ltd. (Shantou, China); TiO2 (anatase, 99.8% metals basis, 100 nm, hydrophobic), silicone elastomer, silicone elastomer curing agent, acetamide (98.5% purity) were purchased from Fuchen Chemical reagent factory (Tianjin, China); ethyl acetate (≧99.5% purity) and acetic acid (≧99.5% purity) were purchased from Beijing chemical factory (Beijing, China). The above three chemicals were used to synthesize acetyl acetone solution. PVDF ultrafiltration membrane (0.22 μm) was purchased from Tianjin Jinteng Co. Ltd China. All chemicals were used without further purification. The deionized (DI) water with a resistivity of 18.2 MΩ·cm was supplied from MilliQ water system (Milli-Q Advantage A10, Millipore, USA) located in our lab.
Preparation of composite membrane
The (PDMS-TiO2)/PVDF composite membrane was prepared in the following procedure. First, silicone elastomer, curing agent and hexane were mixed in a beaker with a weight ratio of 10:1:62. After the solution was well mixed, the TiO2 nanoparticle was added into the solution in the same weight as the silicone. That is, in the solution the weight ratio of silicone elastomer, curing agent, hexane, and TiO2, is 10:1:62:11. The solution was sonicated in an ultrasonic cell disrupter for 30 min and degassed. Then, the solution was casted on top of a PVDF membrane using a doctor blade and left in fume hood for hexane evaporation. After that, the dried membrane was placed in a vacuum oven and heated at 80°C to crosslink the PDMS for 12 h. The resulting composite membrane was ready for pervaporation and characterization tests. A similar process was carried on to fabricate the PDMS/ PVDF composite membrane to benchmark the separation performance of the (PDMS-TiO2)/PVDF composite membrane.
General characterization
UV-visible absorption spectroscopy (UV; Puxi TU-1810 spectrophotometer, China) was applied to detect the concentration of formaldehyde. Scanning electron microscopy (SEM; Hitachi S-4700, Japan) was used to observe the morphology of the samples. Contact angle instrument was used to measure the contact angles of pure water and 37-40 wt% formaldehyde solution on the surfaces of PDMS and PDMS/TiO2 membranes. Fourier transform infrared spectroscopy (FT-IR; PerkinElmer Spectrum RX1, USA), was used to analyze the chemical structures of PDMS and PDMS/TiO2 membranes.
Pervaporation equipment
Figure 1 gives the schematic diagram of the pervaporation equipment built in our lab. The pervaporation experiment was carried out in the following procedure. First, a 1000 ppm formaldehyde solution was placed in a sealed feed tank equipped with a temperature controller, and a mixer. The feed tank was put on the top of an electronic balance which was used to measure the weight loss of the formaldehyde solution that was used to estimate the overall flux of the membrane. The downstream pressure was kept at 100 Pa to provide the driving force for pervaporation process and the permeate was condensed in a cold trapper by liquid nitrogen. During the pervaporation test, the feed temperature varied from 30 to 60°C. Because of the very low concentration of formaldehyde in the permeated vapor, it was extremely difficult to condense all the permeated formaldehyde in the cold trapper. This led to some odd experimental results if we directly determined the fluxes of formaldehyde and water by analyzing the composition and weight of the permeate collected in the cold trap. Therefore, an indirect method was adopted to estimate the flux of formaldehyde. Specifically, the weight loss of the feed solution was monitored by the electrical balance. And the formaldehyde concentrations in the feed solution before and after each PV experiment were also detected by the UV spectrophotometry. The total flux (J, g m-2 h-1) was estimated using Eq. (1):

where J is the total flux, M0 (g)-weight of the feed solution at time 0 (h), Mt (g)-weight of the feed solution at time t (h), A- the effective membrane area (m2), and t- the operation time (h). The flux of the formaldehyde (Jf, g m-2 h-1) was estimated by Eq. (2):

where C0 and Ct are the concentrations of formaldehyde at time 0 and time t, respectively. The water flux (Jw, g m-2 h-1) was estimated using Eq. (3):
Jw=J-Jf (3)
The separation factor (α) of formaldehyde over water was estimated using Eq. (4)

where Cf refers to the concentration of formaldehyde in the permeate solution.
Swelling test
The swelling degree was estimated by measuring the weights of the dry membrane and 1000 ppm formaldehyde solution soaked membrane and used Eq. (5):
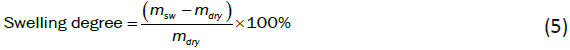
where msw, and mdry, are the weights of water swelled and dry membrane.
