Research Article - (2015) Volume 1, Issue 1
Mara Zeni*, Diana Favero, Kelvin Pacheco and Ana Grisa MC
Materials Chemistry Research Laboratory, Caxias do Sul University, Caxias do Sul
*Corresponding Author:
Mara Zeni
Materials Chemistry Research Laboratory
Caxias do Sul University
Caxias do Sul, RSBrazil
E-mail: mzandrad@ucs.br
Received Date: September 16, 2015; Accepted Date: October 15, 2015; Published Date: December 28, 2015
Citation: Zeni M, Favero D, Pacheco K. Preparation of Microcellulose (Mcc) and Nanocellulose (Ncc) from Eucalyptus Kraft Ssp Pulp. Polym Sci. 2015, 1:1.
Natural fibres have been attracted significant interest due to its renewable nature, its vast source of raw material, besides having good mechanical properties and low cost. The cellulose present in cell walls of plants can be purified and isolated, acting as a reinforcing agent in polymer composites, in order to obtain lighter, resistant and biodegradable composites. The techniques of acid hydrolysis are methods used to isolate crystals of cellulose, using different acids, resulting in different materials. In this study it was possible to verify the effect of the acid hydrolysis process, with hydrochloric acid (HCl) and sulfuric acid (H2SO4), resulting in MCC and NCC by one hydrolysis step, and then were analysed for its chemical and morphological characteristics, presenting significant improvements on these properties.
Keywords
Cellulose; Composites; Micro cellulose; Nano-cellulose; Hydrolysis; Fibers
Introduction
Cellulose is an abundant biopolymer in nature and the major constituent of plants. It can be considered an inexhaustible biodegradable source, presenting hydrophobicity, chirality, large capacity for chemical modification and can form versatile fibres with semi crystalline morphologies. It is a polymer characterized by crystalline regions interspersed by amorphous regions [1,2]. It is the predominant component in vegetable fibres and consist in a polymer comprising multiple units of β-D-glucose linked by β-1,4-glycosidic linkages, generating highly linear chains [3]. The cellulose presents crystalline structure and insolubility in water, while the rest of the components are easily permeable amorphous materials [3,4]. The amorphous regions are the result of malformation of the structure due to the change in the crystallization process. These are called, by some authors, as regions where crystallization occurred with defect [5,6]. The isolated crystalline domains of pulp fibres are known as cellulose Nano crystals [7]. The cellulose can lead to micro cellulose (MCC) and nano-cellulose (NCC), which are named crystalline cellulosic fibres, isolated by acid hydrolysis, and thus improve its performance as reinforcing particle in composites [8]. The natural disorders of cellulose molecules in the amorphous regions favour the accessibility of the acid and consequently the hydrolysis of cellulose chains in these regions. Therefore, the isolation of CNN is facilitated by faster hydrolysis kinetics displayed by amorphous regions compared to the crystalline regions [6]. The MCC has many properties, such as an aqueous insoluble derivative of cellulose, being a rod-shaped particle, which explain its vast application, especially in the food field. Currently it is marketed by Ceolus® and Avicel® in powder form, for use as a thickener in foods and drugs [9]. The interest in nano-materials is not only due to the improvements in performance properties of matrices, but also envisions the development of new products utilizing them as reinforcing materials. Besides the excellent mechanical properties provided by CNC compared to other nanoparticles, the advantage of presenting high specific surface area per mass of material makes them highly attractive when considering the phenomenon of solid-liquid interface [10,11]. Due to the good properties of MCC and NCC, they have attracted the attention of various fields like packaging, agriculture, food, automotive, aerospace, and other industries [12]. The isolation of the NCC using hydrolysis with H2SO4 was initially researched by Rånby [13], when a colloidal suspension was produced. Battista and Smith [1,2], achieved stable suspensions of these particles and proposed that it could be achieved by hydrolysis with HCl, followed by a mechanical treatment step [14]. The variables for cellulose hydrolysis conditions are: acid concentration, time, temperature and acid / feedstock ratio. A study by Araki et al. [15], with bleached Kraft pulp of conifer evaluated the effect of H2SO4 and HCl in the hydrolysis process over the dispersion properties of the CNC. However, the particles obtained from the treatment with H2SO4 had a negatively charged surface due to the introduction of sulphate groups. Moreover, by conduct metric titration of NCC obtained from the treatment with HCl, no surface charge was detected and may having size and thickness reductions [16]. The characterization of these nanomaterial is conducted to assess their dimensional characteristics, its surface charge, crystallinity, stability of the suspension and its performance in polymer matrix. In this article it is presented the acid hydrolysis of cellulose, from Kraft pulp of Eucalyptus ssp. with sulphuric acid (H2SO4) and hydrochloric acid (HCl) to obtain micro cellulose (MCC) and nano-cellulose (NCC), aiming to use the MCC and the NCC as reinforcement for polymeric composites.
