Commentary - (2017) Volume 3, Issue 1
Reiji Yoshimura*
Department of Psychiatry, University of Occupational and Environmental Health, Japan
*Corresponding Author:
Reiji Yoshimura
Department of Psychiatry
University of Occupational and Environmental Health
1-1 Iseigaoka, Yahatanishi-ku
Kitakyushu, Japan
Tel: +81936917253
E-mail: yoshi621@med.uoeh-u.ac.jp
Received Date: April 01, 2017; Accepted Date: April 06, 2017; Published Date: April 13, 2017
Citation: Yoshimura R. Plasma Interleukin 6 Predicts Response to Serotonin Noradrenaline Reuptake Inhibitors in Major Depressive Disorder. Biomark J. 2017, 3:1. doi: 10.21767/2472-1646.100027
Copyright: © 2017 Yoshimura R. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
We investigated the association between the plasma interleukin-6 (IL-6) level and response to Serotonin Noradrenaline Reuptake Inhibitors (SNRIs) in Major Depressive Disorder (MDD) patients. Patients with MDD diagnosed by DSM-IV-TR (M/F: 45/52, mean age 48 years) were enrolled in the present study. All participants were treated by monotherapy of duloxetine (n=48) or milnacipran (n=49). The end-point of the present study was week 8. Patients whose HAMD17 scores decreased 50% or more were defined as responders; others were considered nonresponders. Blood was drawn at baseline and 8 weeks after the treatment. Plasma levels of IL-6 were measured. Plasma IL6 at week 0 was higher in the responder group than in the nonresponders group. Plasma IL6 level in the responder group significantly decreased 8 weeks after treatment with antidepressant. These results suggest that plasma IL-6 is related to the response to SNRIs in MDD patients.
Keywords
Serotonin Noradrenaline Reuptake Inhibitors (SNRIs); Interleukin-6; Major depressive disorder; Plasma
Introduction
Selective Serotonin Reuptake Inhibitors (SSRIs) and Serotonin Noradrenaline Reuptake Inhibitors (RSNEIs) are the first-line treatment for Major Depressive Disorder (MDD). SSRIs work on the Serotonin Transporter (SET) while SNRIs work on both serotonin and noradrenaline transporters. Serotonin and noradrenaline may both play important roles in the pathophysiology of MDD, but the exact etiology of depression remains unknown. There is a growing body of evidence that pro-inflammatory cytokines are involved in the pathology of MDD. Activated microglia exhibit increased production of pro-inflammatory cytokines (e.g., Interleukin (IL)-1β, tumor Necrosis Factor-Alpha (TNF-α), and IL-6) [1]. Cytokines are known to induce changes in the central nervous system when injected peripherally, as well as when secreted internally in response to various conditions, such as an endotoxin challenge or in a model of stress. A recent study reported that overexpressed pro-inflammatory parameters are associated with suicidal ideation [2]. A recent meta-analysis reported significantly higher plasma concentrations of pro-inflammatory cytokines TNF-α and IL-6 in depressed subjects than in control subjects. It is well known that noradrenaline stimulates the release of cytokines from microglia and macrophages, and the effect of noradrenaline and cytokines is regulated by adrenoceptors. We also reported that the plasma levels of IL-6 are elevated during depressive periods [3-5]. According to a meta-analysis [6], treatment with antidepressants decreased serum IL-6 levels in patients with MDD. In other words, plasma IL-6 can serve as a state marker of major depressive episodes. Activation of the immune system has repeatedly been described in MDD, and it has been suggested that cytokines play a key role in this immune activation [3,7]. Within the brain, cytokines exert various effects on neurotransmitters, especially the noradrenaline and serotonin systems, which are thought to play a pivotal role in psychiatric disorders [8]. IL-6 receptors are widely distributed in the brain, especially in the hippocampus and hypothalamus [9]. Maes [10] demonstrated that the plasma concentrations of IL-6 were significantly higher in MDD subjects than in healthy controls. This finding was in accordance with a subsequent meta-analysis [11]. Taken together, these findings suggest that IL-6 might play a role in the pathogenesis of MDD. It may be surmised that IL-6 may play a role in the biology or somatic consequences of MDD. We hypothesized that plasma IL-6 predicts response to SNRIs. The aim of the present study was to investigate the association between the plasma IL-6 level and response to SNRIs in MDD patients.
Materials and Methods
Patients with MDD diagnosed by DSM-IV-TR were enrolled in the present study. All participants were treated by monotherapy of duloxetine (n=48) or milnacipran (n=49). Only benzodiazepines were permitted during the study periods. Patients had a mean age of 48 years and included 45 males and 52 females. The severity of the depressive state was evaluated with the 17-item of Hamilton Rating Scale for Depression (HAMD17) [12]. The end-point point of the present study was week 8. Patients whose HAMD17 scores decreased 50% or more were defined as responders; others were nonresponders. Blood was drawn at baseline and after 8 weeks of treatment. Plasma levels of IL-6 were measured according to the ELISA we previously described [5].