Determination of the formaldehyde concentration
The concentration of formaldehyde was determined using the UV spectrophotometry. Since the formaldehyde solution was colorless, it needed to be dyed before UV analysis. Specifically, in the presence of excessive amount of ammonium salt, acetyl acetone was added into the solution and reacted with formaldehyde to form a yellow compound. The resulting yellowish solution was analyzed by the UV spectrophotometry and exhibited the absorbing spectra as shown in Figure 2. The absorbent value corresponding to the wavelength at 414 nm which was the highest absorbance of the spectra was adopted to generate the calibration curve. Then, a series of formaldehyde solutions with concentrations ranging from 0 ppm to 1.6 ppm were monitored and their corresponding absorbance to the wavelength at 414 nm versus concentrations of formaldehyde was plotted in Figure 3. A linear correlation between absorbance and concentration was obtained with a fitting equation of y=0.039x+0.0188 (where y represents the absorbance of formaldehyde, x represents the concentration of formaldehyde). This equation was applied to estimate the concentration of formaldehyde in the feed solution. Note that, before UV analysis, the testing solution was diluted 100 times to fit the concentration range of the calibration curve.
Swelling degree of PDMS
For pervaporation membranes, swelling property is very important, since a swelled membrane tends to loss its diffusive selectively and mechanical stability [24-32]. Figure 4 shows the swelling degree of the PDMS membrane after being soaked in 1000 ppm formaldehyde solution at different temperatures. As it can be seen, in the whole temperature range, the swelling degree is not higher than 0.2% which is very small for typical pervaporation membranes.
Vapor-liquid equilibrium of water-formaldehyde system
Table 1 lists the experimental data of vapor-liquid equilibrium for water- formaldehyde system at certain ranges of temperatures and concentrations [31]. A curve relative to the partition coefficient of formaldehyde over water versus temperature is plotted in Figure 5 based on the data. One important phenomenon is discovered. That is, at the very low formaldehyde concentration, water is significantly more volatile than formaldehyde. This is because formaldehyde molecule forms hydrogen bond with water molecule which makes formaldehyde more difficult to vaporize than water [30]. Based on the vapor-liquid equilibrium data, we can claim that it is unwise to remove trace amount of formaldehyde using the distillation method. On the contrary, a pervaporation membrane that can selectively permeate formaldehyde over water is preferred for this application.
| xF | Total vapor pressure, mmHg | |||||
|---|---|---|---|---|---|---|
| 40°C | 50°C | 60°C | 70°C | 80°C | 90°C | |
| 0.0000 | 55.3 | 92.5 | 149.4 | 233.7 | 355.3 | 525.7 |
| 0.0295 | 52.9 | 91.8 | 149.3 | 233.9 | 357.2 | 532.0 |
| 0.0364 | 53.9 | 90.8 | 148.0 | 234.4 | 357.6 | 533.6 |
| 0.0627 | 53.1 | 90.1 | 147.4 | 233.3 | 358.2 | 537.5 |
| 0.0672 | 52.5 | 90.3 | 146.9 | 233.2 | 358.3 | 536.6 |
| 0.0688 | 53.1 | 90.7 | 146.7 | 232.9 | 359.8 | 538.8 |
| 0.1103 | 51.8 | 88.6 | 146.0 | 232.0 | 357.7 | 539.4 |
| 0.1566 | 51.0 | 86.8 | 146.1 | 228.3 | 354.6 | 536.9 |
| 0.2088 | 49.1 | 84.4 | 140.5 | 225.7 | 351.9 | 534.1 |
| 0.2302 | 47.4 | 83.3 | 138.6 | 223.6 | 348.4 | 523.0 |
| 0.2795 | 46.1 | 80.5 | 136.8 | 218.5 | 342.4 | … |
| 0.3657 | … | … | 131.0 | 212.2 | 334.6 | 515.8 |
Table 1: Isothermal Vapor-Liquid Equilibrium Data for Water- Formaldehyde System [30].
Membrane characterizations
The FT-IR spectra of PDMS and PDMS/TiO2 composite membranes: To confirm the molecular structures of PDMS and PDMS/TiO2 composite membranes, FT-IR analysis was applied. According to Figure 6, a sharp peak at 600 cm-1 represents the Ti-O stretching, which suggests the existence of TiO2 nanoparticles in the PDMS/TiO2 composite membrane. While peaks at 1260 cm-1, 1084 cm-1, and 800 cm-1, represent the functional groups of Si- CH3, O-Si-O and Si-(CH3)2 of the PDMS molecule [33,34].
Morphologies of the PDMS/PVDF and (PDMS/TiO2)/PVDF composite membrane: Figure 7 shows the surface and cross section SEM morphologies of the PDMS skin layer. As it can be seen, the PDMS skin layer is symmetric, dense with a smooth surface. The SEM morhpologies of the surface and cross section structures of (PDMS/TiO2)/PVDF composite membranes are shown in Figure 8a and 8b show that the hydrophobic TiO2 nanoparticles are uniformly distributed in the PDMS polymer matrix. Figure 8c and 8d show that the PDMS/TiO2 skin layer is well attached to the supported PVDF membrane which is microporous. The micro-porous supporting layer and the thin skin layer of the composite membrane shall lead to the high selectivity and flux of the pervaporation membrane.