Experimental Part
Materials
Eucalyptokraft pulp supplied by CMPC cellulose Riograndense - Guaiba / RS 37% hydrochloric acid (HCl) from Merck (Germany) and sulfuric acid (H2SO4) Merck (Germany).
Methods
Obtaining nano-cellulose (NCC) and micro cellulose (MCC) The obtain ion of NCC and MCC was performed in duplicate with HCl and H2SO4 acid. According to Ellazzouzi et al. and Wang et al. [17,18] the concentrations of the acids used were H2SO4 64% (w/w) and 37% HCl (w/w). 3 g of pulp was weighed and added to 13 mL.g-1 of HCl and 8,75 mL.g-1 of H2SO4, and then acid hydrolysis was carried out under vigorous and constant agitation for 30 min, in the temperature of 45°C. After the acid hydrolysis occurs, the dispersion is washed and made three cycles of centrifugation (Medifriger-BL) at 12,000 rpm. The final wash is done by dialysis in membrane of regenerated cellulose with distilled water until the pH achieves neutrality. The solution containing the supernatant was sonicated (Unique Ultra Clear 1400A) for 15 min, in order to obtain the NCC. The residue obtained (Figure 1) was filtered, stored in an petri plate and placed in an oven at 45°C, ground in cryogenic mill and screened through a sieve of 325 mensch, in order to obtain the MCC (Figure 1).
Figure 1: Supernatant solution with NCC and MCC residue, after the centrifugation process of the hydrolysed solutions of cellulose.
Characterization
Characterization techniques for Eucalyptus Kraft pulp and micro cellulose (MCC) were employed by:X-ray diffraction (XRD), infrared spectroscopy (FTIR) and scanning electron microscopy (SEM). The Nano cellulose (NCC) was characterized by transmission electron microscopy (TEM). In material characterization by X-ray diffraction, the samples were analysed with the emission of radiation of Cu Kα (λ=1.5406 Å) and angular step of 0.05°.Samples were analysed in the angular range of 5° to 40°, ±23°C temperature. The Eucalyptus Kraft pulp, MCC (HCl) and MCC (H2SO4) were analysed using Shimadzu XRD-6000. The morphology of micro celluloses (MCC) were analysed by SEM using the Supers can SSX- 550, Shimadzu equipment. The samples were carefully fractured in liquid nitrogen at-140°C and subjected to plating with gold by sputtering with an exposure time of 2 min to become conductive and the analysis was performed. Tabs KBr were prepared in the ratio 1:10 (fiber/KBr). The analyses were carried out 32 scans; in the range of 4000-500 cm-1.The equipment used was the FTIR - Nicolet IS10 - Thermo Scientific, using a transmission technique. The morphology of Nano celluloses (NCC) were analysed by TEM, using JEM-1200 EX equipment II-120 kV Electron Microscope. The samples were prepared with two drops of the NCC neutral Suspension, under a copper grid with Formvar (400 mensch-Ted Pella) film, then the grid was immersed in a 2% solution of uranyl acetate and remained drying by 24 hours.
Results and Discussions
Characterization of Eucalyptus Kraft pulp and micro cellulose (MCC) hydrolysed with HCl and H2SO4
The technique of X-ray diffraction was used in order to evaluate the degree of crystallinity of the samples (Figure 2). The XRD of Eucalyptus Kraft pulp is characteristic of cellulose type I, where the amorphous halo and the crystalline peak are located between the angles 18º≤2θ≤19º and 22º≤2θ≤23º. After the acid hydrolysis, it was observed the appearance of the characteristic peak from cellulose type II in the diffraction of MCC, situated between the angles 13°≤2θ≤15° and 18°≤2θ≤22° for both acids. When the cellulose I undergoes alkaline treatment, the reagent penetrates the crystal structure breaking the intermolecular bonds, where the chains are mixed to form alkaline cellulose, then occurring a rearrangement in the internal structure of the fibre to a different type of crystallinity, cellulose II [19,20]. With the formation of cellulose II, the crystalline structure is formed in three planes, where some of the intermolecular hydrogen bonds are not formed [17,21], which makes its free energy smaller and the bonds easier to broke. The parallel chains of cellulose I are linked mainly by Van der Waals forces. Consequently, the hydrophobic interaction is much weaker than in the cellulose II [22,23], which may be one of the most important reasons for breaking the chains during hydrolysis of cellulose II. The MCC diffraction indicated that the hydrolysis of MCC with HCl and H2SO4 was suitable for removing part of the amorphous material. Table 1 shows the crystallinity index of Eucalyptus Kraft pulp and MCC obtained by acid hydrolysis method. The acid hydrolysis