Statistical Analysis
The data were normally distributed using the Kolmogorov– Smirnov test. A paired or non-paired t-test was used to compare the plasma IL-6 levels and the HAMD17 scores between week 0 and week 8. The level of significance for all analyses was set at P<0.05.
Results
The demography and response rate of each antidepressant group are shown in Table 1. Plasma IL6 at week 0 was higher in the responder group than in the nonresponders group (p=0.013).
| SNRI | Duloxetine | Milnacipran |
|---|---|---|
| Number | 48 | 49 |
| Sex | 25/23 | 20/29 |
| Age (M/F) | 45 (19) | 49 (17) |
| Daily dose (mg) | 81 (14) | 38 (19) |
| HAMD17 (W0) | 25 (10) | 24 (13) |
| Response rare | 22/48 (45%) | 22/49 (45%) |
Table 1: Demographic data of each SNRI group.
The plasma IL6 level in the responder group was significantly lower after 8 weeks of treatment with the SNRI antidepressant (p=0.024). The plasma IL6 level in the nonresponders group did not change (p=0.861) (Figure 1).
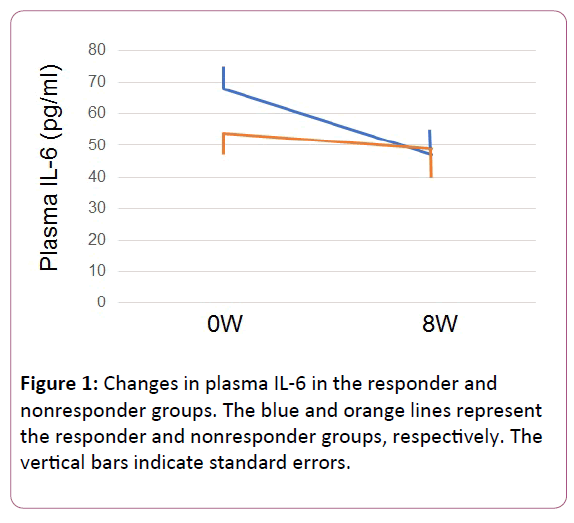
Figure 1: Changes in plasma IL-6 in the responder and nonresponder groups. The blue and orange lines represent the responder and nonresponder groups, respectively. The vertical bars indicate standard errors.
Discussion
The main finding of the present study was that in MDD patients treated with SNRIs, initial plasma IL-6 levels were higher in the responder group than in the nonresponder group. We also demonstrated that after 8 weeks of SNRI treatment, responders showed reduced plasma IL-6 levels but nonresponders did not. Previously, we reported that baseline plasma IL-6 levels were significantly higher in responders to paroxetine or sertraline than in nonresponders. We reconfirmed in the present study that plasma IL-6 is a predicting biomarker for response to SNRIs as well as SSRIs in MDD patients. IL-6 is proposed to be involved in the pathophysiology of MDD. In particular, it was suggested that IL-6 is involved in multiple physiological systems, including the hypothalamic-pituitary-adrenal axis, corticotrophin-releasing hormone activity at limbic sites, noradrenaline utilization, the release of oxidative stress, apoptotic pathways, and kinase signaling, all of which have very close relationships to the pathophysiology of MDD [13-15]. Li et al. recently showed that noradrenaline significantly increased the mRNA and protein expressions of IL-6 in U937 macrophages in time- and concentration-dependent manners [16]. The β-adrenoreceptor inhibitor propranolol blocked noradrenaline-induced IL-6 expression in U937 macrophages at both the mRNA and protein levels; hyper-noradrenergic activity is known to promote IL-6 production. Kong et al. demonstrated that IL-6 directly regulates serotonin transporter levels and consequently serotonin reuptake, and identified STAT3- dependent regulation of the IL-6/serotonin transporter system [17]. Serotonin also regulates IL-6 secretion through the activation of the 5-HT2B receptor and the ERK1/2 signaling pathway [18]. In short, an interaction exists among noradrenergic systems, serotonergic systems, and IL-6.
Microglia is the primary central reservoirs of pro-inflammatory cytokines, and they act as antigen-presenting cells in the brain [19]. Although it is uncertain whether peripheral IL-6 levels represent the levels found in the brain, evidence suggests that IL-6 crosses the blood–brain barrier, and that the peripheral level might represent an overflow from the central nervous system and may be a marker of an inflammatory process in the brain [20].
One limitation of the present study was that the sample size was small and heterogeneous, and lacked a reference group, which might be associated with a type I or type II error. The fact that the dosage of SNRIs was not fixed is another problem. Thus, further research using a larger sample with a reference group, and using different antidepressants, are needed to test the preliminary results achieved in this study. In conclusion, the plasma IL-6 level may be a predictor for response to SNRIs in MDD patients.
Conflict of Interest
The author confirms that this article content has no conflict of interest.
Acknowledgements
The study was supported by a Health and Labor Research grant (#1401010101).