Contact angle: The affinity to formaldehyde of the (PDMS/ TiO2)/PVDF composite membrane was confirmed by the contact angle measurements. Contact angles were measured using the advancing contact angle methods. As shown in Figure 9, the contact angle of water on the membrane surface is 117.87°, while the contact angle of 37%-40% formaldehyde solution is 89.86°. These results suggest that the (PDMS/TiO2)/PVDF composite membrane has a higher affinity to formaldehyde than water [24,25].
The influence of temperature and TiO2 nanoparticles on the separation performances
Figure 10a and 10b show the fluxes and separation factors of the PDMS/PVDF composite membrane at temperatures ranging from 30 to 60°C for separating the 1000 ppm formaldehyde solution. And Figure 11a and 11b show the separation properties of the (PDMS/TiO2)/PVDF composite membrane. As it can be seen, both the water flux and formaldehyde flux increase with temperature for both two composite membranes. This is because of two reasons: (1) the increases in the diffusivity with temperature, and (2) the saturate pressures of both water and formaldehyde are higher at high temperatures. The higher vapor pressure in the membrane feed side with the same low pressure in the permeate side (100 Pa) results in a higher membrane driving force as the temperature increases [26,27]. The separation factors for both two composite membranes reach their highest value at 50°C. The reason for this result will be detailed explained in the next section. In addition, the (PDMS/TiO2)/PVDF composite membrane exhibits a better separation performance than the PDMS/PVDF composite membrane. The total flux and separation factor of the former are 187.72 g·m-2·h-1, and 11.25 at 50°C, respectively, while those of the latter are 115.52 g·m-2·h-1, and 10.66, respectively.
Hence, the addition of TiO2 improves the separation performance significantly.
The Arrhenius plots of fluxes of water and formaldehyde
The relationship between flux of pervaporation membrane and the feed temperature can be well described by the Arrhenius’s law, as shown in Eq. (6) [28,29]:

where Ji represents the permeate flux of membrane; Ai represents the pre- exponential factor; R represents the gas constant; T represents the kelvin temperature; and Epj represents the apparent activation energy.
Figure 12 shows the relations between the logarithm of the water and formaldehyde fluxes of the (PDMS/TiO2)/PVDF composite membrane versus the reciprocal of the kelvin temperature of the feed liquid. It can be seen that the logarithm of the flux of the composite membrane and the reciprocal of the absolute temperature of the feed liquid obeys well to the Arrhenius’s law. It can be also seen that the activation energy of formaldehyde is higher than that of water. Hence, the fluxes of formaldehyde shall increase more significantly than water with temperature so that the separation factor increases as temperature increases from 30 to 50°C. However, it cannot explain why separation factor decreases as the temperature increases from 50 to 60°C.This result is probably due to the solubility of formaldehyde decreases at higher temperature. Since the membrane shows higher affinity to formaldehyde, it is reasonable to assume that the solubility of formaldehyde is higher than water. However, solubility is typically decreases with temperature. With the temperature increases, the permeability of formaldehyde maybe increase less significant than water.
The (PDMS/TiO2)/PVDF composite membrane were successfully prepared, and their structures have been characterized. The contact angle results showed that the membranes had a high affinity to formaldehyde over water. By analyzing the vapor-liquid equilibrium data of the formaldehyde-water system, water was found to be more volatile at low formaldehyde concentrations. Thus, distillation method is not suitable for treating dilute formaldehyde solutions.
The effects of the temperatures and the incorporation of TiO2 on the separation performances of the membrane for separating 1000 ppm formaldehyde solution were studied. The results indicated that (1) the PDMS based composite membranes could effectively enrich formaldehyde from the dilute formaldehyde solution, and (2) the incorporation of TiO2 could significantly increase the membrane flux and separation factor of formaldehyde over water. The best separation performance was obtained for the (PDMS/TiO2)/PVDF composite membrane with a flux of 187.72 g·m-2·h-1, and separation factor of 11.25 at 50°C. This study states that pervaporation technology has a great potential to treat dilute formaldehyde solutions. This may lead to more research interests in the process of industrial formaldehyde wastewater treatment using pervaporation technology.
The authors would like to thank the National Natural Science Foundation of China (contract grant number 51403012), the State Key Laboratory of Organic–Inorganic Composites of Beijing University of Chemical Technology (contract grant number 22010006013), and the Fundamental Research Funds for the Central Universities (contract grant number buctrc201415) to fund this study.