aims to reduce the amorphous regions between micro fibrils while the crystalline segments remain intact because the kinetics of hydrolysis in the amorphous region is faster than the in the crystalline region, due to its higher permeability [6]. The crystalline region of the cellulose increases, so MCC crystallinity becomes higher than the pulp, indicating a partial removal of amorphous regions. The crystallinity of MCC generally lies between 65 and 83% [24], which range is linked to its origin as well as the hydrolysis conditions used in its production [25]. In the SEM micrographs (Figure 3) it can be observed the different morphologies between Eucalyptus Kraft pulp after the acid hydrolysis process with H2SO4 and HCl, to obtain the MCC (Figure 3). According to Araki et al. MCC hydrolysed with HCl does not have surface charge, may having size and thickness reductions [16]. MCC obtained with H2SO4 presents a lot of agglomerations, as viewed in the SEM (Table 2). The Eucalyptus Kraft pulp samples subjected to the acid hydrolysis method with HCl and H2SO4 had a reduction in the dimensions (length and diameter), though similar, and compared with MCC marketed by Ceolus® and Avicel® had lower diameter dimensions. It can be concluded that the hydrolysis occurred effectively in both techniques, with smaller diameter than the commercial MCC and the analysis of X-ray diffraction (XRD) shows an increase in the index of crystallinity of the MCC obtained from Eucalyptus Kraft pulp (Figure 4). The absorbance in the region of 897 cm-1 are assigned to β-glycoside linkages between β-glucose units in cellulose and hemicellulose which shows the evidence that the pulp has a high cellulose content. The absorbance in the region of 1060 cm-1 are assigned to the stretching of cyclical C-O bond of cellulose. It indicates functional groups secondary alcohol and ether [26]. The intensity of peaks is reduced after acid hydrolysis treatment, mainly due to removal of hemicelluloses [26,27]. The absorbance in the region of 1064 cm-1 suggest functional groups C=C and C=O due to slightly incomplete acid hydrolysis of cellulose [28].These bands are present in the spectra of the fibres probably due to the Kraft process. The peak at 1160 cm-1, which appears in all spectra’s, corresponds to the asymmetric stretching of C-O-C of hemicellulose and lignin. The absorbance between 3600 and 2800 cm-1 are due to stretching vibrations of C-H and O-H. Transmission Electron Microscopy (TEM) of the NCC obtained by the acid hydrolysis process with H2SO4 (Figure 5a) and HCl (Figure 5b). This tendency causes birefringence of the dispersion [18]. The NCC when incorporated into a material acts as reinforcement because of the increased crystallinity, which improves the material properties, such as mechanical properties, for example, the strength of the material. These microscopies are very characteristic of this type of system, presenting some clusters that are commonly observed due to the large surface area and strong interactions of hydrogen present in these systems [29] (Table 3). The NCC has diameter smaller than 100 nm [30] and the TEM micrographs show that Nano scale structures were obtained (Figure 5). In suspensions obtained by acid hydrolysis, it was observed that it showed large agglomeration of nanoparticles, especially when hydrolysis was carried out with HCl. In HCl hydrolysis it does not occur insertion of ionic groups on the nano-fiber structure, which despite presenting a weak surface charge between the two individual dipoles, the sum of attractive components of all electric dipoles of the particles results in a relatively high force of attraction, which results in the agglomeration of the particles [4]. The hydrolysis with H2SO4 generates stable aqueous suspensions of NCC, the hydroxyl groups of the surface of NCC are etherified with sulphate groups, stabilize the aqueous suspension by anionic repulsion forces [31-33] and tend not to aggregating. The aspect ratio of the NCC was higher than 10, which is considered the minimum value for which there is a good stress transfer from the matrix to the Nano fillers, generating a significant effect [34]. The averages of ratios aspects of NCC in this work are close to the values found for the NCC from wood, bagasse, golden grass and sisal [35-38] and are among the largest aspect ratios already found in the literature for NCC extracted from lignocellulose materials.
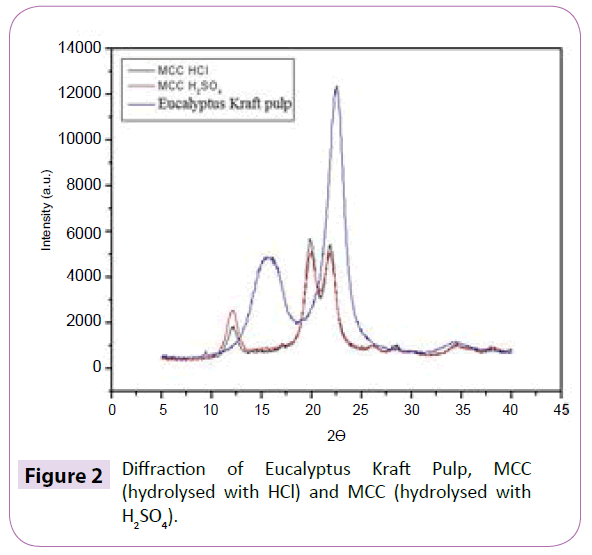
Figure 2: Diffraction of Eucalyptus Kraft Pulp, MCC (hydrolysed with HCl) and MCC (hydrolysed with H2SO4).
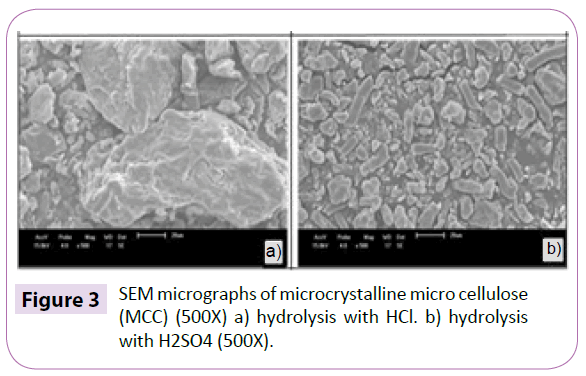
Figure 3: SEM micrographs of microcrystalline micro cellulose (MCC) (500X) a) hydrolysis with HCl. b) hydrolysis with H2SO4 (500X).

Figure 4: Infrared spectroscopy (FTIR) of the spectra’s of the pulp of Eucalyptus, MCC (HCl) and MCC (H2SO4).
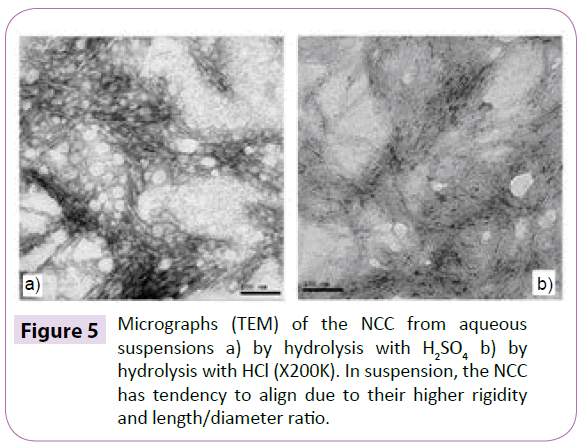
Figure 5: Micrographs (TEM) of the NCC from aqueous suspensions a) by hydrolysis with H2SO4 b) by hydrolysis with HCl (X200K). In suspension, the NCC has tendency to align due to their higher rigidity and length/diameter ratio.
Table 1: Index of crystallinity (%) of MCC.
| Sample | I eucalyptusc(%) |
|---|---|
| EucalyptusKraftPulp | 61.27 |
| MCC HCl | 74.66 |
| MCC H2SO4 | 75.51 |
Table 2: Relationship diameter and length of commercial microcellulose (MCC) and obtained by acid hydrolysis.
| Microceluloses | Avicel FD-101 | Avicel FD-101 | ST-01CM | MCC |
|---|---|---|---|---|
| (MCCs) | (Commercial) | (commercial) | (H2SO4)Eucalyptus | (HCl)Eucalyptus |
| Diameter | 40μm | 60μm | ± 13μm | ± 9μm |
| Length | ------ | ------ | ± 26 mM | ± 21μm |
Table 3: Nanocellulose (NCC) ratio diameter (D), width (W) and aspect ratio (L/D).
| Nanocelluloses (CNs) | L (length) (nm) | D (diameter) (nm) | L/D(aspect ratio) (nm) |
|---|---|---|---|
| NCC H2SO4 | 98.47 ± 3.8 | 3.0 ± 1.03 | 32.82 |
| NCC HCl | 90.11 ± 3.6 | 2.22 ± 1.05 | 40.6 |
Acknowledgments
The authors thank UCS, Compendia, Madeireira Gold Martini for supplying the samples of P. taeda and CNPq for the financial support.
Conclusions
The NCC and MCC can be isolated from Eucalyptus ssp. Kraft pulp and the hydrolysis conditions used resulted in stable aqueous suspensions. Characterization analyses showed that there was an increase incrystallinity index compared to MCC. The acid hydrolysis process with different acids HCl and H2SO4 were effective for obtaining NCC and MCC, with minor variations in their dimensions. The X-DR showed that the crystallinity was increased significantly to the MCC. The FTIR spectra showed the removal of hemicellulose and lignin. The morphological characterization of MCC, compared with commercial MCC from Avicel and Ceolus, showed lower diameter. The obtain ion of MCC and NCC from Eucalyptus Kraft pulp is an efficient alternative for the sustainable development of abundant natural resources in Brazil, in order to develop new products from renewable sources and low cost, adding value and creating wealth.